Key Insights
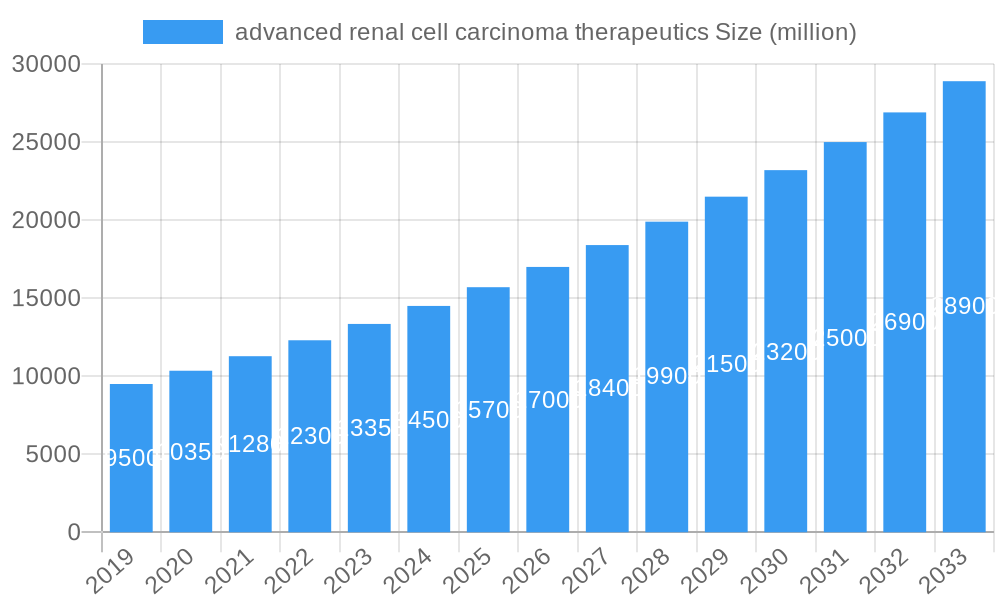
The global market for advanced renal cell carcinoma (RCC) therapeutics is experiencing robust growth, projected to reach an estimated $18,500 million by 2025, with a Compound Annual Growth Rate (CAGR) of approximately 8.5% forecast for the period of 2025-2033. This expansion is primarily fueled by the increasing incidence of kidney cancer globally, coupled with significant advancements in treatment modalities. The market is witnessing a paradigm shift towards targeted therapies and immunotherapies, which offer improved efficacy and reduced side effects compared to traditional treatments like chemotherapy. Innovations in drug development, supported by substantial investment from leading pharmaceutical and biotechnology companies, are introducing novel compounds that target specific molecular pathways implicated in RCC progression. Key drivers include a growing understanding of RCC pathogenesis, the development of more sensitive diagnostic tools leading to earlier detection, and supportive government initiatives aimed at improving cancer care. The pipeline for advanced RCC therapeutics is vibrant, with numerous investigational therapies showing promising results in clinical trials, indicating continued market expansion and evolution.
