Key Insights
The global Pharmaceutical Data Management Software market is poised for significant expansion, projected to reach approximately USD 38,820 million by 2025, driven by an impressive Compound Annual Growth Rate (CAGR) of 6.4% from 2019 to 2033. This robust growth is fundamentally fueled by the escalating complexity and volume of data generated throughout the pharmaceutical lifecycle. Key drivers include the increasing demand for efficient clinical trial management, the imperative for stringent regulatory compliance (e.g., GDPR, HIPAA), and the growing adoption of big data analytics for drug discovery, development, and post-market surveillance. Furthermore, the digital transformation within the pharmaceutical industry, emphasizing real-time data accessibility and improved decision-making, is a critical catalyst. The trend towards cloud-based solutions is accelerating due to their scalability, cost-effectiveness, and enhanced accessibility, while on-premise solutions continue to cater to organizations with specific security or regulatory needs.
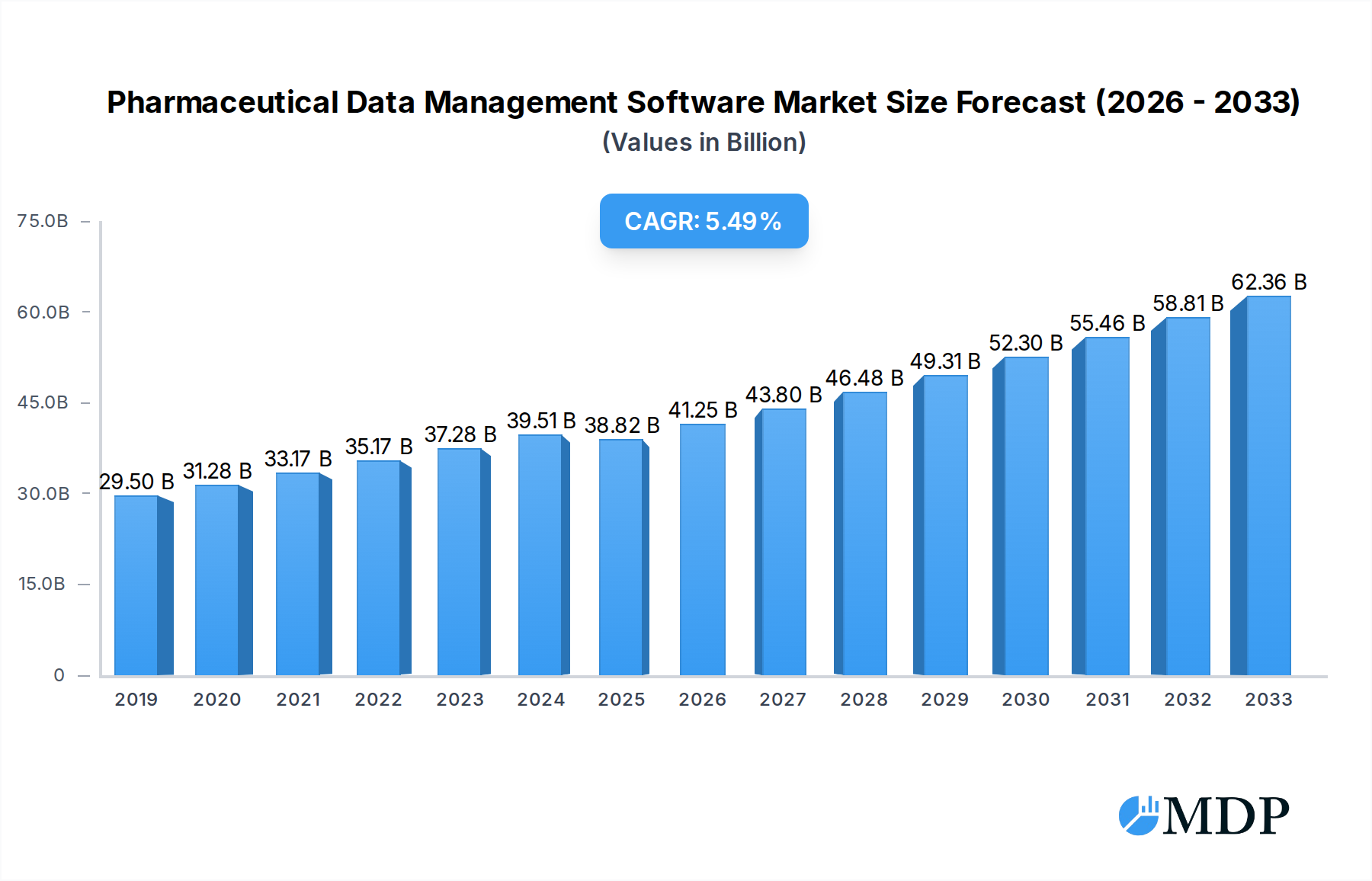
Pharmaceutical Data Management Software Market Size (In Billion)

The market is segmented across various applications, with Laboratory Data Management and Pharmaceutical Supply Chain Data Management emerging as dominant segments, reflecting the industry's focus on R&D efficiency and product integrity. Clinical Data Management also holds substantial importance as regulatory bodies increasingly scrutinize trial data. While the market benefits from strong growth drivers, it faces certain restraints. These include the high initial investment costs for advanced software solutions, concerns regarding data security and privacy, and the challenge of integrating disparate data sources and legacy systems. Addressing these challenges through robust cybersecurity measures, interoperability solutions, and strategic partnerships will be crucial for sustained market development. Key players are actively investing in R&D and strategic acquisitions to enhance their offerings and expand their global footprint, particularly in high-growth regions like Asia Pacific and North America.
Pharmaceutical Data Management Software Company Market Share

This comprehensive report delves into the dynamic Pharmaceutical Data Management Software market, providing in-depth analysis and actionable insights for industry stakeholders. Spanning from 2019 to 2033, with a focus on the base and estimated year of 2025 and a forecast period of 2025–2033, this report offers an unparalleled understanding of market drivers, trends, competitive landscapes, and future opportunities. Discover how leading companies are leveraging advanced data management solutions to accelerate drug discovery, streamline clinical trials, and optimize supply chains. This report is essential for pharmaceutical companies, software providers, investors, and regulatory bodies seeking to navigate this critical sector.
