Key Insights
The Cutaneous T-Cell Lymphoma (CTLC) market is poised for significant expansion, projected to reach an estimated USD 6.84 billion by 2025. This growth trajectory is underpinned by a robust Compound Annual Growth Rate (CAGR) of 12.82% during the forecast period of 2025-2033. A primary driver for this upward trend is the increasing incidence and prevalence of CTCL, particularly Mycosis Fungoides (MF) and Sezary Syndrome (SS), driven by an aging global population and improved diagnostic capabilities. Advancements in treatment modalities, including novel immunotherapies, targeted therapies, and sophisticated radiation techniques, are also playing a crucial role. These innovative treatments offer improved efficacy and reduced side effects compared to traditional chemotherapy, thereby enhancing patient outcomes and driving market demand. Furthermore, growing awareness among healthcare professionals and patients about the disease and available treatment options is contributing to earlier diagnosis and intervention.
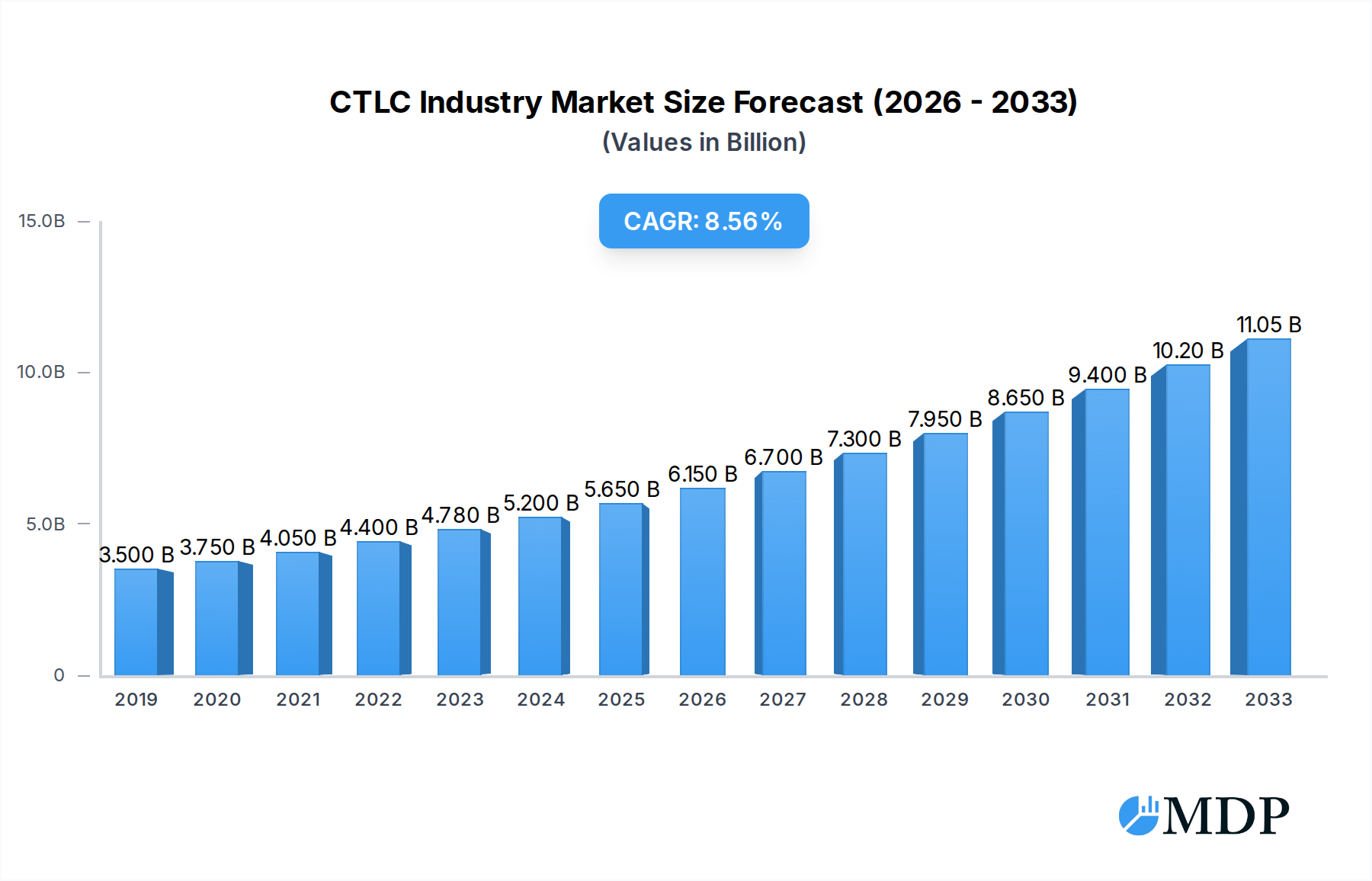
CTLC Industry Market Size (In Billion)

The market's expansion is further fueled by substantial investments in research and development by leading pharmaceutical and biotechnology companies. These entities are actively developing next-generation therapies to address unmet needs in CTCL treatment, particularly for advanced stages of the disease. Emerging markets, especially in the Asia Pacific region, are expected to contribute to market growth due to rising healthcare expenditure and increasing access to advanced medical treatments. However, the market may face certain restraints, such as the high cost of advanced therapies and the potential for treatment side effects. Nonetheless, the continuous innovation in drug development and therapeutic approaches, coupled with a growing understanding of CTCL pathogenesis, indicates a strong and sustained growth outlook for the CTCL market in the coming years.
CTLC Industry Company Market Share

Here's the SEO-optimized and engaging report description for the CTLC Industry:
CTLC Industry Market Dynamics & Concentration
The Cutaneous T-cell Lymphoma (CTLC) industry is characterized by a dynamic market landscape and evolving concentration. This report analyzes the key factors shaping its trajectory, including innovation drivers that are pushing the boundaries of treatment efficacy and patient outcomes. Regulatory frameworks, particularly those set forth by the US FDA, play a pivotal role in approving novel therapies and ensuring patient safety, with recent advancements from Citius Pharmaceuticals Inc. and Soligenix