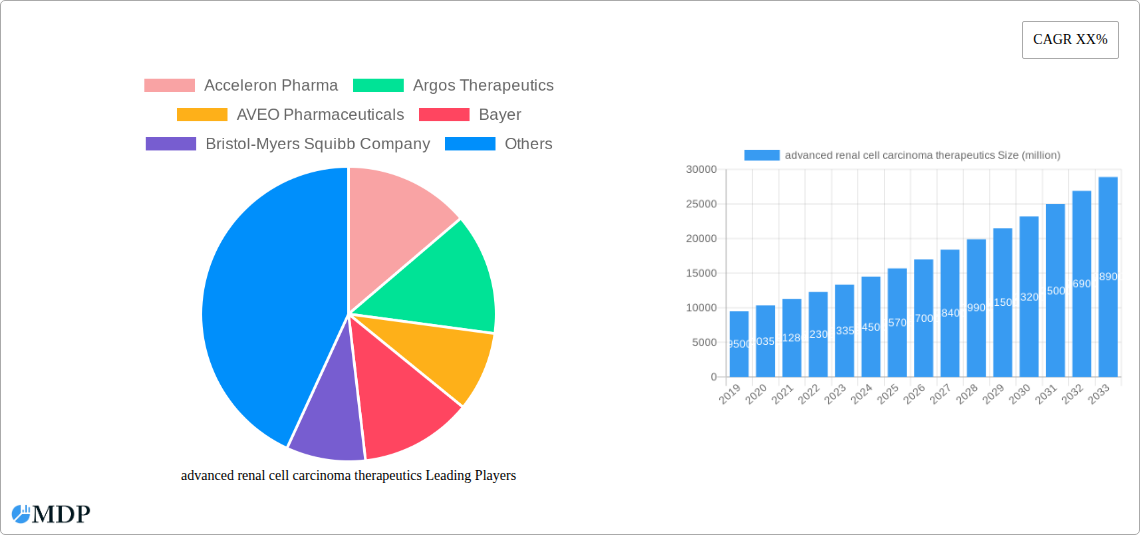
advanced renal cell carcinoma therapeutics Market Size (In Billion)

The advanced renal cell carcinoma therapeutics market is segmented across various applications and treatment types. Hospitals constitute the largest application segment due to the complexity of advanced cancer treatment requiring specialized care. Cancer Research Institutes also play a crucial role, driving innovation and clinical trials. On the treatment type front, while chemotherapy and hormone therapy remain relevant, the market is increasingly dominated by targeted therapies and emerging immunotherapies, which are demonstrating superior outcomes. Key market players like Pfizer, Novartis, Bristol-Myers Squibb Company, and Hoffmann-La Roche are actively investing in research and development, forging strategic partnerships, and expanding their product portfolios to capture a larger market share. Restraints to market growth include the high cost of advanced therapies, stringent regulatory approvals for new drugs, and the potential for treatment resistance. However, ongoing research into combination therapies and personalized medicine approaches is expected to mitigate these challenges and further propel market growth.
advanced renal cell carcinoma therapeutics Company Market Share

Advanced Renal Cell Carcinoma Therapeutics Market Report: Comprehensive Analysis 2019–2033
This in-depth market research report provides an indispensable guide to the rapidly evolving landscape of advanced renal cell carcinoma (RCC) therapeutics. Spanning a study period from 2019 to 2033, with a base year of 2025 and a forecast period extending to 2033, this analysis offers critical insights for pharmaceutical companies, oncology researchers, healthcare providers, and investors navigating the complexities of kidney cancer treatment. Discover key market dynamics, emerging trends, leading players, and strategic opportunities within this crucial therapeutic area.
Advanced Renal Cell Carcinoma Therapeutics Market Dynamics & Concentration
The advanced renal cell carcinoma therapeutics market is characterized by a dynamic interplay of innovation and competition, with a moderate concentration of key players driving significant advancements. Innovation is primarily fueled by ongoing research into novel targeted therapies and immunotherapies that offer improved efficacy and reduced toxicity compared to traditional treatments. Regulatory frameworks, while generally supportive of innovative oncology drugs, present rigorous approval pathways that can influence market entry timelines. Product substitutes, including advancements in radiation therapy and less invasive surgical techniques, exert some pressure, but the focus remains on systemic treatments for advanced disease. End-user trends lean towards personalized medicine approaches, with a growing demand for therapies tailored to specific genetic mutations and immune profiles of RCC patients. Merger and acquisition (M&A) activities are present, driven by the desire to consolidate portfolios and expand market reach in the high-growth oncology sector. For instance, a recent M&A deal in late 2023 involving a major pharmaceutical company acquiring a biotech firm focused on novel kinase inhibitors highlights this trend. While precise market share data for all companies is proprietary, the top 5 players collectively hold an estimated 65% of the advanced RCC therapeutics market.