Pharmaceutical Data Management Software Market Dynamics & Concentration
The Pharmaceutical Data Management Software market is characterized by a moderate to high concentration, with key players like Medidata, Oracle, IBM, and PAREXEL Informatics holding significant market shares, estimated in the hundreds of millions of dollars each. Innovation remains a primary driver, fueled by the increasing complexity of clinical trials and the growing need for real-time data analysis. Regulatory frameworks, such as GDPR and FDA guidelines, also play a pivotal role in shaping software development and adoption, ensuring data integrity and patient privacy. Product substitutes exist in the form of generic data handling tools, but specialized pharmaceutical solutions offer superior functionality. End-user trends indicate a strong preference for cloud-based solutions due to their scalability and accessibility, with an estimated 70% of new implementations leaning towards cloud. Mergers and acquisitions (M&A) activities are moderately prevalent, with an estimated 20 to 30 major deals annually, valued in the hundreds of millions, aimed at consolidating market positions and expanding technological capabilities.
Pharmaceutical Data Management Software Industry Trends & Analysis
The pharmaceutical data management software industry is poised for robust growth, driven by the escalating need for efficient and compliant data handling throughout the drug lifecycle. The Compound Annual Growth Rate (CAGR) is projected to be approximately 12% over the forecast period, indicating a substantial expansion. Market penetration is steadily increasing, reaching an estimated 75% for core clinical data management solutions. Technological disruptions, such as the integration of Artificial Intelligence (AI) and Machine Learning (ML) for predictive analytics and anomaly detection, are transforming how data is processed and interpreted. These advancements empower researchers to identify potential drug candidates faster and optimize trial designs, leading to significant cost savings estimated in the hundreds of millions annually per major pharmaceutical company. Consumer preferences are shifting towards integrated platforms that offer seamless data flow across different applications, from laboratory data management to pharmaceutical supply chain data management. This demand is pushing vendors to develop comprehensive suites rather than standalone solutions. The competitive dynamics are intense, with established players investing heavily in R&D and new entrants offering niche, innovative solutions. The increasing volume and complexity of real-world evidence (RWE) further propel the demand for sophisticated data management tools, enabling pharmaceutical companies to demonstrate drug efficacy and safety more effectively. The global market for pharmaceutical data management software is estimated to reach over 50,000 million dollars by 2025.
