Key Insights
The global Atherectomy Devices Market is poised for significant expansion, projected to reach an estimated USD 939 million in 2025 and grow at a robust Compound Annual Growth Rate (CAGR) of 9.24% through 2033. This upward trajectory is fueled by a confluence of factors, including the increasing prevalence of cardiovascular and peripheral vascular diseases worldwide, driven by lifestyle changes, an aging population, and a greater focus on preventative healthcare. Technological advancements in atherectomy devices, leading to less invasive procedures with improved patient outcomes, are also a major impetus. The demand for directional, orbital, photo-ablative (laser), and rotational atherectomy systems is expected to rise as healthcare providers increasingly adopt these sophisticated treatment modalities to manage plaque buildup in arteries effectively. The market's growth is further supported by a rising number of minimally invasive procedures performed in hospitals, surgical centers, and ambulatory care settings, driven by faster recovery times and reduced healthcare costs.

Atherectomy Devices Market Market Size (In Million)

The market segmentation reveals key areas of growth and opportunity. Peripheral vascular applications are expected to lead the charge, owing to the escalating incidence of peripheral artery disease (PAD). Cardiovascular applications, particularly for coronary artery disease, will continue to be a substantial segment. Neurovascular applications, though smaller, are anticipated to witness considerable growth as advanced atherectomy techniques become more refined for treating cerebrovascular conditions. Key players like Medtronic, Abbott Laboratories, Boston Scientific Corporation, and Becton Dickinson and Company are actively investing in research and development, introducing innovative solutions and expanding their market reach across North America, Europe, and the rapidly growing Asia Pacific region. While the market demonstrates strong growth potential, factors such as high procedural costs and the need for specialized training for healthcare professionals may present minor challenges to its exponential growth.

Atherectomy Devices Market Company Market Share

Unlocking the Potential: A Comprehensive Analysis of the Global Atherectomy Devices Market (2019–2033)
This in-depth report provides an indispensable analysis of the global Atherectomy Devices Market, a rapidly evolving sector critical for treating cardiovascular and peripheral vascular diseases. Spanning from 2019 to 2033, with a base year of 2025 and a forecast period of 2025–2033, this study offers unparalleled insights into market dynamics, industry trends, leading segments, and strategic opportunities. We delve into the market's structure, from its key players and product innovations to the growth drivers, challenges, and future outlook.
The Atherectomy Devices Market is projected to witness significant expansion, driven by an aging global population, increasing prevalence of cardiovascular and peripheral artery diseases, and advancements in minimally invasive surgical techniques. This report leverages high-traffic keywords such as "atherectomy devices," "peripheral vascular intervention," "cardiovascular devices," "minimally invasive surgery," "medical devices market," "rotational atherectomy," "orbital atherectomy," and "laser atherectomy" to maximize search visibility and attract a broad spectrum of industry stakeholders, including medical device manufacturers, healthcare providers, investors, and regulatory bodies.
This comprehensive report includes detailed breakdowns of market segments by Product (Directional Atherectomy Systems, Orbital Atherectomy Systems, Photo-ablative (Laser) Atherectomy Systems, Rotational Atherectomy Systems), Application (Peripheral Vascular Applications, Cardiovascular Applications, Neurovascular Applications), and End User (Hospitals and Surgical Centers, Ambulatory Care Centers, Other End Users). We also highlight key industry developments, leading players, and strategic recommendations, providing a robust foundation for informed decision-making in this dynamic market.
Atherectomy Devices Market Dynamics & Concentration
The Atherectomy Devices Market exhibits a moderate to high concentration, with several key players dominating the landscape. Innovation remains a primary driver, fueled by the continuous development of less invasive and more effective atherectomy technologies designed to improve patient outcomes and reduce procedural complications. Regulatory frameworks, while essential for ensuring device safety and efficacy, can also present hurdles to market entry and product adoption. Product substitutes, such as angioplasty balloons and stents, continue to pose a competitive challenge, necessitating constant innovation and differentiation in atherectomy device offerings.
End-user trends are shifting towards less invasive procedures and outpatient settings, driving demand for atherectomy devices that facilitate these approaches. Hospitals and surgical centers remain the primary end-users, but ambulatory care centers are emerging as significant growth areas. Mergers and acquisitions (M&A) activities are a notable feature of the market, as larger companies seek to expand their portfolios and technological capabilities. The market has seen notable M&A deals, with an estimated XX M&A deal counts in the historical period. Market share is currently led by companies like Medtronic and Abbott Laboratories, with their respective market shares estimated at XX% and XX% respectively.
Atherectomy Devices Market Industry Trends & Analysis
The global Atherectomy Devices Market is poised for substantial growth, projected to expand at a Compound Annual Growth Rate (CAGR) of approximately XX% during the forecast period. This robust growth is underpinned by several intertwined industry trends. The escalating prevalence of cardiovascular diseases (CVDs) and peripheral artery diseases (PADs) worldwide serves as a primary growth catalyst. As populations age and lifestyle-related health issues become more common, the demand for effective treatments for atherosclerosis and other vascular blockages intensifies. Atherectomy devices offer a crucial solution by mechanically removing plaque buildup, thereby restoring blood flow and preventing severe complications like limb amputation or myocardial infarction.