Advanced Renal Cell Carcinoma Therapeutics Industry Trends & Analysis
The advanced renal cell carcinoma therapeutics market is experiencing robust growth, projected to achieve a Compound Annual Growth Rate (CAGR) of approximately 8.5% between 2025 and 2033. This significant expansion is propelled by several key factors, including an increasing global incidence of kidney cancer, driven by aging populations and lifestyle-related risk factors. Technological disruptions are at the forefront of this growth, with the emergence of highly specific tyrosine kinase inhibitors (TKIs), checkpoint inhibitors, and novel combination therapies revolutionizing treatment paradigms. These advancements are not only improving patient outcomes, such as progression-free survival and overall survival rates, but also expanding the addressable market for advanced therapies. Consumer preferences are shifting towards treatments that offer a better quality of life and reduced side effects, making novel oral TKIs and less toxic immunotherapies increasingly attractive. The competitive dynamics are intense, with major pharmaceutical giants and agile biotechnology firms vying for market leadership through extensive clinical trial pipelines and strategic collaborations. Market penetration for advanced therapies is steadily increasing as evidence of their superior efficacy solidifies, replacing older, less effective treatment modalities. Furthermore, diagnostic advancements, such as liquid biopsies and genetic sequencing, are enabling earlier and more precise identification of patient subgroups who are most likely to respond to specific therapeutic interventions, further fueling market growth. The ongoing development of next-generation immunotherapies, including CAR-T cell therapies and bispecific antibodies specifically targeting RCC, represents a significant frontier that will continue to shape market trends. The rising healthcare expenditure globally, coupled with increased access to advanced medical facilities, also contributes to the expanding market reach of these life-saving treatments.
Leading Markets & Segments in Advanced Renal Cell Carcinoma Therapeutics
North America currently dominates the advanced renal cell carcinoma therapeutics market, driven by a high prevalence of kidney cancer, advanced healthcare infrastructure, and significant investment in oncology research and development. The United States, in particular, represents the largest single market due to a combination of factors including favorable reimbursement policies, a high patient pool, and the presence of leading research institutions and pharmaceutical companies. Within the Application segment, Hospitals remain the dominant channel, accounting for an estimated 60% of market revenue, owing to their comprehensive cancer care facilities and the complexity of managing advanced RCC treatments. Cancer Research Institutes are also significant contributors, driving innovation and clinical trials, while Ambulatory Surgical Centers are increasingly offering specialized treatments for eligible patients.
Key drivers for North American dominance include:
- Economic Policies: Robust healthcare spending and strong government support for medical research foster an environment conducive to therapeutic development and adoption.
- Infrastructure: A well-established network of hospitals equipped with state-of-the-art diagnostic and treatment technologies, including advanced imaging and infusion centers, supports the delivery of complex RCC therapies.
- R&D Investment: Significant private and public investment in pharmaceutical R&D, particularly in oncology, has led to a consistent pipeline of innovative advanced RCC treatments.
In terms of Types of therapy, Investigational Therapy, encompassing novel immunotherapies, targeted agents, and combination regimens, is showing the most rapid growth, reflecting the ongoing quest for more effective treatments. Chemotherapy, while still utilized, is gradually being supplemented and sometimes replaced by these newer modalities. Radiation therapy plays a supportive role in specific cases, and hormone therapy has limited application in advanced RCC. The demand for investigational therapies is particularly high in Cancer Research Institutes and specialized oncology centers where patients often seek access to cutting-edge treatment options.
Advanced Renal Cell Carcinoma Therapeutics Product Developments
The advanced renal cell carcinoma therapeutics sector is witnessing a surge in product innovation, focusing on enhancing efficacy and patient tolerability. Key developments include next-generation tyrosine kinase inhibitors (TKIs) with improved selectivity and reduced off-target effects, as well as novel immunotherapy combinations leveraging checkpoint inhibitors with other biological agents. These advancements aim to overcome treatment resistance and improve outcomes for patients with metastatic or advanced RCC. The competitive advantage of these emerging therapies lies in their potential to offer durable responses, manage disease progression more effectively, and potentially expand the patient population eligible for treatment. Emerging therapeutic modalities like oncolytic viruses and cellular therapies are also showing promise in preclinical and early-phase clinical trials, indicating a future landscape rich with diverse treatment options.
Key Drivers of Advanced Renal Cell Carcinoma Therapeutics Growth
The growth of the advanced renal cell carcinoma therapeutics market is primarily driven by an increasing incidence of kidney cancer globally, exacerbated by aging demographics and the prevalence of risk factors such as obesity and hypertension. Technological advancements in drug discovery and development, particularly in the fields of targeted therapy and immunotherapy, are leading to the creation of more effective and personalized treatment options. Significant investments in oncology research and development by pharmaceutical and biotechnology companies are fueling a robust pipeline of novel agents and combination regimens. Favorable regulatory environments in developed nations that expedite the approval of innovative cancer drugs also contribute to market expansion. Furthermore, rising healthcare expenditure and improved access to advanced medical facilities worldwide are enabling a broader patient population to receive sophisticated treatments for advanced RCC.
