Key Insights
The Biologics Contract Development and Manufacturing Organization (CDMO) market is experiencing robust growth, projected to reach \$15.32 billion in 2025 and exhibiting a Compound Annual Growth Rate (CAGR) of 12.78% from 2025 to 2033. This expansion is driven by several key factors. Firstly, the increasing demand for biologics, particularly biosimilars, across various therapeutic areas like oncology, immunology, and infectious diseases, fuels the need for specialized CDMO services. Secondly, the rising adoption of advanced technologies such as single-use systems and continuous manufacturing processes enhances efficiency and reduces production costs, further stimulating market growth. Finally, the outsourcing trend among pharmaceutical and biotechnology companies, driven by cost optimization and focus on core competencies, significantly contributes to the market's expansion. The market is segmented by type (mammalian and non-mammalian/microbial), product type (biologics and biosimilars), and geography. While precise regional market shares are not provided, it is reasonable to expect that North America and Europe will hold significant shares, given their established biopharmaceutical industries and regulatory frameworks. Asia Pacific is expected to witness substantial growth, driven by increasing investments in research and development and a growing biologics market.
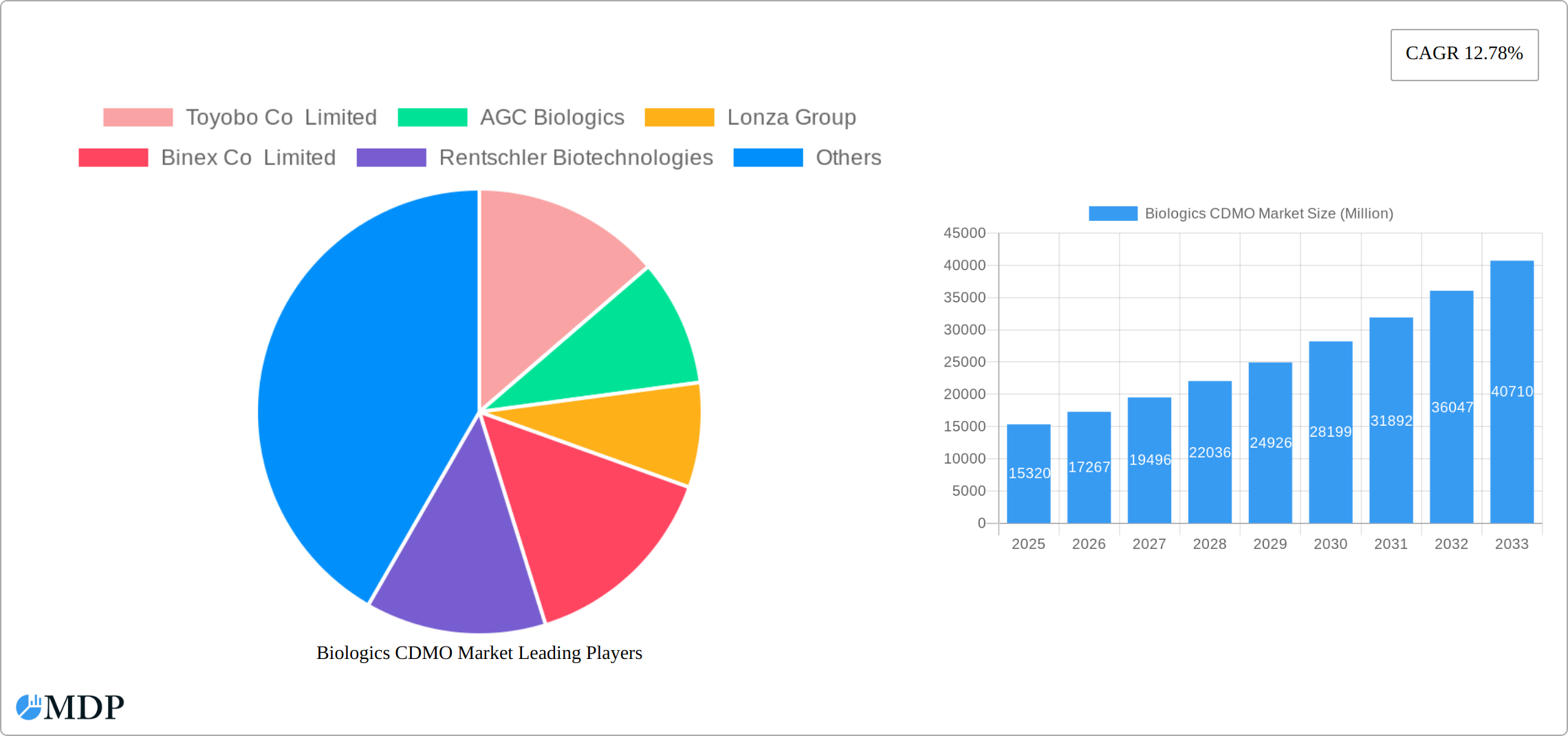
Biologics CDMO Market Market Size (In Billion)

Key players like Samsung Biologics, Lonza, Wuxi Biologics, and Catalent are prominent in this competitive landscape, constantly investing in capacity expansion and technological advancements to meet the burgeoning demand. The market's growth is, however, subject to certain restraints, including stringent regulatory approvals for biologics and the complex manufacturing processes involved. Despite these challenges, the overall outlook for the Biologics CDMO market remains highly positive, indicating sustained growth throughout the forecast period. The increasing prevalence of chronic diseases, advancements in biotechnology, and the growing demand for personalized medicine are expected to further propel market expansion in the coming years.