Inc. underscoring their significance. Product substitutes, while present, are increasingly challenged by the development of targeted and personalized CTCL treatments. End-user trends are shifting towards minimally invasive and more effective therapeutic options, driven by a growing awareness of CTCL and its impact on quality of life. Merger and acquisition (M&A) activities, while not yet at a billion-dollar scale, indicate strategic consolidation and investment within the sector, with an estimated X M&A deals recorded in the historical period. The market share distribution is a key focus, with leading pharmaceutical and biotechnology companies vying for dominance in this specialized oncological field.
CTLC Industry Industry Trends & Analysis
The Cutaneous T-cell Lymphoma (CTLC) industry is poised for significant growth, propelled by a confluence of market growth drivers, technological disruptions, evolving consumer preferences, and intensifying competitive dynamics. The compounded annual growth rate (CAGR) for the CTCL market is projected to be XX% from 2025 to 2033, reaching an estimated market size of over a billion dollars by the end of the forecast period. Technological disruptions, particularly in the realm of immunotherapy and targeted therapy, are revolutionizing treatment paradigms, offering new hope for patients with persistent or recurrent CTCL. Consumer preferences are increasingly leaning towards therapies that offer improved quality of life, reduced side effects, and personalized treatment plans. This shift is driving research and development into novel drug modalities and delivery systems. Competitive dynamics are intensifying as major players like Merck & Co. Inc. and Bristol Myers Squibb, alongside innovative biotechs such as Innate Pharma and Seattle Genetics, invest heavily in pipeline development and strategic partnerships. Market penetration of advanced therapies is on an upward trend, reflecting the growing acceptance and efficacy of newer treatment approaches. The overall market penetration is estimated to reach XX% by 2033. Factors such as an aging global population, increasing incidence rates of CTCL, and advancements in diagnostic technologies are further contributing to the robust expansion of the CTCL market. The historical period from 2019 to 2024 has witnessed consistent innovation and investment, setting a strong foundation for future growth.
Leading Markets & Segments in CTLC Industry
The Cutaneous T-cell Lymphoma (CTLC) industry's market dominance is clearly bifurcated across key segments, with a significant focus on Mycosis Fungoides (MF) and Sezary Syndrome (SS) due to their higher prevalence.