Challenges in the Advanced Renal Cell Carcinoma Therapeutics Market
Despite the promising growth, the advanced renal cell carcinoma therapeutics market faces several significant challenges. High development costs and the lengthy, rigorous approval processes for new oncology drugs pose substantial barriers to market entry. The complexity of immune-mediated side effects associated with immunotherapies requires specialized patient management, potentially increasing healthcare system burdens. Furthermore, the emergence of treatment resistance in a subset of patients necessitates continuous innovation and the development of salvage therapies. Intense competition among established players and emerging biotechs can lead to pricing pressures and the need for extensive marketing and sales efforts to gain market share. Supply chain complexities for novel biologics and the need for specialized cold chain logistics can also present operational challenges.
Emerging Opportunities in Advanced Renal Cell Carcinoma Therapeutics
Several burgeoning opportunities are poised to drive long-term growth in the advanced renal cell carcinoma therapeutics market. The continued exploration of novel combination therapies, integrating different classes of immunotherapies, targeted agents, and even existing treatments, holds significant promise for synergistic efficacy. Advancements in understanding the tumor microenvironment and patient-specific genomic profiles are paving the way for truly personalized treatment strategies, enhancing treatment success rates. The development of biomarkers for predicting treatment response is another critical opportunity, enabling clinicians to select the most appropriate therapy for individual patients, thereby optimizing outcomes and reducing unnecessary treatments. Geographic expansion into emerging markets with growing healthcare capacities and increasing access to advanced cancer care also presents substantial growth potential. Furthermore, strategic partnerships between pharmaceutical companies, academic institutions, and contract research organizations are crucial for accelerating drug development and bringing innovative therapies to patients more efficiently.
Leading Players in the Advanced Renal Cell Carcinoma Therapeutics Sector
- Acceleron Pharma
- Argos Therapeutics
- AVEO Pharmaceuticals
- Bayer
- Bristol-Myers Squibb Company
- Eisai
- Exelixi
- Genentech
- Immatics Biotechnologies
- Merck
- Novartis
- Ono Pharmaceutical
- Pfizer
- Rexahn Pharmaceuticals
- Hoffmann-La Roche
Key Milestones in Advanced Renal Cell Carcinoma Therapeutics Industry
- 2019: Launch of cabozantinib for advanced RCC, expanding treatment options.
- 2020: Approval of nivolumab plus ipilimumab combination therapy, establishing a new standard of care for some advanced RCC patients.
- 2021: FDA approval of belzutifan for von Hippel-Lindau (VHL) disease-associated RCC, targeting a specific genetic pathway.
- 2022: Emergence of real-world evidence supporting the efficacy of advanced immunotherapies and targeted agents in specific RCC subtypes.
- 2023: Significant advancements in clinical trials for novel combination therapies involving PD-1/PD-L1 inhibitors and TKI agents, demonstrating improved progression-free survival.
- Early 2024: Continued progress in the development of novel checkpoint inhibitors and bispecific antibodies targeting renal cell carcinoma.
Strategic Outlook for Advanced Renal Cell Carcinoma Therapeutics Market
The strategic outlook for the advanced renal cell carcinoma therapeutics market is exceptionally positive, driven by continuous innovation and unmet clinical needs. Future growth will be accelerated by the development of precision medicine approaches, leveraging biomarker-driven patient selection and novel combination strategies to overcome treatment resistance. Increased investment in early-stage research, particularly in the realm of novel immunotherapy targets and advanced molecular therapies, will sustain the pipeline of future treatments. Strategic collaborations and partnerships will be crucial for navigating complex regulatory landscapes and ensuring market access for innovative therapies. The focus will increasingly shift towards improving long-term survival and quality of life for patients with advanced RCC, positioning this market for sustained and significant expansion in the coming years.