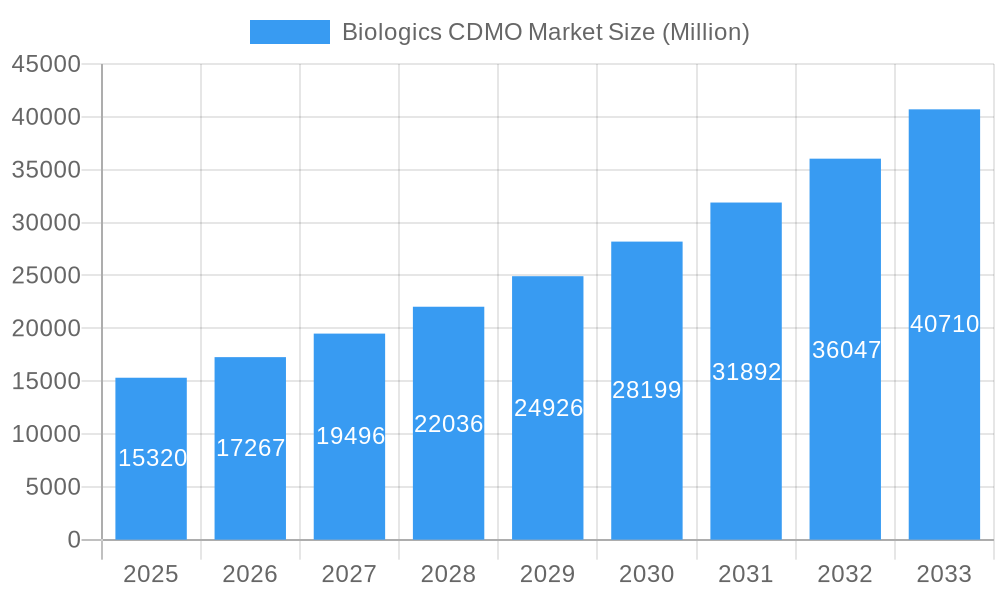
Biologics CDMO Market Company Market Share

Biologics CDMO Market Report: A Comprehensive Analysis (2019-2033)
This comprehensive report provides an in-depth analysis of the Biologics CDMO market, offering invaluable insights for stakeholders, investors, and industry professionals. With a detailed examination of market dynamics, leading players, and future trends, this report serves as an essential resource for navigating the complexities of this rapidly evolving sector. The report covers the period 2019-2033, with a base year of 2025 and a forecast period of 2025-2033. The market is segmented by type (Mammalian, Non-mammalian (Microbial)), product type (Biologics, Biosimilars), and other relevant factors. Expected market value estimations will be provided throughout the report.
Biologics CDMO Market Market Dynamics & Concentration
The Biologics Contract Development and Manufacturing Organization (CDMO) market is a dynamic and evolving landscape, shaped by a confluence of technological advancements, regulatory considerations, and strategic industry movements. Market concentration is currently assessed as moderate to high, with a discernible presence of large, established players alongside a growing number of specialized and emerging CDMOs. Mergers and acquisitions (M&A) are a significant force in this sector, driving consolidation and enabling companies to expand their capabilities, geographical reach, and service offerings. The market is fueled by several key growth drivers:
- Technological Advancements: Continuous innovation is paramount. This includes breakthroughs in cell line development for enhanced protein expression, process intensification for higher yields and reduced footprint, and the widespread adoption of single-use technologies for greater flexibility and reduced contamination risks.