Dominant Segments by Type:
- Mycosis Fungoides (MF): This is the most prevalent subtype of CTCL, representing approximately XX% of all CTCL diagnoses. Its widespread occurrence drives substantial demand for effective treatments, making it a primary focus for pharmaceutical companies. Economic policies that encourage R&D for rare diseases, coupled with growing awareness campaigns, contribute to its market leadership.
- Sezary Syndrome (SS): While less common than MF, SS is a more aggressive form of CTCL, necessitating advanced treatment modalities. The higher unmet medical need in SS patients fuels innovation and market growth within this segment. Infrastructure development in specialized cancer centers capable of managing complex cases further supports its market presence.
- Other Types: This segment, encompassing less common CTCL variants, presents niche opportunities for specialized therapies and is expected to witness growth driven by advancements in personalized medicine.
Dominant Segments by Treatment:
- Immunotherapy: This treatment modality is emerging as a cornerstone in CTCL management, demonstrating superior efficacy in many cases. Market growth in this segment is driven by the development of novel immune checkpoint inhibitors and cell-based therapies. Regulatory frameworks that expedite the approval of innovative immunotherapies are crucial drivers.
- Targeted Therapy: With a deeper understanding of the molecular drivers of CTCL, targeted therapies are gaining traction. These treatments offer precision and improved patient outcomes. Technological advancements in genomics and proteomics are key enablers for this segment's expansion.
- Chemotherapy: While still a significant treatment option, particularly for advanced stages, the role of chemotherapy is evolving with the advent of more targeted and less toxic alternatives. Its market share is expected to see a gradual shift towards combination therapies and supportive care.
- Radiation Therapy: Remains a vital component for localized CTCL, particularly for MF. Infrastructure in specialized radiation oncology centers and government incentives for advanced radiotherapy adoption contribute to its sustained market presence.
- Other Treatments: This broad category includes topical agents, phototherapy, and supportive care, which collectively address a significant portion of CTCL patient needs and contribute to market diversification.
The dominance of MF and SS, coupled with the ascendancy of immunotherapy and targeted therapy, defines the current and future landscape of the CTCL industry.
CTLC Industry Product Developments
The CTLC industry is witnessing a surge in product innovations aimed at enhancing therapeutic efficacy and patient outcomes. Key developments include the advancement of engineered IL-2-diphtheria toxin fusion proteins like denileukin diftitox by Citius Pharmaceuticals Inc., which has seen its biologics license application accepted by the US FDA for persistent or recurrent CTCL. Soligenix Inc.'s HyBryte (synthetic hypericin) has also advanced with an iPSP agreement from the FDA for CTCL treatment. These innovations highlight a trend towards precision medicine, leveraging biotechnological advancements to create more targeted and effective treatments. Competitive advantages are being forged through novel drug formulations, improved delivery mechanisms, and the exploration of combination therapies. The emphasis on developing treatments with reduced side effects and improved quality of life for patients is a significant technological trend shaping the market fit of new products.
Key Drivers of CTLC Industry Growth
The growth of the CTLC industry is primarily propelled by several key drivers. Technological advancements in understanding the pathogenesis of CTCL are leading to the development of novel immunotherapies and targeted therapies, offering more effective treatment options. An increasing global incidence of CTCL, coupled with an aging population prone to the disease, contributes to a growing patient pool. Favorable regulatory environments, exemplified by the FDA's expedited review pathways for orphan drugs and innovative oncology treatments, are encouraging investment and faster market entry. Economic factors, such as increased healthcare spending and insurance coverage for advanced cancer treatments, also play a crucial role. For instance, the growing investment in oncology research and development by major pharmaceutical companies, estimated to be over a billion dollars annually, directly fuels market expansion.
Challenges in the CTLC Industry Market
The CTLC industry faces several significant challenges that can impede its growth trajectory. Regulatory hurdles, while streamlined for some innovations, can still be complex and time-consuming, particularly for novel drug classes. Supply chain disruptions and manufacturing complexities for highly specialized biologics can lead to availability issues and increased costs, impacting market access. The high cost of advanced CTCL therapies poses a significant barrier for many patients and healthcare systems, raising concerns about affordability and equitable access. Furthermore, intense competitive pressures from established pharmaceutical giants and emerging biotechs necessitate continuous innovation and differentiation. The limited patient population for specific CTCL subtypes can also make market penetration and achieving economies of scale challenging, despite overall market growth.