advanced renal cell carcinoma therapeutics Segmentation
-
1. Application
- 1.1. Hospitals
- 1.2. Cancer Research Institutes
- 1.3. Ambulatory Surgical Centers
- 1.4. Others
-
2. Types
- 2.1. Radiation Therapy
- 2.2. Chemotherapy
- 2.3. Hormone Therapy
- 2.4. Investigational Therapy
advanced renal cell carcinoma therapeutics Segmentation By Geography
- 1. CA
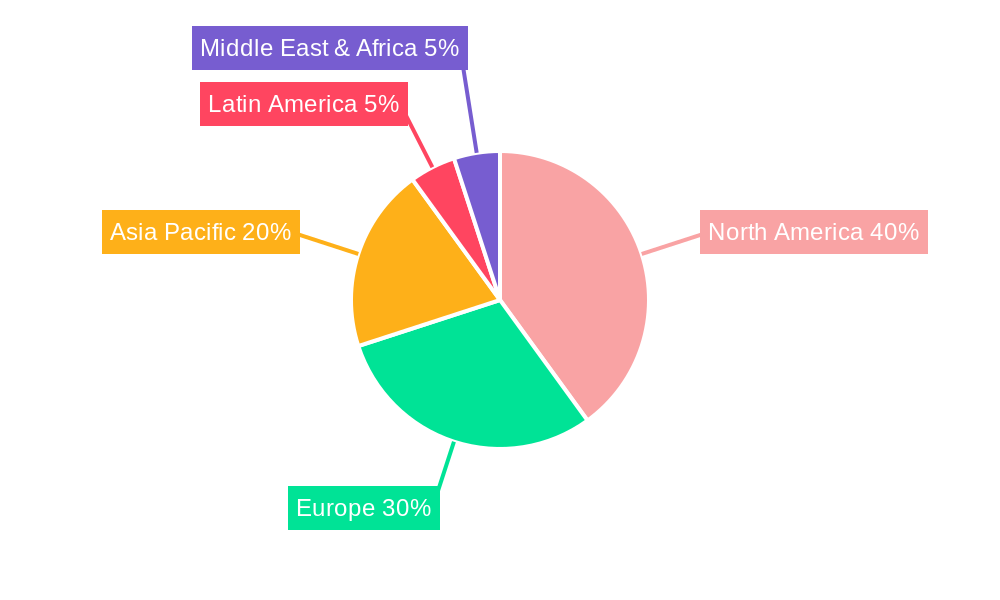
advanced renal cell carcinoma therapeutics Regional Market Share

Geographic Coverage of advanced renal cell carcinoma therapeutics
advanced renal cell carcinoma therapeutics REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. advanced renal cell carcinoma therapeutics Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals
- 5.1.2. Cancer Research Institutes
- 5.1.3. Ambulatory Surgical Centers
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Radiation Therapy
- 5.2.2. Chemotherapy
- 5.2.3. Hormone Therapy
- 5.2.4. Investigational Therapy
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. CA
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Competitive Analysis
- 6.1. Market Share Analysis 2025
- 6.2. Company Profiles
- 6.2.1 Acceleron Pharma
- 6.2.1.1. Overview
- 6.2.1.2. Products
- 6.2.1.3. SWOT Analysis
- 6.2.1.4. Recent Developments
- 6.2.1.5. Financials (Based on Availability)
- 6.2.2 Argos Therapeutics
- 6.2.2.1. Overview
- 6.2.2.2. Products
- 6.2.2.3. SWOT Analysis
- 6.2.2.4. Recent Developments
- 6.2.2.5. Financials (Based on Availability)
- 6.2.3 AVEO Pharmaceuticals
- 6.2.3.1. Overview
- 6.2.3.2. Products
- 6.2.3.3. SWOT Analysis
- 6.2.3.4. Recent Developments
- 6.2.3.5. Financials (Based on Availability)
- 6.2.4 Bayer
- 6.2.4.1. Overview
- 6.2.4.2. Products
- 6.2.4.3. SWOT Analysis
- 6.2.4.4. Recent Developments
- 6.2.4.5. Financials (Based on Availability)
- 6.2.5 Bristol-Myers Squibb Company
- 6.2.5.1. Overview
- 6.2.5.2. Products
- 6.2.5.3. SWOT Analysis
- 6.2.5.4. Recent Developments
- 6.2.5.5. Financials (Based on Availability)
- 6.2.6 Eisai
- 6.2.6.1. Overview
- 6.2.6.2. Products
- 6.2.6.3. SWOT Analysis
- 6.2.6.4. Recent Developments
- 6.2.6.5. Financials (Based on Availability)
- 6.2.7 Exelixi
- 6.2.7.1. Overview
- 6.2.7.2. Products
- 6.2.7.3. SWOT Analysis
- 6.2.7.4. Recent Developments
- 6.2.7.5. Financials (Based on Availability)
- 6.2.8 Genentech
- 6.2.8.1. Overview
- 6.2.8.2. Products
- 6.2.8.3. SWOT Analysis
- 6.2.8.4. Recent Developments
- 6.2.8.5. Financials (Based on Availability)
- 6.2.9 Immatics Biotechnologies
- 6.2.9.1. Overview
- 6.2.9.2. Products
- 6.2.9.3. SWOT Analysis
- 6.2.9.4. Recent Developments
- 6.2.9.5. Financials (Based on Availability)
- 6.2.10 Merck
- 6.2.10.1. Overview
- 6.2.10.2. Products
- 6.2.10.3. SWOT Analysis
- 6.2.10.4. Recent Developments
- 6.2.10.5. Financials (Based on Availability)