- Evolving Regulatory Frameworks: The stringent and ever-changing global regulatory landscape, encompassing quality control, Good Manufacturing Practices (GMP), and validation requirements, significantly influences market dynamics. CDMOs must continually invest in robust quality systems and regulatory expertise to ensure compliance and facilitate client product approvals.
- Therapeutic Innovation and Substitutes: While biologics offer distinct therapeutic advantages for complex diseases, the emergence of novel treatment modalities and the increasing maturity of the biosimilar market present both opportunities and challenges, demanding adaptability and specialized expertise from CDMOs.
- Surging End-User Demand: The escalating global prevalence of chronic and rare diseases is a primary driver, leading to a sustained and growing demand for sophisticated biologic therapeutics from pharmaceutical and biotechnology companies.
- Strategic M&A Activity: The high volume of M&A demonstrates a clear trend towards industry consolidation. We observe a substantial number of M&A transactions occurring within the 2019-2024 period, reshaping market share and competitive positioning. Larger CDMOs are actively pursuing acquisitions to gain access to specialized technologies, acquire new clients, expand their manufacturing capacities, and enhance their end-to-end service portfolios. Smaller, agile companies are often acquired to leverage their niche expertise or proprietary technologies.
Biologics CDMO Market Industry Trends & Analysis
The Biologics CDMO market is on a trajectory of robust expansion, with a projected Compound Annual Growth Rate (CAGR) of approximately [Insert Specific CAGR Here]% during the forecast period of 2025-2033. This impressive growth is propelled by a synergistic interplay of several critical factors:
The increasing incidence of chronic and complex diseases worldwide is a fundamental driver, directly translating into a higher demand for advanced biologic treatments. Concurrent with this, significant technological advancements are revolutionizing manufacturing processes. The widespread adoption of single-use technologies, coupled with increasing automation, is significantly boosting operational efficiency, reducing lead times, and driving down manufacturing costs. Furthermore, a pronounced trend of outsourcing among pharmaceutical and biotechnology companies, driven by the need for cost optimization, enhanced manufacturing flexibility, and access to specialized expertise, is creating substantial opportunities for CDMOs. Evolving consumer preferences for highly effective and safe therapeutic options further underscore the demand for high-quality biologic products. The competitive landscape is characterized by intense rivalry, compelling leading players to make substantial investments in capacity expansion, technological upgrades, and strategic partnerships to secure and maintain their market leadership. Market penetration is anticipated to deepen considerably as outsourcing continues to rise and the demand for sophisticated biological manufacturing capabilities grows.
Leading Markets & Segments in Biologics CDMO Market
North America currently commands the largest share in the Biologics CDMO market, followed closely by Europe. However, the Asia-Pacific region is emerging as a powerhouse of rapid growth, fueled by substantial investments in healthcare infrastructure and a rapidly expanding pharmaceutical industry. This growth is further bolstered by increasing government initiatives aimed at fostering domestic biopharmaceutical manufacturing.
-
Dominant Segment: The mammalian cell-based biologics segment remains the largest and most significant contributor to the market. This dominance is attributed to the inherent ability of mammalian cell lines to produce complex therapeutic proteins with intricate post-translational modifications, which are crucial for efficacy.
-
Key Drivers by Region:
- North America: Characterized by its well-established and robust regulatory pathways, a highly developed pharmaceutical and biotechnology ecosystem, and substantial investment in research and development activities.
- Europe: Benefits from significant and ongoing investments in healthcare infrastructure, adherence to stringent quality and safety standards, and a large, diverse patient population driving demand for advanced therapies.
- Asia-Pacific: Driven by rapid economic expansion, increasing government support and incentives for the biopharmaceutical sector, and a vast and growing population that presents a significant market opportunity.
A granular analysis of country-level market dominance within each major region, detailing specific economic policies and infrastructure development initiatives that propel growth in individual nations, will be provided in the comprehensive market report. The biosimilars segment is also witnessing accelerated growth, a trend largely driven by the increasing expiration of patents for blockbuster biologics and a growing global emphasis on affordable healthcare solutions.