Emerging Opportunities in CTLC Industry
Emerging opportunities within the CTLC industry are primarily catalysts for long-term growth, driven by groundbreaking technological breakthroughs and strategic market expansion initiatives. The increasing understanding of the tumor microenvironment in CTCL is paving the way for next-generation immunotherapies and combination treatments that target specific cellular pathways, offering significant therapeutic potential. Strategic partnerships between academic research institutions and pharmaceutical companies are accelerating the discovery and development of novel drug candidates. Market expansion strategies targeting underdiagnosed or underserved geographic regions, coupled with increased disease awareness campaigns, can unlock new patient populations. Furthermore, the development of companion diagnostics that enable personalized treatment selection represents a significant opportunity to optimize patient care and market penetration for targeted therapies.
Leading Players in the CTLC Industry Sector
- Innate Pharma
- Soligenix Inc.
- Kyowa Hakko Kirin Co Ltd
- Citius Pharmaceuticals Inc.
- Celgene Corporation
- Merck & Co Inc.
- Seattle Genetics
- Helsinn Healthcare SA
- Bristol Myers Squibb
- Equillium Inc (Bioniz Therapeutics)
- Bausch Health Companies Inc.
- miRagen Therapeutics
Key Milestones in CTLC Industry Industry
- Dec 2022: Citius Pharmaceuticals Inc. announced that the US FDA accepted the company's biologics license application (BLA) for denileukin diftitox, an engineered IL-2-diphtheria toxin fusion protein for the treatment of patients with persistent or recurrent CTCL. This milestone signifies a significant step towards potential market approval for a novel CTCL therapy.
- Jul 2022: Soligenix Inc. received an agreement from the US FDA on an initial pediatric study plan (iPSP) for HyBryte (synthetic hypericin) for the treatment of cutaneous T-cell lymphoma (CTCL). This agreement paves the way for further clinical development and potential regulatory submission for HyBryte.
Strategic Outlook for CTLC Industry Market
The strategic outlook for the CTLC industry is one of sustained growth and innovation. Future market potential will be significantly shaped by the continued advancement of personalized medicine approaches, including gene therapies and advanced immunotherapies tailored to specific CTCL subtypes and patient genetic profiles. Increased collaboration between pharmaceutical companies, research institutions, and patient advocacy groups will be crucial for driving clinical trial success and accelerating product development. Strategic opportunities lie in expanding the therapeutic armamentarium to address refractory disease and improve long-term remission rates. Furthermore, the development of cost-effective treatment strategies and improved access models will be critical for maximizing market penetration and ensuring that innovative therapies reach all eligible patients worldwide, contributing to a market size projected to exceed a billion dollars.
CTLC Industry Segmentation
-
1. Type
- 1.1. Mycosis Fungoides (MF)
- 1.2. Sezary Syndrome (SS)
- 1.3. Other Types
-
2. Treatment
- 2.1. Radiation Therapy
- 2.2. Chemotherapy
- 2.3. Immunotherapy
- 2.4. Targeted therapy
- 2.5. Other Treatments
CTLC Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
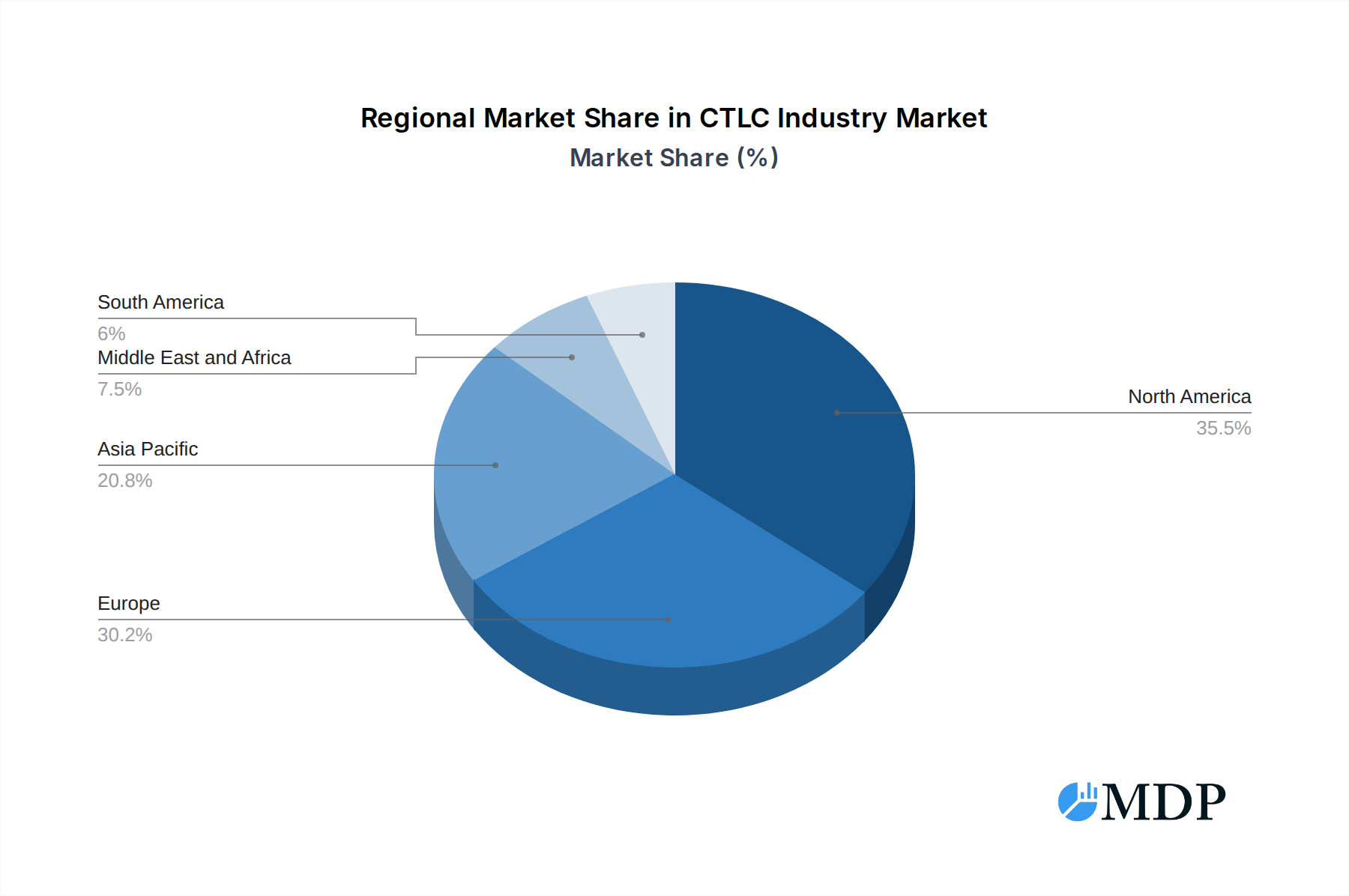
CTLC Industry Regional Market Share

Geographic Coverage of CTLC Industry
CTLC Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 12.82% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. MDP Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Type