- 6.2.11 Novartis
- 6.2.11.1. Overview
- 6.2.11.2. Products
- 6.2.11.3. SWOT Analysis
- 6.2.11.4. Recent Developments
- 6.2.11.5. Financials (Based on Availability)
- 6.2.12 Ono Pharmaceutical
- 6.2.12.1. Overview
- 6.2.12.2. Products
- 6.2.12.3. SWOT Analysis
- 6.2.12.4. Recent Developments
- 6.2.12.5. Financials (Based on Availability)
- 6.2.13 Pfizer
- 6.2.13.1. Overview
- 6.2.13.2. Products
- 6.2.13.3. SWOT Analysis
- 6.2.13.4. Recent Developments
- 6.2.13.5. Financials (Based on Availability)
- 6.2.14 Rexahn Pharmaceuticals
- 6.2.14.1. Overview
- 6.2.14.2. Products
- 6.2.14.3. SWOT Analysis
- 6.2.14.4. Recent Developments
- 6.2.14.5. Financials (Based on Availability)
- 6.2.15 Hoffmann-La Roche
- 6.2.15.1. Overview
- 6.2.15.2. Products
- 6.2.15.3. SWOT Analysis
- 6.2.15.4. Recent Developments
- 6.2.15.5. Financials (Based on Availability)
- 6.2.1 Acceleron Pharma
List of Figures
- Figure 1: advanced renal cell carcinoma therapeutics Revenue Breakdown (undefined, %) by Product 2025 & 2033
- Figure 2: advanced renal cell carcinoma therapeutics Share (%) by Company 2025
List of Tables
- Table 1: advanced renal cell carcinoma therapeutics Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: advanced renal cell carcinoma therapeutics Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: advanced renal cell carcinoma therapeutics Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: advanced renal cell carcinoma therapeutics Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: advanced renal cell carcinoma therapeutics Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: advanced renal cell carcinoma therapeutics Revenue undefined Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the advanced renal cell carcinoma therapeutics?
The projected CAGR is approximately 6.4%.
2. Which companies are prominent players in the advanced renal cell carcinoma therapeutics?
Key companies in the market include Acceleron Pharma, Argos Therapeutics, AVEO Pharmaceuticals, Bayer, Bristol-Myers Squibb Company, Eisai, Exelixi, Genentech, Immatics Biotechnologies, Merck, Novartis, Ono Pharmaceutical, Pfizer, Rexahn Pharmaceuticals, Hoffmann-La Roche.
3. What are the main segments of the advanced renal cell carcinoma therapeutics?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3400.00, USD 5100.00, and USD 6800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "advanced renal cell carcinoma therapeutics," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the advanced renal cell carcinoma therapeutics report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the advanced renal cell carcinoma therapeutics?
To stay informed about further developments, trends, and reports in the advanced renal cell carcinoma therapeutics, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