Biologics CDMO Market Product Developments
Recent product innovations focus on enhancing manufacturing efficiency, reducing costs, and improving product quality. Advancements in single-use technologies, continuous manufacturing processes, and process analytical technologies (PAT) are transforming the industry. These innovations offer competitive advantages by accelerating development timelines and reducing manufacturing costs, thus increasing market fit and customer adoption. The market continues to see an increasing adoption of these technologically advanced methods.
Key Drivers of Biologics CDMO Market Growth
Technological advancements (automation, single-use systems), rising prevalence of chronic diseases, increasing outsourcing by pharmaceutical companies, and supportive regulatory frameworks all contribute to market growth. For example, the adoption of continuous manufacturing significantly reduces production time and costs.
Challenges in the Biologics CDMO Market Market
The Biologics CDMO market, despite its promising outlook, navigates a complex set of challenges. Prominent among these are stringent and often lengthy regulatory hurdles, which can lead to significant project delays and escalated compliance costs. Furthermore, the intricate global supply chains for biologics are susceptible to disruptions, potentially causing production delays and impacting overall costs. The highly competitive nature of the market necessitates continuous innovation, operational efficiency improvements, and strategic differentiation. These challenges, if not effectively managed, could collectively impact the market's revenue potential by an estimated [Insert Specific Revenue Impact Here] Million annually.
Emerging Opportunities in Biologics CDMO Market
Emerging opportunities arise from technological breakthroughs in cell and gene therapies, increasing strategic partnerships between CDMOs and pharmaceutical companies, and expanding into new geographic markets. These opportunities present significant potential for long-term market growth.
Leading Players in the Biologics CDMO Market Sector
- Toyobo Co Limited
- AGC Biologics
- Lonza Group
- Binex Co Limited
- Rentschler Biotechnologies
- Wuxi Biologics
- AbbVie Contract Manufacturing
- Parexel International Corporation
- Sandoz Biopharmaceuticals (Novartis AG)
- Catalent Inc
- JRS Pharma
- Fujifilm Diosynth Biotechnologies USA Inc
- Samsung Biologics
- Boehringer Ingelheim Group
- Icon PLC
Key Milestones in Biologics CDMO Market Industry
- December 2021: AstraZeneca and Samsung Biologics formed a strategic biopharmaceutical manufacturing partnership, expanding their collaboration to include a cancer immunotherapy product worth approximately USD 380 Million.
- March 2022: Oasmia Pharmaceutical AB and Lonza signed a large-scale manufacturing agreement for Cantrixil, an investigational drug candidate.
- April 2022: FUJIFILM Corporation completed the acquisition of a cell therapy manufacturing facility from Atara Biotherapeutics Inc., expanding its global CDMO network.
Strategic Outlook for Biologics CDMO Market Market
The Biologics CDMO market is poised for substantial and sustained long-term growth. This optimistic outlook is underpinned by continuous technological advancements, the escalating trend of outsourcing by biopharmaceutical companies, and the ever-expanding global demand for sophisticated biologic therapeutics. Strategic opportunities abound for CDMOs that focus on specialized therapeutic areas, invest proactively in cutting-edge manufacturing technologies such as advanced cell culture techniques and continuous manufacturing, and cultivate strong, long-term strategic partnerships to secure reliable contract streams. The market is expected to continue its expansion, with forward-thinking players prioritizing innovation, strategic collaborations, and a commitment to quality and regulatory excellence to thrive in this dynamic and competitive ecosystem.