Leading Markets & Segments in Pharmaceutical Data Management Software
The Clinical Data Management segment is the dominant force within the pharmaceutical data management software market, accounting for an estimated 55% of the total market share. This dominance is driven by the intricate requirements of clinical trials, including data capture, cleaning, validation, and reporting, which are critical for regulatory submissions and drug approvals. North America, particularly the United States, remains the leading geographical market, contributing an estimated 35% to global revenue. This is attributed to a strong pharmaceutical R&D ecosystem, a high concentration of major pharmaceutical companies, and favorable regulatory environments that encourage technological adoption.
- Key Drivers for Clinical Data Management Dominance:
- Increasing number and complexity of clinical trials globally.
- Stringent regulatory requirements for data accuracy and integrity.
- Growing emphasis on real-world evidence (RWE) collection and analysis.
- Demand for faster trial timelines and reduced operational costs.
The Cloud-Based Software type is rapidly gaining traction, projected to capture over 60% of the market by 2025, supplanting traditional on-premise solutions. The scalability, flexibility, and cost-effectiveness of cloud platforms are particularly appealing to pharmaceutical companies of all sizes, from large enterprises to smaller biotechs.
- Key Drivers for Cloud-Based Software Adoption:
- Reduced IT infrastructure costs and maintenance overhead.
- Enhanced accessibility and collaboration for geographically dispersed teams.
- Faster deployment and easier updates compared to on-premise solutions.
- Improved disaster recovery and business continuity capabilities.
While Laboratory Data Management and Pharmaceutical Supply Chain Data Management are crucial segments, their combined market share currently stands at approximately 35%, with the remaining 10% attributed to ‘Others’ encompassing areas like pharmacovigilance data management. The growth in these segments is steady, propelled by the need for end-to-end visibility and data-driven decision-making across the entire pharmaceutical value chain.
Pharmaceutical Data Management Software Product Developments
Recent product developments in pharmaceutical data management software are centered on enhancing data integration, AI-powered analytics, and user experience. Companies are introducing advanced modules for real-time data monitoring, predictive analytics for trial outcomes, and streamlined data submission capabilities. Innovations in blockchain technology are also emerging, promising increased data security and traceability. Competitive advantages are being built around specialized functionalities for niche therapeutic areas, interoperability with diverse data sources, and robust compliance features. The market is witnessing a trend towards unified platforms that consolidate various data management needs, reducing silos and improving operational efficiency, with an estimated R&D investment of several hundred million dollars annually by leading players.
Key Drivers of Pharmaceutical Data Management Software Growth
The pharmaceutical data management software market is experiencing significant growth, fueled by several key factors. The relentless pace of drug discovery and development necessitates efficient handling of vast datasets, driving the adoption of advanced data management solutions. Regulatory mandates, such as enhanced data integrity requirements and the increasing focus on real-world evidence (RWE), are compelling companies to invest in robust data management systems, estimated to drive market growth by over 10% annually. Furthermore, the economic imperative to reduce clinical trial costs and accelerate time-to-market is pushing pharmaceutical organizations towards scalable and efficient data management platforms. Technological advancements, including AI and machine learning, are unlocking new capabilities for data analysis and predictive modeling, further spurring growth.
Challenges in the Pharmaceutical Data Management Software Market
Despite its growth, the pharmaceutical data management software market faces several challenges. Data security and privacy remain paramount concerns, with the constant threat of cyberattacks and stringent regulatory compliance, such as HIPAA and GDPR, requiring substantial investment in security infrastructure. The integration of disparate data sources and legacy systems can be complex and time-consuming, leading to implementation delays and increased costs, estimated to add 10-15% to project budgets. Furthermore, the high cost of specialized pharmaceutical data management software can be a barrier for smaller companies, and the availability of skilled personnel to manage these sophisticated systems is also a limiting factor.