- 5.1.1. Mycosis Fungoides (MF)
- 5.1.2. Sezary Syndrome (SS)
- 5.1.3. Other Types
- 5.2. Market Analysis, Insights and Forecast - by Treatment
- 5.2.1. Radiation Therapy
- 5.2.2. Chemotherapy
- 5.2.3. Immunotherapy
- 5.2.4. Targeted therapy
- 5.2.5. Other Treatments
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. Europe
- 5.3.3. Asia Pacific
- 5.3.4. Middle East and Africa
- 5.3.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Type
- 6. Global CTLC Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Type
- 6.1.1. Mycosis Fungoides (MF)
- 6.1.2. Sezary Syndrome (SS)
- 6.1.3. Other Types
- 6.2. Market Analysis, Insights and Forecast - by Treatment
- 6.2.1. Radiation Therapy
- 6.2.2. Chemotherapy
- 6.2.3. Immunotherapy
- 6.2.4. Targeted therapy
- 6.2.5. Other Treatments
- 6.1. Market Analysis, Insights and Forecast - by Type
- 7. North America CTLC Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Type
- 7.1.1. Mycosis Fungoides (MF)
- 7.1.2. Sezary Syndrome (SS)
- 7.1.3. Other Types
- 7.2. Market Analysis, Insights and Forecast - by Treatment
- 7.2.1. Radiation Therapy
- 7.2.2. Chemotherapy
- 7.2.3. Immunotherapy
- 7.2.4. Targeted therapy
- 7.2.5. Other Treatments
- 7.1. Market Analysis, Insights and Forecast - by Type
- 8. Europe CTLC Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Type
- 8.1.1. Mycosis Fungoides (MF)
- 8.1.2. Sezary Syndrome (SS)
- 8.1.3. Other Types
- 8.2. Market Analysis, Insights and Forecast - by Treatment
- 8.2.1. Radiation Therapy
- 8.2.2. Chemotherapy
- 8.2.3. Immunotherapy
- 8.2.4. Targeted therapy
- 8.2.5. Other Treatments
- 8.1. Market Analysis, Insights and Forecast - by Type
- 9. Asia Pacific CTLC Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Type
- 9.1.1. Mycosis Fungoides (MF)
- 9.1.2. Sezary Syndrome (SS)
- 9.1.3. Other Types
- 9.2. Market Analysis, Insights and Forecast - by Treatment
- 9.2.1. Radiation Therapy
- 9.2.2. Chemotherapy
- 9.2.3. Immunotherapy
- 9.2.4. Targeted therapy
- 9.2.5. Other Treatments
- 9.1. Market Analysis, Insights and Forecast - by Type
- 10. Middle East and Africa CTLC Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Type
- 10.1.1. Mycosis Fungoides (MF)
- 10.1.2. Sezary Syndrome (SS)
- 10.1.3. Other Types
- 10.2. Market Analysis, Insights and Forecast - by Treatment
- 10.2.1. Radiation Therapy
- 10.2.2. Chemotherapy
- 10.2.3. Immunotherapy
- 10.2.4. Targeted therapy
- 10.2.5. Other Treatments
- 10.1. Market Analysis, Insights and Forecast - by Type
- 11. South America CTLC Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Type
- 11.1.1. Mycosis Fungoides (MF)
- 11.1.2. Sezary Syndrome (SS)
- 11.1.3. Other Types
- 11.2. Market Analysis, Insights and Forecast - by Treatment
- 11.2.1. Radiation Therapy
- 11.2.2. Chemotherapy
- 11.2.3. Immunotherapy
- 11.2.4. Targeted therapy
- 11.2.5. Other Treatments
- 11.1. Market Analysis, Insights and Forecast - by Type
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Innate Pharma
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Soligenix Inc
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Kyowa Hakko Kirin Co Ltd
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Citius Pharmaceuticals Inc
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Celgene Corporation
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Merck & Co Inc
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Seattle Genetics
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Helsinn Healthcare SA
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Bristol Myers Squibb*List Not Exhaustive