Technological disruptions are continuously reshaping the market landscape. Innovations in catheter design, imaging guidance, and energy-based atherectomy technologies are leading to the development of devices that are more precise, less traumatic to vessel walls, and capable of treating a wider range of lesion complexities. For instance, advancements in rotational atherectomy systems have enhanced their ability to tackle calcified lesions, while orbital atherectomy offers a different approach for plaque modification. Photo-ablative (laser) atherectomy systems are gaining traction for their ability to vaporize plaque with high precision. These technological advancements are not only improving clinical efficacy but also enhancing procedural efficiency, which is a key consideration for healthcare providers seeking to optimize resource utilization.
Consumer preferences are increasingly leaning towards minimally invasive procedures due to their associated benefits, including reduced recovery times, lower pain, and fewer complications compared to traditional open surgeries. Atherectomy devices are at the forefront of this trend, enabling physicians to perform complex interventions through small incisions or punctures. This patient preference, coupled with physician adoption of these advanced techniques, is a significant driver of market penetration. The focus on improving quality of life and reducing long-term healthcare costs further bolsters the appeal of atherectomy as a therapeutic option.
Competitive dynamics within the atherectomy devices market are characterized by intense research and development, strategic partnerships, and a focus on gaining regulatory approvals for new indications and improved device performance. Companies are investing heavily in clinical trials to demonstrate the safety and effectiveness of their technologies, which is crucial for securing market access and physician trust. The market penetration for atherectomy devices, while still growing, is expected to increase as awareness of their benefits expands and reimbursement policies become more favorable. The overall market value is estimated to reach approximately $XX billion by 2033.
Leading Markets & Segments in Atherectomy Devices Market
The Atherectomy Devices Market is geographically diverse, with North America currently leading in terms of market share and adoption. This dominance is attributed to a well-established healthcare infrastructure, high disposable incomes, a high prevalence of cardiovascular diseases, and early adoption of advanced medical technologies. Within North America, the United States is the primary market, driven by a robust reimbursement framework and a significant volume of cardiovascular procedures performed annually. The region's advanced regulatory landscape also fosters innovation and rapid market entry for new atherectomy devices.
The market can be segmented by Product, Application, and End User, each exhibiting unique growth dynamics.
Product Segment Dominance:
- Rotational Atherectomy Systems: These systems have historically held a significant market share due to their established efficacy in treating calcified lesions, a common challenge in peripheral and coronary arteries. Key drivers for their continued dominance include their proven track record, physician familiarity, and ongoing improvements in device design for better control and minimal vessel trauma.
- Orbital Atherectomy Systems: This segment has witnessed substantial growth, offering a unique approach to plaque modification. Drivers include its effectiveness in treating challenging calcified lesions, its ability to prepare the vessel for subsequent interventions like stenting, and ongoing clinical evidence supporting its benefits.
- Directional Atherectomy Systems: These systems are crucial for plaque excision and have a strong presence in treating specific types of lesions. Their dominance is supported by their targeted approach to plaque removal and their utility in complex anatomies.
- Photo-ablative (Laser) Atherectomy Systems: While a smaller segment currently, laser atherectomy systems are experiencing rapid growth. Their key drivers include their ability to precisely ablate plaque with minimal thermal damage to surrounding tissues and their potential for treating occlusive and complex lesions, particularly in the peripheral vasculature.
Application Segment Dominance:
- Peripheral Vascular Applications: This application segment is the largest and fastest-growing within the atherectomy devices market. The escalating incidence of Peripheral Artery Disease (PAD), coupled with an aging population and rising rates of diabetes and obesity, fuels the demand for effective treatments. Economic policies promoting limb salvage and reducing amputation rates further contribute to the dominance of this segment.
- Cardiovascular Applications: This segment, primarily focusing on coronary interventions, remains a significant area for atherectomy devices. Drivers include the persistent burden of coronary artery disease and the need for advanced tools to treat complex coronary lesions.
- Neurovascular Applications: While a nascent but growing segment, neurovascular applications hold significant future potential. Advancements in device technology are enabling atherectomy for treating stroke-related blockages in the brain. Infrastructure development in interventional neurology and increasing research are key drivers.
End User Segment Dominance:
- Hospitals and Surgical Centers: These institutions represent the largest end-user segment due to their comprehensive facilities and expertise in performing complex interventional procedures. Economic policies that support advanced medical treatments and the availability of specialized medical professionals are key drivers.
- Ambulatory Care Centers: This segment is experiencing rapid growth as healthcare systems move towards outpatient care for less complex procedures. Their dominance is influenced by cost-effectiveness and patient convenience. Other End Users, including research institutions and specialized clinics, also contribute to market demand.