Emerging Opportunities in Pharmaceutical Data Management Software
Emerging opportunities in the pharmaceutical data management software market are ripe for exploration, driven by technological breakthroughs and strategic market expansion. The increasing adoption of AI and ML for drug discovery and clinical trial optimization presents a significant growth catalyst, enabling predictive analytics and personalized medicine approaches. Strategic partnerships between software vendors and pharmaceutical companies, as well as collaborations with academic institutions, are fostering innovation and accelerating the development of tailored solutions. Furthermore, the growing demand for data management solutions in emerging markets, particularly in Asia-Pacific, offers substantial opportunities for global expansion and market penetration. The potential for leveraging real-world data for post-market surveillance and health economics outcomes research is also a significant area of future growth, estimated to unlock billions in value.
Leading Players in the Pharmaceutical Data Management Software Sector
Medidata Oracle IBM PAREXEL Informatics BioClinica Medrio Forte Research Systems Taimei Technology Ofni Systems Fortress Medical Systems OpenClinica ClinCapture Logan Data Fortra Thermo Fisher Scientific Axtria Bentham Science
Key Milestones in Pharmaceutical Data Management Software Industry
- 2019: Increased adoption of cloud-based EDC (Electronic Data Capture) systems, marking a shift from traditional paper-based methods.
- 2020: Heightened demand for remote monitoring and data management solutions due to the global pandemic, accelerating digital transformation.
- 2021: Growing integration of AI and ML for advanced data analytics and predictive modeling in clinical trials.
- 2022: Increased focus on data interoperability standards and the development of unified data platforms.
- 2023: Emergence of blockchain technology for enhanced data security and traceability in pharmaceutical supply chains.
- 2024: Significant investments in real-world data (RWD) management and analytics capabilities.
Strategic Outlook for Pharmaceutical Data Management Software Market
The strategic outlook for the Pharmaceutical Data Management Software market remains exceptionally positive, driven by the continuous evolution of the pharmaceutical industry and the increasing reliance on data-driven decision-making. Future growth will be propelled by further integration of AI and ML for sophisticated predictive analytics, real-time data insights, and personalized medicine initiatives, with an estimated market expansion of over 15% annually. Strategic partnerships and M&A activities will continue to shape the competitive landscape, fostering innovation and market consolidation. The expansion into emerging markets and the development of specialized solutions for niche therapeutic areas will also be key growth accelerators, ensuring the continued dynamism and significant value creation within this vital sector.
Pharmaceutical Data Management Software Segmentation
-
1. Application
- 1.1. Laboratory Data Management
- 1.2. Pharmaceutical Supply Chain Data Management
- 1.3. Clinical Data Management
- 1.4. Others
-
2. Type
- 2.1. On-premise Software
- 2.2. Cloud-Based Software
Pharmaceutical Data Management Software Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
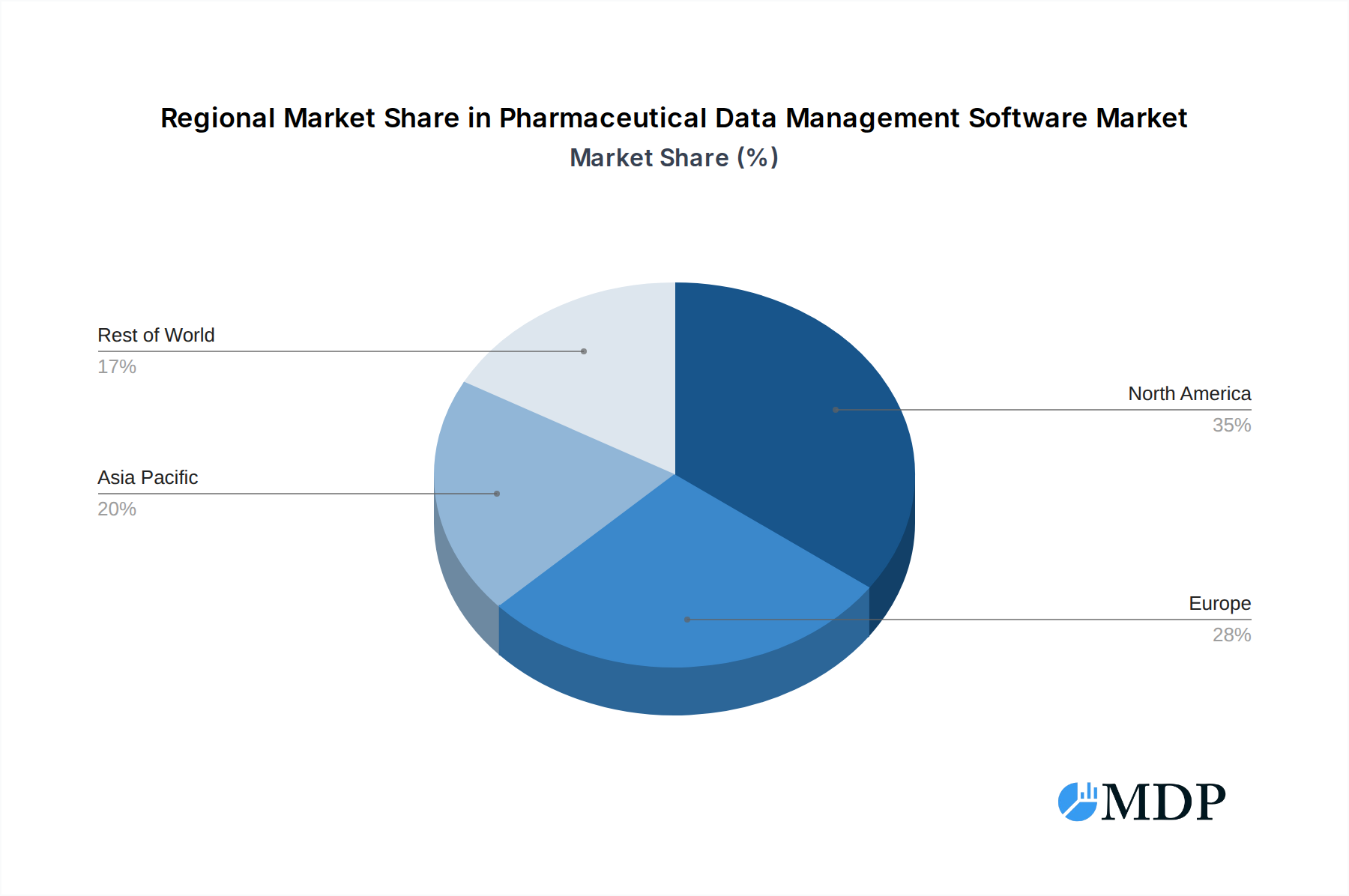
Pharmaceutical Data Management Software Regional Market Share

Geographic Coverage of Pharmaceutical Data Management Software
Pharmaceutical Data Management Software REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Pharmaceutical Data Management Software Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Laboratory Data Management
- 5.1.2. Pharmaceutical Supply Chain Data Management
- 5.1.3. Clinical Data Management
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. On-premise Software
- 5.2.2. Cloud-Based Software
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Pharmaceutical Data Management Software Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Laboratory Data Management
- 6.1.2. Pharmaceutical Supply Chain Data Management
- 6.1.3. Clinical Data Management
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. On-premise Software
- 6.2.2. Cloud-Based Software