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Equillium Inc (Bioniz Therapeutics)
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Bausch Health Companies Inc
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 miRagen Therapeutics
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.1 Innate Pharma
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global CTLC Industry Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America CTLC Industry Revenue (billion), by Type 2025 & 2033
- Figure 3: North America CTLC Industry Revenue Share (%), by Type 2025 & 2033
- Figure 4: North America CTLC Industry Revenue (billion), by Treatment 2025 & 2033
- Figure 5: North America CTLC Industry Revenue Share (%), by Treatment 2025 & 2033
- Figure 6: North America CTLC Industry Revenue (billion), by Country 2025 & 2033
- Figure 7: North America CTLC Industry Revenue Share (%), by Country 2025 & 2033
- Figure 8: Europe CTLC Industry Revenue (billion), by Type 2025 & 2033
- Figure 9: Europe CTLC Industry Revenue Share (%), by Type 2025 & 2033
- Figure 10: Europe CTLC Industry Revenue (billion), by Treatment 2025 & 2033
- Figure 11: Europe CTLC Industry Revenue Share (%), by Treatment 2025 & 2033
- Figure 12: Europe CTLC Industry Revenue (billion), by Country 2025 & 2033
- Figure 13: Europe CTLC Industry Revenue Share (%), by Country 2025 & 2033
- Figure 14: Asia Pacific CTLC Industry Revenue (billion), by Type 2025 & 2033
- Figure 15: Asia Pacific CTLC Industry Revenue Share (%), by Type 2025 & 2033
- Figure 16: Asia Pacific CTLC Industry Revenue (billion), by Treatment 2025 & 2033
- Figure 17: Asia Pacific CTLC Industry Revenue Share (%), by Treatment 2025 & 2033
- Figure 18: Asia Pacific CTLC Industry Revenue (billion), by Country 2025 & 2033
- Figure 19: Asia Pacific CTLC Industry Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East and Africa CTLC Industry Revenue (billion), by Type 2025 & 2033
- Figure 21: Middle East and Africa CTLC Industry Revenue Share (%), by Type 2025 & 2033
- Figure 22: Middle East and Africa CTLC Industry Revenue (billion), by Treatment 2025 & 2033
- Figure 23: Middle East and Africa CTLC Industry Revenue Share (%), by Treatment 2025 & 2033
- Figure 24: Middle East and Africa CTLC Industry Revenue (billion), by Country 2025 & 2033
- Figure 25: Middle East and Africa CTLC Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America CTLC Industry Revenue (billion), by Type 2025 & 2033
- Figure 27: South America CTLC Industry Revenue Share (%), by Type 2025 & 2033
- Figure 28: South America CTLC Industry Revenue (billion), by Treatment 2025 & 2033
- Figure 29: South America CTLC Industry Revenue Share (%), by Treatment 2025 & 2033
- Figure 30: South America CTLC Industry Revenue (billion), by Country 2025 & 2033
- Figure 31: South America CTLC Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global CTLC Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 2: Global CTLC Industry Revenue billion Forecast, by Treatment 2020 & 2033
- Table 3: Global CTLC Industry Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Global CTLC Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 5: Global CTLC Industry Revenue billion Forecast, by Treatment 2020 & 2033
- Table 6: Global CTLC Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 7: United States CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Canada CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 9: Mexico CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Global CTLC Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 11: Global CTLC Industry Revenue billion Forecast, by Treatment 2020 & 2033