Atherectomy Devices Market Product Developments
The atherectomy devices market is characterized by relentless product innovation aimed at enhancing efficacy, safety, and user-friendliness. Recent developments have focused on miniaturization of devices for improved navigability in complex anatomies, enhanced energy delivery mechanisms for efficient plaque removal, and advanced imaging integration for real-time visualization during procedures. Companies are developing systems with improved debulking capabilities, reduced risk of distal embolization, and the ability to treat a wider range of plaque types, including highly calcified lesions. These innovations are crucial for expanding the indications for atherectomy and increasing physician adoption, thereby providing significant competitive advantages.
Key Drivers of Atherectomy Devices Market Growth
The growth of the atherectomy devices market is propelled by a confluence of technological, economic, and regulatory factors. Technologically, advancements in catheter design, energy sources (e.g., laser, rotational, orbital), and imaging guidance are enabling more precise and less invasive plaque removal. Economically, the increasing burden of cardiovascular and peripheral artery diseases, coupled with an aging global population, is driving demand for effective treatment solutions. Regulatory bodies are increasingly approving innovative atherectomy devices, recognizing their potential to improve patient outcomes and reduce healthcare costs. For instance, the growing emphasis on limb salvage in peripheral artery disease cases further fuels the demand for atherectomy devices that can effectively treat occlusive lesions.
Challenges in the Atherectomy Devices Market Market
Despite the promising growth trajectory, the atherectomy devices market faces several challenges. Regulatory hurdles, including the rigorous approval processes for novel devices and the need for extensive clinical data, can slow down market entry. Competition from established alternatives like angioplasty balloons and stenting procedures necessitates continuous demonstration of superior clinical and economic benefits. Reimbursement policies, though generally supportive, can vary by region and procedure, impacting adoption rates. Supply chain disruptions and the high cost of advanced atherectomy systems also present significant barriers, limiting accessibility for some healthcare providers and patient populations.
Emerging Opportunities in Atherectomy Devices Market
Emerging opportunities in the atherectomy devices market are driven by unmet clinical needs and technological breakthroughs. The expanding understanding and treatment of complex atherosclerotic plaques, particularly in the neurovascular space for stroke prevention, presents a significant growth avenue. Strategic partnerships between device manufacturers and research institutions are accelerating the development of next-generation atherectomy technologies, such as biodegradable atherectomy devices or those incorporating AI-driven procedural guidance. Furthermore, the increasing focus on value-based healthcare is creating opportunities for atherectomy devices that demonstrate cost-effectiveness and improved long-term patient outcomes, encouraging market expansion into underserved regions and patient demographics.
Leading Players in the Atherectomy Devices Market Sector
- Becton Dickinson and Company
- Medtronic
- Terumo Corporation
- Abbott Laboratories
- Cardinal Health Inc
- AngioDynamics Inc
- Koninklijke Philips NV
- Cardiovascular Systems Inc
- RA Medical Systems
- Biomerics LLC
- Boston Scientific Corporation
- B Braun SE
Key Milestones in Atherectomy Devices Market Industry
- August 2022: AngioDynamics, Inc. announced that the Auryon Atherectomy System has gained 510(k) clearance for an expanded indication that includes arterial thrombectomy.
- February 2022: RA Medical Systems, announced that enrollment has reached 95 subjects in the pivotal clinical trial to evaluate the safety and effectiveness of the DABRA excimer laser system as an atherectomy device for the treatment of peripheral arterial disease.
Strategic Outlook for Atherectomy Devices Market Market
The strategic outlook for the atherectomy devices market remains exceptionally positive, driven by continuous innovation and a growing global demand for effective treatments for vascular diseases. Future market growth will likely be accelerated by the development of more intelligent, adaptable atherectomy systems capable of real-time lesion analysis and personalized treatment delivery. Strategic opportunities lie in expanding the therapeutic applications of atherectomy devices, particularly in the neurovascular domain and for complex peripheral lesions. Companies that invest in demonstrating the cost-effectiveness and superior clinical outcomes of their atherectomy technologies will be well-positioned to capture market share and drive long-term sustainable growth.