Biologics CDMO Market Segmentation
-
1. Type
- 1.1. Mammalian
- 1.2. Non-mammalian (Microbial)
-
2. Product Type
-
2.1. Biologics
- 2.1.1. Monoclon
- 2.1.2. Recombinant Proteins
- 2.1.3. Antisense and Molecular Therapy
- 2.1.4. Vaccines
- 2.1.5. Other Biologics
- 2.2. Biosimilars
-
2.1. Biologics
Biologics CDMO Market Segmentation By Geography
- 1. North America
- 2. Europe
- 3. Asia
- 4. Australia and New Zealand
- 5. Latin America
- 6. Middle East and Africa
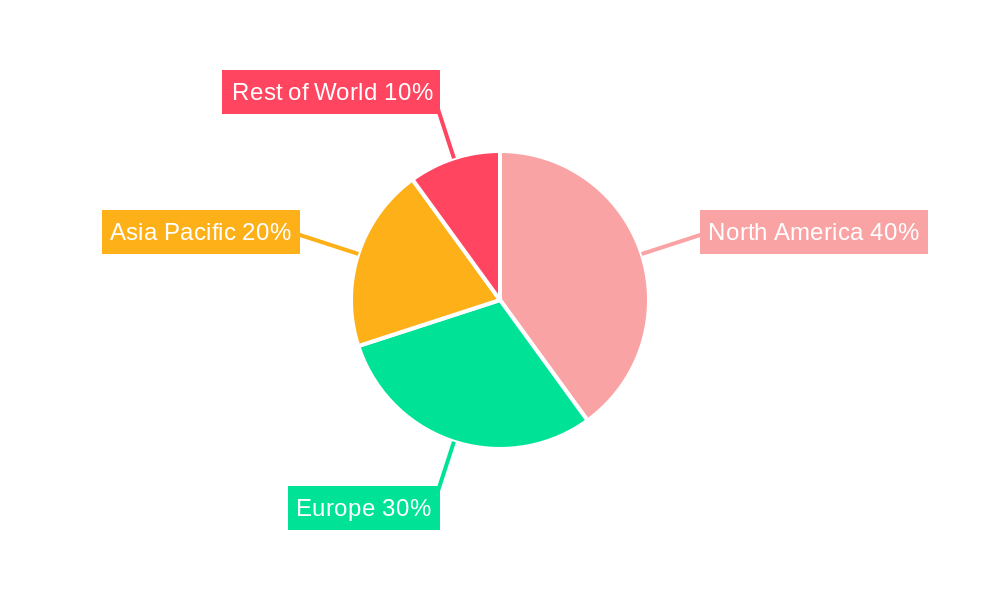
Biologics CDMO Market Regional Market Share

Geographic Coverage of Biologics CDMO Market
Biologics CDMO Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 12.78% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. MDP Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Type
- 5.1.1. Mammalian
- 5.1.2. Non-mammalian (Microbial)
- 5.2. Market Analysis, Insights and Forecast - by Product Type
- 5.2.1. Biologics
- 5.2.1.1. Monoclon
- 5.2.1.2. Recombinant Proteins
- 5.2.1.3. Antisense and Molecular Therapy
- 5.2.1.4. Vaccines
- 5.2.1.5. Other Biologics
- 5.2.2. Biosimilars
- 5.2.1. Biologics
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. Europe
- 5.3.3. Asia
- 5.3.4. Australia and New Zealand
- 5.3.5. Latin America
- 5.3.6. Middle East and Africa
- 5.1. Market Analysis, Insights and Forecast - by Type
- 6. Global Biologics CDMO Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Type
- 6.1.1. Mammalian
- 6.1.2. Non-mammalian (Microbial)
- 6.2. Market Analysis, Insights and Forecast - by Product Type
- 6.2.1. Biologics
- 6.2.1.1. Monoclon
- 6.2.1.2. Recombinant Proteins
- 6.2.1.3. Antisense and Molecular Therapy
- 6.2.1.4. Vaccines
- 6.2.1.5. Other Biologics
- 6.2.2. Biosimilars
- 6.2.1. Biologics
- 6.1. Market Analysis, Insights and Forecast - by Type
- 7. North America Biologics CDMO Market Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Type
- 7.1.1. Mammalian
- 7.1.2. Non-mammalian (Microbial)
- 7.2. Market Analysis, Insights and Forecast - by Product Type
- 7.2.1. Biologics
- 7.2.1.1. Monoclon
- 7.2.1.2. Recombinant Proteins
- 7.2.1.3. Antisense and Molecular Therapy
- 7.2.1.4. Vaccines
- 7.2.1.5. Other Biologics
- 7.2.2. Biosimilars
- 7.2.1. Biologics
- 7.1. Market Analysis, Insights and Forecast - by Type
- 8. Europe Biologics CDMO Market Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Type
- 8.1.1. Mammalian
- 8.1.2. Non-mammalian (Microbial)
- 8.2. Market Analysis, Insights and Forecast - by Product Type
- 8.2.1. Biologics
- 8.2.1.1. Monoclon
- 8.2.1.2. Recombinant Proteins
- 8.2.1.3. Antisense and Molecular Therapy
- 8.2.1.4. Vaccines
- 8.2.1.5. Other Biologics
- 8.2.2. Biosimilars
- 8.2.1. Biologics
- 8.1. Market Analysis, Insights and Forecast - by Type
- 9. Asia Biologics CDMO Market Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Type
- 9.1.1. Mammalian
- 9.1.2. Non-mammalian (Microbial)
- 9.2. Market Analysis, Insights and Forecast - by Product Type
- 9.2.1. Biologics
- 9.2.1.1. Monoclon
- 9.2.1.2. Recombinant Proteins
- 9.2.1.3. Antisense and Molecular Therapy
- 9.2.1.4. Vaccines
- 9.2.1.5. Other Biologics
- 9.2.2. Biosimilars
- 9.2.1. Biologics
- 9.1. Market Analysis, Insights and Forecast - by Type
- 10. Australia and New Zealand Biologics CDMO Market Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Type
- 10.1.1. Mammalian
- 10.1.2. Non-mammalian (Microbial)
- 10.2. Market Analysis, Insights and Forecast - by Product Type
- 10.2.1. Biologics
- 10.2.1.1. Monoclon
- 10.2.1.2. Recombinant Proteins
- 10.2.1.3. Antisense and Molecular Therapy
- 10.2.1.4. Vaccines
- 10.2.1.5. Other Biologics
- 10.2.2. Biosimilars