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Pharmaceutical Data Management Software Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Laboratory Data Management
- 7.1.2. Pharmaceutical Supply Chain Data Management
- 7.1.3. Clinical Data Management
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. On-premise Software
- 7.2.2. Cloud-Based Software
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Pharmaceutical Data Management Software Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Laboratory Data Management
- 8.1.2. Pharmaceutical Supply Chain Data Management
- 8.1.3. Clinical Data Management
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. On-premise Software
- 8.2.2. Cloud-Based Software
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Pharmaceutical Data Management Software Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Laboratory Data Management
- 9.1.2. Pharmaceutical Supply Chain Data Management
- 9.1.3. Clinical Data Management
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. On-premise Software
- 9.2.2. Cloud-Based Software
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Pharmaceutical Data Management Software Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Laboratory Data Management
- 10.1.2. Pharmaceutical Supply Chain Data Management
- 10.1.3. Clinical Data Management
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. On-premise Software
- 10.2.2. Cloud-Based Software
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Medidata
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Oracle
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 IBM
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 PAREXEL Informatics
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 BioClinica
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Medrio
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Forte Research Systems
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Taimei Technology
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Ofni Systems
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Fortress Medical Systems
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 OpenClinica
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 ClinCapture
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Logan Data
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Fortra
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Thermo Fisher Scientific
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Axtria
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 Bentham Science
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.1 Medidata
List of Figures
- Figure 1: Global Pharmaceutical Data Management Software Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Pharmaceutical Data Management Software Revenue (million), by Application 2025 & 2033
- Figure 3: North America Pharmaceutical Data Management Software Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Pharmaceutical Data Management Software Revenue (million), by Type 2025 & 2033
- Figure 5: North America Pharmaceutical Data Management Software Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Pharmaceutical Data Management Software Revenue (million), by Country 2025 & 2033
- Figure 7: North America Pharmaceutical Data Management Software Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Pharmaceutical Data Management Software Revenue (million), by Application 2025 & 2033
- Figure 9: South America Pharmaceutical Data Management Software Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Pharmaceutical Data Management Software Revenue (million), by Type 2025 & 2033
- Figure 11: South America Pharmaceutical Data Management Software Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Pharmaceutical Data Management Software Revenue (million), by Country 2025 & 2033
- Figure 13: South America Pharmaceutical Data Management Software Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Pharmaceutical Data Management Software Revenue (million), by Application 2025 & 2033
- Figure 15: Europe Pharmaceutical Data Management Software Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Pharmaceutical Data Management Software Revenue (million), by Type 2025 & 2033