Atherectomy Devices Market Segmentation
-
1. Product
- 1.1. Directional Atherectomy Systems
- 1.2. Orbital Atherectomy Systems
- 1.3. Photo-ablative (Laser) Atherectomy Systems
- 1.4. Rotational Atherectomy Systems
-
2. Application
- 2.1. Peripheral Vascular Applications
- 2.2. Cardiovascular Applications
- 2.3. Neurovascular Applications
-
3. End User
- 3.1. Hospitals and Surgical Centers
- 3.2. Ambulatory Care Centers
- 3.3. Other End Users
Atherectomy Devices Market Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
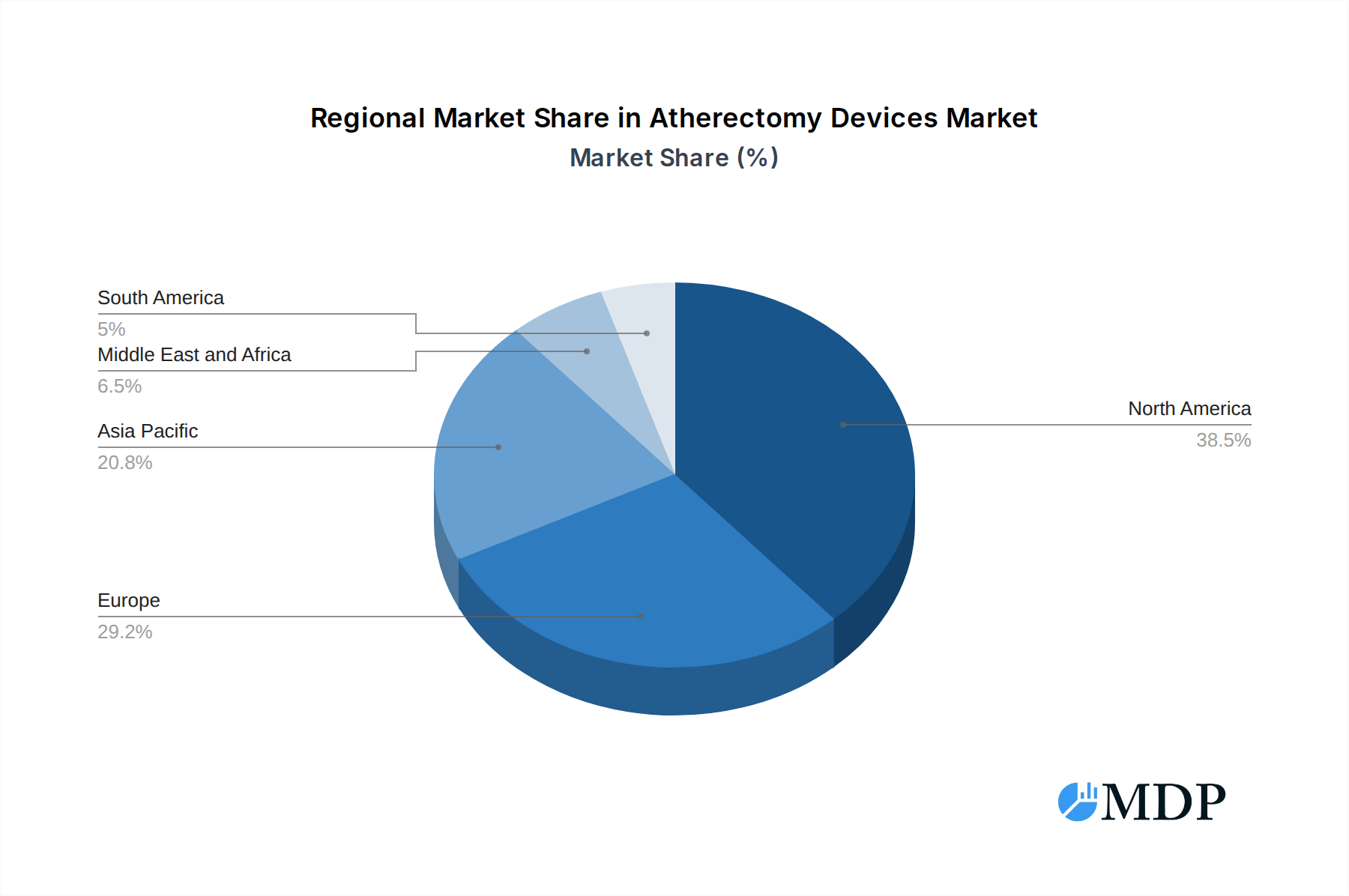
Atherectomy Devices Market Regional Market Share

Geographic Coverage of Atherectomy Devices Market
Atherectomy Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9.24% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. MDP Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product
- 5.1.1. Directional Atherectomy Systems
- 5.1.2. Orbital Atherectomy Systems
- 5.1.3. Photo-ablative (Laser) Atherectomy Systems
- 5.1.4. Rotational Atherectomy Systems
- 5.2. Market Analysis, Insights and Forecast - by Application
- 5.2.1. Peripheral Vascular Applications
- 5.2.2. Cardiovascular Applications
- 5.2.3. Neurovascular Applications
- 5.3. Market Analysis, Insights and Forecast - by End User
- 5.3.1. Hospitals and Surgical Centers
- 5.3.2. Ambulatory Care Centers
- 5.3.3. Other End Users
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. North America
- 5.4.2. Europe
- 5.4.3. Asia Pacific
- 5.4.4. Middle East and Africa
- 5.4.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Product
- 6. Global Atherectomy Devices Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product
- 6.1.1. Directional Atherectomy Systems
- 6.1.2. Orbital Atherectomy Systems
- 6.1.3. Photo-ablative (Laser) Atherectomy Systems
- 6.1.4. Rotational Atherectomy Systems
- 6.2. Market Analysis, Insights and Forecast - by Application
- 6.2.1. Peripheral Vascular Applications
- 6.2.2. Cardiovascular Applications
- 6.2.3. Neurovascular Applications
- 6.3. Market Analysis, Insights and Forecast - by End User
- 6.3.1. Hospitals and Surgical Centers
- 6.3.2. Ambulatory Care Centers
- 6.3.3. Other End Users
- 6.1. Market Analysis, Insights and Forecast - by Product
- 7. North America Atherectomy Devices Market Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product
- 7.1.1. Directional Atherectomy Systems
- 7.1.2. Orbital Atherectomy Systems
- 7.1.3. Photo-ablative (Laser) Atherectomy Systems
- 7.1.4. Rotational Atherectomy Systems
- 7.2. Market Analysis, Insights and Forecast - by Application
- 7.2.1. Peripheral Vascular Applications
- 7.2.2. Cardiovascular Applications
- 7.2.3. Neurovascular Applications
- 7.3. Market Analysis, Insights and Forecast - by End User
- 7.3.1. Hospitals and Surgical Centers
- 7.3.2. Ambulatory Care Centers
- 7.3.3. Other End Users
- 7.1. Market Analysis, Insights and Forecast - by Product
- 8. Europe Atherectomy Devices Market Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product
- 8.1.1. Directional Atherectomy Systems
- 8.1.2. Orbital Atherectomy Systems
- 8.1.3. Photo-ablative (Laser) Atherectomy Systems
- 8.1.4. Rotational Atherectomy Systems
- 8.2. Market Analysis, Insights and Forecast - by Application
- 8.2.1. Peripheral Vascular Applications
- 8.2.2. Cardiovascular Applications
- 8.2.3. Neurovascular Applications
- 8.3. Market Analysis, Insights and Forecast - by End User
- 8.3.1. Hospitals and Surgical Centers
- 8.3.2. Ambulatory Care Centers
- 8.3.3. Other End Users
- 8.1. Market Analysis, Insights and Forecast - by Product
- 9. Asia Pacific Atherectomy Devices Market Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product
- 9.1.1. Directional Atherectomy Systems
- 9.1.2. Orbital Atherectomy Systems
- 9.1.3. Photo-ablative (Laser) Atherectomy Systems
- 9.1.4. Rotational Atherectomy Systems
- 9.2. Market Analysis, Insights and Forecast - by Application
- 9.2.1. Peripheral Vascular Applications
- 9.2.2. Cardiovascular Applications
- 9.2.3. Neurovascular Applications
- 9.3. Market Analysis, Insights and Forecast - by End User
- 9.3.1. Hospitals and Surgical Centers
- 9.3.2. Ambulatory Care Centers
- 9.3.3. Other End Users
- 9.1. Market Analysis, Insights and Forecast - by Product
- 10. Middle East and Africa Atherectomy Devices Market Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Product
- 10.1.1. Directional Atherectomy Systems
- 10.1.2. Orbital Atherectomy Systems
- 10.1.3. Photo-ablative (Laser) Atherectomy Systems
- 10.1.4. Rotational Atherectomy Systems
- 10.2. Market Analysis, Insights and Forecast - by Application
- 10.2.1. Peripheral Vascular Applications
- 10.2.2. Cardiovascular Applications
- 10.2.3. Neurovascular Applications
- 10.3. Market Analysis, Insights and Forecast - by End User