- 10.2.1. Biologics
- 10.1. Market Analysis, Insights and Forecast - by Type
- 11. Latin America Biologics CDMO Market Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Type
- 11.1.1. Mammalian
- 11.1.2. Non-mammalian (Microbial)
- 11.2. Market Analysis, Insights and Forecast - by Product Type
- 11.2.1. Biologics
- 11.2.1.1. Monoclon
- 11.2.1.2. Recombinant Proteins
- 11.2.1.3. Antisense and Molecular Therapy
- 11.2.1.4. Vaccines
- 11.2.1.5. Other Biologics
- 11.2.2. Biosimilars
- 11.2.1. Biologics
- 11.1. Market Analysis, Insights and Forecast - by Type
- 12. Middle East and Africa Biologics CDMO Market Analysis, Insights and Forecast, 2020-2032
- 12.1. Market Analysis, Insights and Forecast - by Type
- 12.1.1. Mammalian
- 12.1.2. Non-mammalian (Microbial)
- 12.2. Market Analysis, Insights and Forecast - by Product Type
- 12.2.1. Biologics
- 12.2.1.1. Monoclon
- 12.2.1.2. Recombinant Proteins
- 12.2.1.3. Antisense and Molecular Therapy
- 12.2.1.4. Vaccines
- 12.2.1.5. Other Biologics
- 12.2.2. Biosimilars
- 12.2.1. Biologics
- 12.1. Market Analysis, Insights and Forecast - by Type
- 13. Competitive Analysis
- 13.1. Company Profiles
- 13.1.1 Toyobo Co Limited
- 13.1.1.1. Company Overview
- 13.1.1.2. Products
- 13.1.1.3. Company Financials
- 13.1.1.4. SWOT Analysis
- 13.1.2 AGC Biologics
- 13.1.2.1. Company Overview
- 13.1.2.2. Products
- 13.1.2.3. Company Financials
- 13.1.2.4. SWOT Analysis
- 13.1.3 Lonza Group
- 13.1.3.1. Company Overview
- 13.1.3.2. Products
- 13.1.3.3. Company Financials
- 13.1.3.4. SWOT Analysis
- 13.1.4 Binex Co Limited
- 13.1.4.1. Company Overview
- 13.1.4.2. Products
- 13.1.4.3. Company Financials
- 13.1.4.4. SWOT Analysis
- 13.1.5 Rentschler Biotechnologies
- 13.1.5.1. Company Overview
- 13.1.5.2. Products
- 13.1.5.3. Company Financials
- 13.1.5.4. SWOT Analysis
- 13.1.6 Wuxi Biologics
- 13.1.6.1. Company Overview
- 13.1.6.2. Products
- 13.1.6.3. Company Financials
- 13.1.6.4. SWOT Analysis
- 13.1.7 AbbVie Contract Manufacturing*List Not Exhaustive
- 13.1.7.1. Company Overview
- 13.1.7.2. Products
- 13.1.7.3. Company Financials
- 13.1.7.4. SWOT Analysis
- 13.1.8 Parexel International Corporation
- 13.1.8.1. Company Overview
- 13.1.8.2. Products
- 13.1.8.3. Company Financials
- 13.1.8.4. SWOT Analysis
- 13.1.9 Sandoz Biopharmaceuticals (Novartis AG)
- 13.1.9.1. Company Overview
- 13.1.9.2. Products
- 13.1.9.3. Company Financials
- 13.1.9.4. SWOT Analysis
- 13.1.10 Catalent Inc
- 13.1.10.1. Company Overview
- 13.1.10.2. Products
- 13.1.10.3. Company Financials
- 13.1.10.4. SWOT Analysis
- 13.1.11 JRS Pharma
- 13.1.11.1. Company Overview
- 13.1.11.2. Products
- 13.1.11.3. Company Financials
- 13.1.11.4. SWOT Analysis
- 13.1.12 Fujifilm Diosynth Biotechnologies USA Inc
- 13.1.12.1. Company Overview
- 13.1.12.2. Products
- 13.1.12.3. Company Financials
- 13.1.12.4. SWOT Analysis
- 13.1.13 Samsung Biologics
- 13.1.13.1. Company Overview
- 13.1.13.2. Products
- 13.1.13.3. Company Financials
- 13.1.13.4. SWOT Analysis
- 13.1.14 Boehringer Ingelheim Group
- 13.1.14.1. Company Overview
- 13.1.14.2. Products
- 13.1.14.3. Company Financials
- 13.1.14.4. SWOT Analysis
- 13.1.15 Icon PLC
- 13.1.15.1. Company Overview
- 13.1.15.2. Products
- 13.1.15.3. Company Financials
- 13.1.15.4. SWOT Analysis
- 13.1.1 Toyobo Co Limited
- 13.2. Market Entropy
- 13.2.1 Company's Key Areas Served
- 13.2.2 Recent Developments
- 13.3. Company Market Share Analysis 2025
- 13.3.1 Top 5 Companies Market Share Analysis
- 13.3.2 Top 3 Companies Market Share Analysis
- 13.4. List of Potential Customers
- 14. Research Methodology
List of Figures
