Key Insights
The global Buparlisib market is poised for significant expansion, projected to reach an estimated USD 6.04 billion in 2025. This robust growth is propelled by a remarkable Compound Annual Growth Rate (CAGR) of 16.57% during the forecast period of 2025-2033. A primary driver behind this surge is the increasing prevalence of cancers where Buparlisib, an inhibitor of the PI3K pathway, demonstrates therapeutic potential. Advancements in precision medicine and ongoing clinical trials exploring its efficacy in combination therapies are further fueling market demand. The research segment, crucial for drug discovery and development, is expected to remain a dominant force, alongside the expanding medical applications for treating various oncological conditions. The market is also witnessing a growing demand for higher purity grades of Buparlisib, particularly those exceeding 99%, essential for stringent pharmaceutical research and development protocols.
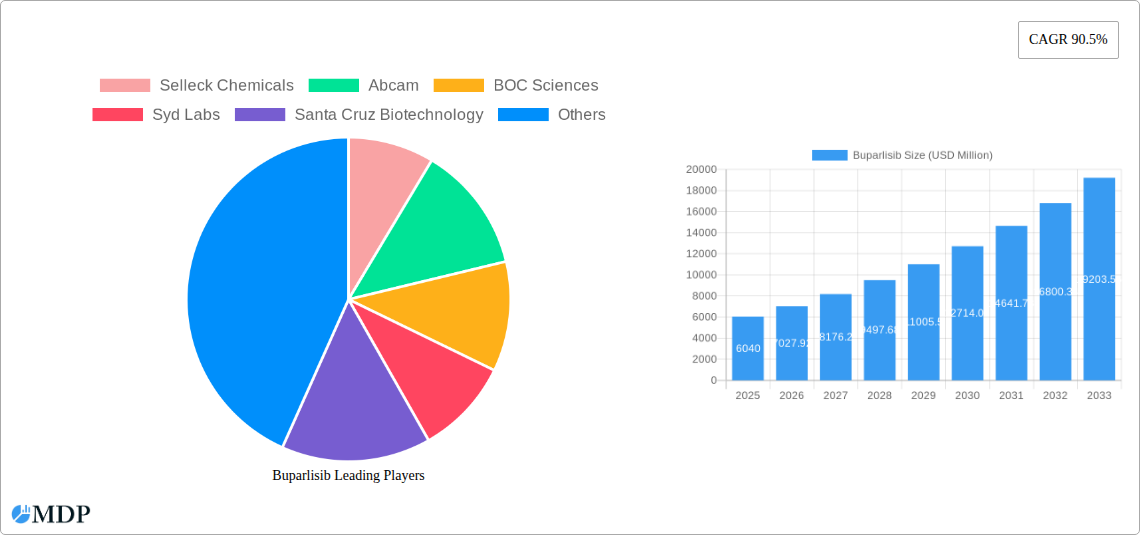
Buparlisib Market Size (In Billion)

Key trends shaping the Buparlisib market include the relentless pursuit of novel cancer treatments and a deeper understanding of the PI3K signaling pathway's role in tumorigenesis. The increasing investment in pharmaceutical R&D, coupled with collaborations between research institutions and biopharmaceutical companies, is accelerating the development and commercialization of Buparlisib-based therapies. Geographically, North America and Europe are anticipated to lead market share, driven by advanced healthcare infrastructure, significant R&D spending, and the presence of major pharmaceutical players. However, the Asia Pacific region is emerging as a high-growth market due to increasing healthcare expenditure, a growing patient population, and favorable government initiatives supporting drug development. While the market is largely driven by its therapeutic potential, potential challenges such as stringent regulatory approvals for new indications and the high cost of drug development may present some restraints.