- 10.3.1. Hospitals and Surgical Centers
- 10.3.2. Ambulatory Care Centers
- 10.3.3. Other End Users
- 10.1. Market Analysis, Insights and Forecast - by Product
- 11. South America Atherectomy Devices Market Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Product
- 11.1.1. Directional Atherectomy Systems
- 11.1.2. Orbital Atherectomy Systems
- 11.1.3. Photo-ablative (Laser) Atherectomy Systems
- 11.1.4. Rotational Atherectomy Systems
- 11.2. Market Analysis, Insights and Forecast - by Application
- 11.2.1. Peripheral Vascular Applications
- 11.2.2. Cardiovascular Applications
- 11.2.3. Neurovascular Applications
- 11.3. Market Analysis, Insights and Forecast - by End User
- 11.3.1. Hospitals and Surgical Centers
- 11.3.2. Ambulatory Care Centers
- 11.3.3. Other End Users
- 11.1. Market Analysis, Insights and Forecast - by Product
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Becton Dickinson and Company
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Medtronic
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Terumo Corporation
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Abbott Laboratories
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Cardinal Health Inc
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 AngioDynamics Inc *List Not Exhaustive
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Koninklijke Philips NV
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Cardiovascular Systems Inc
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 RA Medical Systems
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Biomerics LLC
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Boston Scientific Corporation
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 B Braun SE
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.1 Becton Dickinson and Company
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Atherectomy Devices Market Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: Global Atherectomy Devices Market Volume Breakdown (K Units, %) by Region 2025 & 2033
- Figure 3: North America Atherectomy Devices Market Revenue (million), by Product 2025 & 2033
- Figure 4: North America Atherectomy Devices Market Volume (K Units), by Product 2025 & 2033
- Figure 5: North America Atherectomy Devices Market Revenue Share (%), by Product 2025 & 2033
- Figure 6: North America Atherectomy Devices Market Volume Share (%), by Product 2025 & 2033
- Figure 7: North America Atherectomy Devices Market Revenue (million), by Application 2025 & 2033
- Figure 8: North America Atherectomy Devices Market Volume (K Units), by Application 2025 & 2033
- Figure 9: North America Atherectomy Devices Market Revenue Share (%), by Application 2025 & 2033
- Figure 10: North America Atherectomy Devices Market Volume Share (%), by Application 2025 & 2033
- Figure 11: North America Atherectomy Devices Market Revenue (million), by End User 2025 & 2033
- Figure 12: North America Atherectomy Devices Market Volume (K Units), by End User 2025 & 2033
- Figure 13: North America Atherectomy Devices Market Revenue Share (%), by End User 2025 & 2033
- Figure 14: North America Atherectomy Devices Market Volume Share (%), by End User 2025 & 2033
- Figure 15: North America Atherectomy Devices Market Revenue (million), by Country 2025 & 2033
- Figure 16: North America Atherectomy Devices Market Volume (K Units), by Country 2025 & 2033
- Figure 17: North America Atherectomy Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 18: North America Atherectomy Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 19: Europe Atherectomy Devices Market Revenue (million), by Product 2025 & 2033
- Figure 20: Europe Atherectomy Devices Market Volume (K Units), by Product 2025 & 2033
- Figure 21: Europe Atherectomy Devices Market Revenue Share (%), by Product 2025 & 2033
- Figure 22: Europe Atherectomy Devices Market Volume Share (%), by Product 2025 & 2033
- Figure 23: Europe Atherectomy Devices Market Revenue (million), by Application 2025 & 2033
- Figure 24: Europe Atherectomy Devices Market Volume (K Units), by Application 2025 & 2033
- Figure 25: Europe Atherectomy Devices Market Revenue Share (%), by Application 2025 & 2033
- Figure 26: Europe Atherectomy Devices Market Volume Share (%), by Application 2025 & 2033
- Figure 27: Europe Atherectomy Devices Market Revenue (million), by End User 2025 & 2033
- Figure 28: Europe Atherectomy Devices Market Volume (K Units), by End User 2025 & 2033
- Figure 29: Europe Atherectomy Devices Market Revenue Share (%), by End User 2025 & 2033
- Figure 30: Europe Atherectomy Devices Market Volume Share (%), by End User 2025 & 2033
- Figure 31: Europe Atherectomy Devices Market Revenue (million), by Country 2025 & 2033