- Table 12: Global CTLC Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Germany CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: United Kingdom CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: France CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Italy CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 17: Spain CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 18: Rest of Europe CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 19: Global CTLC Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 20: Global CTLC Industry Revenue billion Forecast, by Treatment 2020 & 2033
- Table 21: Global CTLC Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 22: China CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Japan CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: India CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 25: Australia CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: South Korea CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Rest of Asia Pacific CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Global CTLC Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 29: Global CTLC Industry Revenue billion Forecast, by Treatment 2020 & 2033
- Table 30: Global CTLC Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 31: GCC CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: South Africa CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 33: Rest of Middle East and Africa CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: Global CTLC Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 35: Global CTLC Industry Revenue billion Forecast, by Treatment 2020 & 2033
- Table 36: Global CTLC Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 37: Brazil CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 38: Argentina CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 39: Rest of South America CTLC Industry Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the CTLC Industry?
The projected CAGR is approximately 12.82%.
2. Which companies are prominent players in the CTLC Industry?
Key companies in the market include Innate Pharma, Soligenix Inc, Kyowa Hakko Kirin Co Ltd, Citius Pharmaceuticals Inc, Celgene Corporation, Merck & Co Inc, Seattle Genetics, Helsinn Healthcare SA, Bristol Myers Squibb*List Not Exhaustive, Equillium Inc (Bioniz Therapeutics), Bausch Health Companies Inc, miRagen Therapeutics.
3. What are the main segments of the CTLC Industry?
The market segments include Type, Treatment.
4. Can you provide details about the market size?
The market size is estimated to be USD 6.84 billion as of 2022.
5. What are some drivers contributing to market growth?
Rise in Lymphoma Cancer Incidence; Growing Geriatric Population.
6. What are the notable trends driving market growth?
Mycosis Fungoides Segment is Expected to Register a Significant Growth in the Forecast Years.
7. Are there any restraints impacting market growth?
High Cost of Treatment.
8. Can you provide examples of recent developments in the market?
Dec 2022: Citius Pharmaceuticals Inc. announced that the US FDA accepted the company's biologics license application (BLA) for denileukin diftitox, an engineered IL-2-diphtheria toxin fusion protein for the treatment of patients with persistent or recurrent CTCL.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "CTLC Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the CTLC Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the CTLC Industry?
To stay informed about further developments, trends, and reports in the CTLC Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