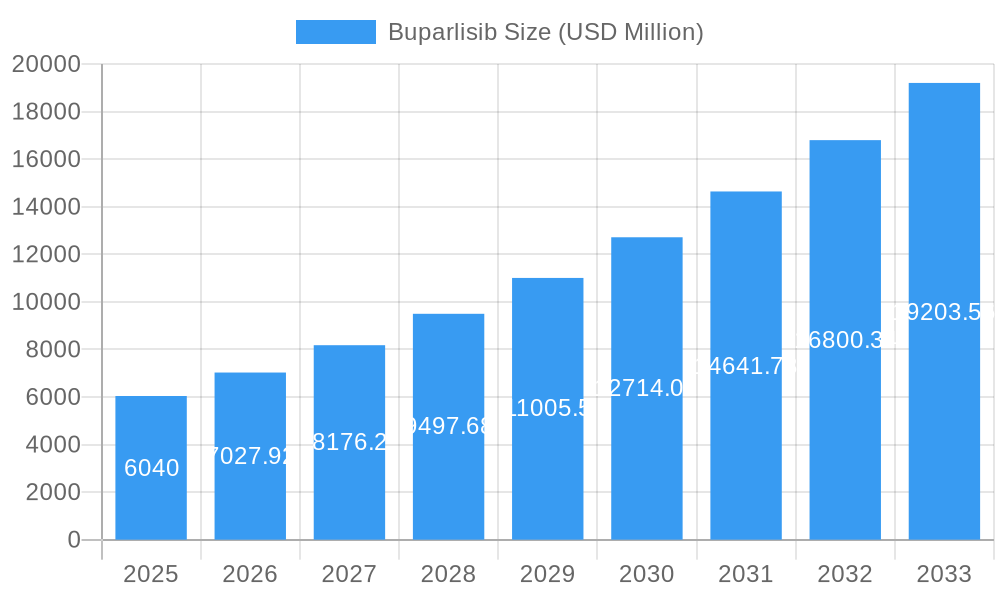
Buparlisib Company Market Share

Buparlisib Market Report: Innovation, Growth, and Strategic Opportunities (2019-2033)
This comprehensive Buparlisib market report provides an in-depth analysis of a crucial therapeutic agent, offering unparalleled insights for industry stakeholders. Covering a dynamic study period from 2019 to 2033, with a base and estimated year of 2025, this report delves into market dynamics, emerging trends, leading segments, product developments, growth drivers, challenges, and strategic opportunities within the Buparlisib sector. Leveraging high-traffic keywords and structured for maximum SEO visibility, this report is essential for researchers, pharmaceutical companies, investors, and regulatory bodies seeking to navigate and capitalize on the evolving Buparlisib landscape.
Buparlisib Market Dynamics & Concentration
The Buparlisib market is characterized by a moderate concentration of key players, with innovation being a primary driver of competition. Regulatory frameworks, particularly those governing pharmaceutical research and clinical trials, play a significant role in shaping market entry and product development. The primary application of Buparlisib lies in research settings and burgeoning medical applications, where its PI3K inhibitory properties are being extensively investigated. Product substitutes, though present in the broader oncology landscape, are generally not direct replacements for Buparlisib's specific mechanism of action. End-user trends indicate a growing demand for targeted therapies, fueling advancements in Buparlisib research and development. Mergers and acquisitions (M&A) activities within the broader oncology drug development space may indirectly impact Buparlisib's market position, with an estimated XX M&A deals observed in the historical period (2019-2024) and an anticipated XX deals in the forecast period (2025-2033). Market share analysis reveals that leading research chemical suppliers hold substantial influence, with estimates suggesting that the top five companies collectively command over 50% of the Buparlisib research reagent market.
Buparlisib Industry Trends & Analysis
The Buparlisib industry is poised for significant expansion, driven by a confluence of factors including the increasing prevalence of cancer and the growing demand for targeted therapies. Technological disruptions, particularly in drug discovery and development, are accelerating research into Buparlisib's therapeutic potential. Consumer preferences are shifting towards personalized medicine and treatments with improved efficacy and reduced side effects, a trend that Buparlisib, as a PI3K inhibitor, is well-positioned to address. Competitive dynamics are intensifying as more research institutions and pharmaceutical companies explore Buparlisib's applications in various oncological indications. The Compound Annual Growth Rate (CAGR) for the Buparlisib market is projected to be xx% during the forecast period of 2025–2033. Market penetration is currently robust in the research segment, with increasing efforts to translate these findings into clinical applications. The ongoing exploration of Buparlisib's role in combination therapies, its potential in treating specific genetic mutations, and the continuous refinement of its delivery mechanisms are key trends influencing market trajectory. Furthermore, advancements in diagnostic tools that identify patient populations most likely to respond to PI3K inhibition are directly impacting the demand for Buparlisib and similar compounds. The economic landscape also plays a role, with increasing healthcare expenditure globally supporting the development and adoption of novel cancer treatments. Regulatory bodies are closely monitoring research and clinical trial outcomes, which will ultimately shape the approval pathways and market access for Buparlisib in medical applications. The commitment of research institutions and pharmaceutical giants to uncovering the full therapeutic spectrum of Buparlisib underscores its significance in the future of cancer treatment.
Leading Markets & Segments in Buparlisib
The Buparlisib market is predominantly led by the Research application segment, driven by its extensive use in preclinical studies and academic investigations into cancer biology. Within the Types segmentation, the Min Purity 98%-99% and Min Purity More Than 99% categories hold significant dominance, reflecting the stringent purity requirements for reliable scientific research and potential therapeutic development. The Medical application segment, while currently smaller, is experiencing rapid growth due to ongoing clinical trials and the increasing focus on targeted therapies in oncology.
- Dominant Region: North America is currently the leading market for Buparlisib, propelled by substantial investment in cancer research, a well-established pharmaceutical industry, and a high incidence of cancer.