- Figure 32: Europe Atherectomy Devices Market Volume (K Units), by Country 2025 & 2033
- Figure 33: Europe Atherectomy Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 34: Europe Atherectomy Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 35: Asia Pacific Atherectomy Devices Market Revenue (million), by Product 2025 & 2033
- Figure 36: Asia Pacific Atherectomy Devices Market Volume (K Units), by Product 2025 & 2033
- Figure 37: Asia Pacific Atherectomy Devices Market Revenue Share (%), by Product 2025 & 2033
- Figure 38: Asia Pacific Atherectomy Devices Market Volume Share (%), by Product 2025 & 2033
- Figure 39: Asia Pacific Atherectomy Devices Market Revenue (million), by Application 2025 & 2033
- Figure 40: Asia Pacific Atherectomy Devices Market Volume (K Units), by Application 2025 & 2033
- Figure 41: Asia Pacific Atherectomy Devices Market Revenue Share (%), by Application 2025 & 2033
- Figure 42: Asia Pacific Atherectomy Devices Market Volume Share (%), by Application 2025 & 2033
- Figure 43: Asia Pacific Atherectomy Devices Market Revenue (million), by End User 2025 & 2033
- Figure 44: Asia Pacific Atherectomy Devices Market Volume (K Units), by End User 2025 & 2033
- Figure 45: Asia Pacific Atherectomy Devices Market Revenue Share (%), by End User 2025 & 2033
- Figure 46: Asia Pacific Atherectomy Devices Market Volume Share (%), by End User 2025 & 2033
- Figure 47: Asia Pacific Atherectomy Devices Market Revenue (million), by Country 2025 & 2033
- Figure 48: Asia Pacific Atherectomy Devices Market Volume (K Units), by Country 2025 & 2033
- Figure 49: Asia Pacific Atherectomy Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 50: Asia Pacific Atherectomy Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 51: Middle East and Africa Atherectomy Devices Market Revenue (million), by Product 2025 & 2033
- Figure 52: Middle East and Africa Atherectomy Devices Market Volume (K Units), by Product 2025 & 2033
- Figure 53: Middle East and Africa Atherectomy Devices Market Revenue Share (%), by Product 2025 & 2033
- Figure 54: Middle East and Africa Atherectomy Devices Market Volume Share (%), by Product 2025 & 2033
- Figure 55: Middle East and Africa Atherectomy Devices Market Revenue (million), by Application 2025 & 2033
- Figure 56: Middle East and Africa Atherectomy Devices Market Volume (K Units), by Application 2025 & 2033
- Figure 57: Middle East and Africa Atherectomy Devices Market Revenue Share (%), by Application 2025 & 2033
- Figure 58: Middle East and Africa Atherectomy Devices Market Volume Share (%), by Application 2025 & 2033
- Figure 59: Middle East and Africa Atherectomy Devices Market Revenue (million), by End User 2025 & 2033
- Figure 60: Middle East and Africa Atherectomy Devices Market Volume (K Units), by End User 2025 & 2033
- Figure 61: Middle East and Africa Atherectomy Devices Market Revenue Share (%), by End User 2025 & 2033
- Figure 62: Middle East and Africa Atherectomy Devices Market Volume Share (%), by End User 2025 & 2033
- Figure 63: Middle East and Africa Atherectomy Devices Market Revenue (million), by Country 2025 & 2033
- Figure 64: Middle East and Africa Atherectomy Devices Market Volume (K Units), by Country 2025 & 2033
- Figure 65: Middle East and Africa Atherectomy Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 66: Middle East and Africa Atherectomy Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 67: South America Atherectomy Devices Market Revenue (million), by Product 2025 & 2033
- Figure 68: South America Atherectomy Devices Market Volume (K Units), by Product 2025 & 2033
- Figure 69: South America Atherectomy Devices Market Revenue Share (%), by Product 2025 & 2033
- Figure 70: South America Atherectomy Devices Market Volume Share (%), by Product 2025 & 2033
- Figure 71: South America Atherectomy Devices Market Revenue (million), by Application 2025 & 2033
- Figure 72: South America Atherectomy Devices Market Volume (K Units), by Application 2025 & 2033
- Figure 73: South America Atherectomy Devices Market Revenue Share (%), by Application 2025 & 2033
- Figure 74: South America Atherectomy Devices Market Volume Share (%), by Application 2025 & 2033
- Figure 75: South America Atherectomy Devices Market Revenue (million), by End User 2025 & 2033
- Figure 76: South America Atherectomy Devices Market Volume (K Units), by End User 2025 & 2033
- Figure 77: South America Atherectomy Devices Market Revenue Share (%), by End User 2025 & 2033
- Figure 78: South America Atherectomy Devices Market Volume Share (%), by End User 2025 & 2033
- Figure 79: South America Atherectomy Devices Market Revenue (million), by Country 2025 & 2033
- Figure 80: South America Atherectomy Devices Market Volume (K Units), by Country 2025 & 2033