- Figure 1: Global Biologics CDMO Market Revenue Breakdown (Million, %) by Region 2025 & 2033
- Figure 2: North America Biologics CDMO Market Revenue (Million), by Type 2025 & 2033
- Figure 3: North America Biologics CDMO Market Revenue Share (%), by Type 2025 & 2033
- Figure 4: North America Biologics CDMO Market Revenue (Million), by Product Type 2025 & 2033
- Figure 5: North America Biologics CDMO Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 6: North America Biologics CDMO Market Revenue (Million), by Country 2025 & 2033
- Figure 7: North America Biologics CDMO Market Revenue Share (%), by Country 2025 & 2033
- Figure 8: Europe Biologics CDMO Market Revenue (Million), by Type 2025 & 2033
- Figure 9: Europe Biologics CDMO Market Revenue Share (%), by Type 2025 & 2033
- Figure 10: Europe Biologics CDMO Market Revenue (Million), by Product Type 2025 & 2033
- Figure 11: Europe Biologics CDMO Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 12: Europe Biologics CDMO Market Revenue (Million), by Country 2025 & 2033
- Figure 13: Europe Biologics CDMO Market Revenue Share (%), by Country 2025 & 2033
- Figure 14: Asia Biologics CDMO Market Revenue (Million), by Type 2025 & 2033
- Figure 15: Asia Biologics CDMO Market Revenue Share (%), by Type 2025 & 2033
- Figure 16: Asia Biologics CDMO Market Revenue (Million), by Product Type 2025 & 2033
- Figure 17: Asia Biologics CDMO Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 18: Asia Biologics CDMO Market Revenue (Million), by Country 2025 & 2033
- Figure 19: Asia Biologics CDMO Market Revenue Share (%), by Country 2025 & 2033
- Figure 20: Australia and New Zealand Biologics CDMO Market Revenue (Million), by Type 2025 & 2033
- Figure 21: Australia and New Zealand Biologics CDMO Market Revenue Share (%), by Type 2025 & 2033
- Figure 22: Australia and New Zealand Biologics CDMO Market Revenue (Million), by Product Type 2025 & 2033
- Figure 23: Australia and New Zealand Biologics CDMO Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 24: Australia and New Zealand Biologics CDMO Market Revenue (Million), by Country 2025 & 2033
- Figure 25: Australia and New Zealand Biologics CDMO Market Revenue Share (%), by Country 2025 & 2033
- Figure 26: Latin America Biologics CDMO Market Revenue (Million), by Type 2025 & 2033
- Figure 27: Latin America Biologics CDMO Market Revenue Share (%), by Type 2025 & 2033
- Figure 28: Latin America Biologics CDMO Market Revenue (Million), by Product Type 2025 & 2033
- Figure 29: Latin America Biologics CDMO Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 30: Latin America Biologics CDMO Market Revenue (Million), by Country 2025 & 2033
- Figure 31: Latin America Biologics CDMO Market Revenue Share (%), by Country 2025 & 2033
- Figure 32: Middle East and Africa Biologics CDMO Market Revenue (Million), by Type 2025 & 2033
- Figure 33: Middle East and Africa Biologics CDMO Market Revenue Share (%), by Type 2025 & 2033
- Figure 34: Middle East and Africa Biologics CDMO Market Revenue (Million), by Product Type 2025 & 2033
- Figure 35: Middle East and Africa Biologics CDMO Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 36: Middle East and Africa Biologics CDMO Market Revenue (Million), by Country 2025 & 2033
- Figure 37: Middle East and Africa Biologics CDMO Market Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Biologics CDMO Market Revenue Million Forecast, by Type 2020 & 2033
- Table 2: Global Biologics CDMO Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 3: Global Biologics CDMO Market Revenue Million Forecast, by Region 2020 & 2033