- Figure 17: Europe Pharmaceutical Data Management Software Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Pharmaceutical Data Management Software Revenue (million), by Country 2025 & 2033
- Figure 19: Europe Pharmaceutical Data Management Software Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Pharmaceutical Data Management Software Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa Pharmaceutical Data Management Software Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Pharmaceutical Data Management Software Revenue (million), by Type 2025 & 2033
- Figure 23: Middle East & Africa Pharmaceutical Data Management Software Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Pharmaceutical Data Management Software Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa Pharmaceutical Data Management Software Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Pharmaceutical Data Management Software Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific Pharmaceutical Data Management Software Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Pharmaceutical Data Management Software Revenue (million), by Type 2025 & 2033
- Figure 29: Asia Pacific Pharmaceutical Data Management Software Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Pharmaceutical Data Management Software Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific Pharmaceutical Data Management Software Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Pharmaceutical Data Management Software Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Pharmaceutical Data Management Software Revenue million Forecast, by Type 2020 & 2033
- Table 3: Global Pharmaceutical Data Management Software Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Pharmaceutical Data Management Software Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Pharmaceutical Data Management Software Revenue million Forecast, by Type 2020 & 2033
- Table 6: Global Pharmaceutical Data Management Software Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Pharmaceutical Data Management Software Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Pharmaceutical Data Management Software Revenue million Forecast, by Type 2020 & 2033
- Table 12: Global Pharmaceutical Data Management Software Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global Pharmaceutical Data Management Software Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global Pharmaceutical Data Management Software Revenue million Forecast, by Type 2020 & 2033
- Table 18: Global Pharmaceutical Data Management Software Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Pharmaceutical Data Management Software Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Pharmaceutical Data Management Software Revenue million Forecast, by Type 2020 & 2033
- Table 30: Global Pharmaceutical Data Management Software Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global Pharmaceutical Data Management Software Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global Pharmaceutical Data Management Software Revenue million Forecast, by Type 2020 & 2033
- Table 39: Global Pharmaceutical Data Management Software Revenue million Forecast, by Country 2020 & 2033
- Table 40: China Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Pharmaceutical Data Management Software Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Pharmaceutical Data Management Software?
The projected CAGR is approximately 6.4%.
2. Which companies are prominent players in the Pharmaceutical Data Management Software?
Key companies in the market include Medidata, Oracle, IBM, PAREXEL Informatics, BioClinica, Medrio, Forte Research Systems, Taimei Technology, Ofni Systems, Fortress Medical Systems, OpenClinica, ClinCapture, Logan Data, Fortra, Thermo Fisher Scientific, Axtria, Bentham Science.
3. What are the main segments of the Pharmaceutical Data Management Software?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 38820 million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Pharmaceutical Data Management Software," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Pharmaceutical Data Management Software report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Pharmaceutical Data Management Software?
To stay informed about further developments, trends, and reports in the Pharmaceutical Data Management Software, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