- Figure 81: South America Atherectomy Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 82: South America Atherectomy Devices Market Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Atherectomy Devices Market Revenue million Forecast, by Product 2020 & 2033
- Table 2: Global Atherectomy Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 3: Global Atherectomy Devices Market Revenue million Forecast, by Application 2020 & 2033
- Table 4: Global Atherectomy Devices Market Volume K Units Forecast, by Application 2020 & 2033
- Table 5: Global Atherectomy Devices Market Revenue million Forecast, by End User 2020 & 2033
- Table 6: Global Atherectomy Devices Market Volume K Units Forecast, by End User 2020 & 2033
- Table 7: Global Atherectomy Devices Market Revenue million Forecast, by Region 2020 & 2033
- Table 8: Global Atherectomy Devices Market Volume K Units Forecast, by Region 2020 & 2033
- Table 9: Global Atherectomy Devices Market Revenue million Forecast, by Product 2020 & 2033
- Table 10: Global Atherectomy Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 11: Global Atherectomy Devices Market Revenue million Forecast, by Application 2020 & 2033
- Table 12: Global Atherectomy Devices Market Volume K Units Forecast, by Application 2020 & 2033
- Table 13: Global Atherectomy Devices Market Revenue million Forecast, by End User 2020 & 2033
- Table 14: Global Atherectomy Devices Market Volume K Units Forecast, by End User 2020 & 2033
- Table 15: Global Atherectomy Devices Market Revenue million Forecast, by Country 2020 & 2033
- Table 16: Global Atherectomy Devices Market Volume K Units Forecast, by Country 2020 & 2033
- Table 17: United States Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 18: United States Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 19: Canada Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Canada Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 21: Mexico Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Mexico Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 23: Global Atherectomy Devices Market Revenue million Forecast, by Product 2020 & 2033
- Table 24: Global Atherectomy Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 25: Global Atherectomy Devices Market Revenue million Forecast, by Application 2020 & 2033
- Table 26: Global Atherectomy Devices Market Volume K Units Forecast, by Application 2020 & 2033
- Table 27: Global Atherectomy Devices Market Revenue million Forecast, by End User 2020 & 2033
- Table 28: Global Atherectomy Devices Market Volume K Units Forecast, by End User 2020 & 2033
- Table 29: Global Atherectomy Devices Market Revenue million Forecast, by Country 2020 & 2033
- Table 30: Global Atherectomy Devices Market Volume K Units Forecast, by Country 2020 & 2033
- Table 31: Germany Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Germany Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 33: United Kingdom Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: United Kingdom Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 35: France Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: France Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 37: Italy Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 38: Italy Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 39: Spain Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 40: Spain Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 41: Rest of Europe Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Rest of Europe Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 43: Global Atherectomy Devices Market Revenue million Forecast, by Product 2020 & 2033
- Table 44: Global Atherectomy Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 45: Global Atherectomy Devices Market Revenue million Forecast, by Application 2020 & 2033
- Table 46: Global Atherectomy Devices Market Volume K Units Forecast, by Application 2020 & 2033
- Table 47: Global Atherectomy Devices Market Revenue million Forecast, by End User 2020 & 2033
- Table 48: Global Atherectomy Devices Market Volume K Units Forecast, by End User 2020 & 2033
- Table 49: Global Atherectomy Devices Market Revenue million Forecast, by Country 2020 & 2033
- Table 50: Global Atherectomy Devices Market Volume K Units Forecast, by Country 2020 & 2033