- Table 4: Global Biologics CDMO Market Revenue Million Forecast, by Type 2020 & 2033
- Table 5: Global Biologics CDMO Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 6: Global Biologics CDMO Market Revenue Million Forecast, by Country 2020 & 2033
- Table 7: Global Biologics CDMO Market Revenue Million Forecast, by Type 2020 & 2033
- Table 8: Global Biologics CDMO Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 9: Global Biologics CDMO Market Revenue Million Forecast, by Country 2020 & 2033
- Table 10: Global Biologics CDMO Market Revenue Million Forecast, by Type 2020 & 2033
- Table 11: Global Biologics CDMO Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 12: Global Biologics CDMO Market Revenue Million Forecast, by Country 2020 & 2033
- Table 13: Global Biologics CDMO Market Revenue Million Forecast, by Type 2020 & 2033
- Table 14: Global Biologics CDMO Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 15: Global Biologics CDMO Market Revenue Million Forecast, by Country 2020 & 2033
- Table 16: Global Biologics CDMO Market Revenue Million Forecast, by Type 2020 & 2033
- Table 17: Global Biologics CDMO Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 18: Global Biologics CDMO Market Revenue Million Forecast, by Country 2020 & 2033
- Table 19: Global Biologics CDMO Market Revenue Million Forecast, by Type 2020 & 2033
- Table 20: Global Biologics CDMO Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 21: Global Biologics CDMO Market Revenue Million Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Biologics CDMO Market?
The projected CAGR is approximately 12.78%.
2. Which companies are prominent players in the Biologics CDMO Market?
Key companies in the market include Toyobo Co Limited, AGC Biologics, Lonza Group, Binex Co Limited, Rentschler Biotechnologies, Wuxi Biologics, AbbVie Contract Manufacturing*List Not Exhaustive, Parexel International Corporation, Sandoz Biopharmaceuticals (Novartis AG), Catalent Inc, JRS Pharma, Fujifilm Diosynth Biotechnologies USA Inc, Samsung Biologics, Boehringer Ingelheim Group, Icon PLC.
3. What are the main segments of the Biologics CDMO Market?
The market segments include Type, Product Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 15.32 Million as of 2022.
5. What are some drivers contributing to market growth?
Access to New Technologies and Higher Speed of Execution Realized by CDMOs; Need for High Capital Investments to Develop Capabilities Has Led to Firms Choosing the Outsourcing Model; Lack of In-house Capacity among Emerging Drug Development Companies.
6. What are the notable trends driving market growth?
CDMOs’ Access to New Technologies and Higher Speed of Execution Driving Market Growth.
7. Are there any restraints impacting market growth?
Presence of Alternative Printing Technology.
8. Can you provide examples of recent developments in the market?
April 2022 - FUJIFILM Corporation announced that it had completed the acquisition of a dedicated cell therapy manufacturing facility from Atara Biotherapeutics Inc. The facility, located in Thousand Oaks, California, will be operated as part of FUJIFILM DiosynthBiotechnologies' global network, a subsidiary of FUJIFILM Corporation and a world-leading contract development and manufacturing organization (CDMO).
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Biologics CDMO Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Biologics CDMO Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Biologics CDMO Market?
To stay informed about further developments, trends, and reports in the Biologics CDMO Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