- Table 51: China Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 52: China Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 53: Japan Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 54: Japan Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 55: India Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 56: India Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 57: Australia Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 58: Australia Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 59: South Korea Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 60: South Korea Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 61: Rest of Asia Pacific Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 62: Rest of Asia Pacific Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 63: Global Atherectomy Devices Market Revenue million Forecast, by Product 2020 & 2033
- Table 64: Global Atherectomy Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 65: Global Atherectomy Devices Market Revenue million Forecast, by Application 2020 & 2033
- Table 66: Global Atherectomy Devices Market Volume K Units Forecast, by Application 2020 & 2033
- Table 67: Global Atherectomy Devices Market Revenue million Forecast, by End User 2020 & 2033
- Table 68: Global Atherectomy Devices Market Volume K Units Forecast, by End User 2020 & 2033
- Table 69: Global Atherectomy Devices Market Revenue million Forecast, by Country 2020 & 2033
- Table 70: Global Atherectomy Devices Market Volume K Units Forecast, by Country 2020 & 2033
- Table 71: GCC Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 72: GCC Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 73: South Africa Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 74: South Africa Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 75: Rest of Middle East and Africa Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 76: Rest of Middle East and Africa Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 77: Global Atherectomy Devices Market Revenue million Forecast, by Product 2020 & 2033
- Table 78: Global Atherectomy Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 79: Global Atherectomy Devices Market Revenue million Forecast, by Application 2020 & 2033
- Table 80: Global Atherectomy Devices Market Volume K Units Forecast, by Application 2020 & 2033
- Table 81: Global Atherectomy Devices Market Revenue million Forecast, by End User 2020 & 2033
- Table 82: Global Atherectomy Devices Market Volume K Units Forecast, by End User 2020 & 2033
- Table 83: Global Atherectomy Devices Market Revenue million Forecast, by Country 2020 & 2033
- Table 84: Global Atherectomy Devices Market Volume K Units Forecast, by Country 2020 & 2033
- Table 85: Brazil Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 86: Brazil Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 87: Argentina Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 88: Argentina Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
- Table 89: Rest of South America Atherectomy Devices Market Revenue (million) Forecast, by Application 2020 & 2033
- Table 90: Rest of South America Atherectomy Devices Market Volume (K Units) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Atherectomy Devices Market?
The projected CAGR is approximately 9.24%.
2. Which companies are prominent players in the Atherectomy Devices Market?
Key companies in the market include Becton Dickinson and Company, Medtronic, Terumo Corporation, Abbott Laboratories, Cardinal Health Inc, AngioDynamics Inc *List Not Exhaustive, Koninklijke Philips NV, Cardiovascular Systems Inc, RA Medical Systems, Biomerics LLC, Boston Scientific Corporation, B Braun SE.
3. What are the main segments of the Atherectomy Devices Market?
The market segments include Product, Application, End User.
4. Can you provide details about the market size?
The market size is estimated to be USD 939 million as of 2022.
5. What are some drivers contributing to market growth?
Continuous Product Development and Commercialization; Favourable Medical Reimbursement Scenario in Mature Markets; Growing Demand for Minimally Invasive Surgical Procedures.
6. What are the notable trends driving market growth?
Directional Atherectomy Segment Holds a Significant Market Share in the Studied Market Over the Forecast Period.
7. Are there any restraints impacting market growth?
Stringent Regulatory Scenario; Lack of Well-trained Surgeons.
8. Can you provide examples of recent developments in the market?
August 2022: AngioDynamics, Inc. announced that the Auryon Atherectomy System has gained 510(k) clearance for an expanded indication that includes arterial thrombectomy.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million and volume, measured in K Units.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Atherectomy Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Atherectomy Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Atherectomy Devices Market?
To stay informed about further developments, trends, and reports in the Atherectomy Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