- Key Country: The United States, within North America, stands out as a primary hub for Buparlisib consumption and research, benefiting from extensive funding for academic institutions and biopharmaceutical companies.
- Dominant Application Segment (Research): The "Research" application segment is fueled by its indispensable role in understanding PI3K pathway dysregulation in various cancers. Key drivers include:
- Technological Advancements in Oncology: Sophisticated research tools and techniques enable deeper exploration of Buparlisib's mechanisms.
- Increased Funding for Cancer Research: Government and private sector grants support extensive preclinical and clinical investigations.
- Growing Understanding of PI3K Pathway: Scientific literature continuously highlights the PI3K pathway's critical role in tumor growth and survival.
- Dominant Purity Segment (Min Purity 98%-99% & Min Purity More Than 99%): High-purity Buparlisib is crucial for:
- Reproducible Research Outcomes: Ensuring the reliability and validity of scientific findings.
- Minimizing Off-Target Effects: Critical for both research and potential therapeutic applications.
- Regulatory Compliance: Meeting the stringent quality standards for pharmaceutical-grade compounds.
- Emerging Medical Applications: The "Medical" segment's growth is driven by:
- Clinical Trial Advancements: Positive outcomes in ongoing trials for various cancer types.
- Targeted Therapy Demand: Growing patient and physician preference for personalized treatment strategies.
- Biomarker Discovery: Identification of biomarkers that predict patient response to PI3K inhibitors.
Buparlisib Product Developments
Product developments in the Buparlisib sector are primarily focused on enhancing its therapeutic efficacy and expanding its application spectrum. Innovations include the development of optimized formulations for improved bioavailability and reduced toxicity, as well as the exploration of Buparlisib in combination therapies with other anti-cancer agents to overcome resistance mechanisms. Competitive advantages are being carved out through stringent quality control measures ensuring high purity levels (Min Purity More Than 99%), crucial for both research and clinical translation. Furthermore, ongoing research into specific genetic mutations associated with PI3K pathway activation is enabling the development of more targeted therapeutic strategies involving Buparlisib.
Key Drivers of Buparlisib Growth
Several key factors are propelling the growth of the Buparlisib market. Technological advancements in cancer research, including sophisticated molecular biology techniques and high-throughput screening, are uncovering new therapeutic avenues for Buparlisib. Economic factors, such as increasing healthcare expenditure and investment in biopharmaceutical R&D globally, provide the financial impetus for further development. Regulatory frameworks that encourage the approval of innovative oncology treatments, coupled with a growing understanding of the PI3K pathway's role in various cancers, are also significant growth drivers. The increasing demand for targeted therapies, driven by a desire for more effective and less toxic cancer treatments, further fuels market expansion.
Challenges in the Buparlisib Market
Despite its promising therapeutic potential, the Buparlisib market faces certain challenges. Regulatory hurdles associated with drug approval processes, especially for novel targeted therapies, can be lengthy and demanding. Supply chain complexities for specialized research chemicals and potential pharmaceutical-grade compounds can also pose logistical challenges, impacting availability and cost. Competitive pressures from other PI3K inhibitors and alternative cancer treatment modalities necessitate continuous innovation and demonstration of superior efficacy. Furthermore, understanding and managing potential off-target effects and toxicity profiles remain critical areas of focus for widespread clinical adoption, impacting market penetration.
Emerging Opportunities in Buparlisib
Emerging opportunities in the Buparlisib market are primarily driven by technological breakthroughs in precision medicine and a deeper understanding of cancer genomics. The identification of specific biomarkers that predict patient response to PI3K inhibition presents a significant opportunity for developing personalized treatment strategies. Strategic partnerships between research institutions and pharmaceutical companies are crucial for accelerating clinical trials and bringing Buparlisib closer to market approval for various oncological indications. Market expansion strategies targeting underserved cancer patient populations and exploring Buparlisib's potential in rare or refractory cancers represent significant long-term growth catalysts.
Leading Players in the Buparlisib Sector
- Selleck Chemicals
- Abcam
- BOC Sciences
- Syd Labs
- Santa Cruz Biotechnology
- Cayman Chemical
- BioVision
- Shimadzu
- Biosynth Carbosynth
- Adooq Bioscience
- Axon Medchem
- TargetMol
- Enzo Biochem
- LGC
- LKT Laboratories
- InvivoChem
- J&K Scientific
Key Milestones in Buparlisib Industry
- 2019: Initial preclinical studies demonstrating Buparlisib's efficacy in various cancer models published.
- 2020: Initiation of early-phase clinical trials investigating Buparlisib for specific solid tumors.
- 2021: Publication of research detailing Buparlisib's mechanism of action and potential synergistic effects with other agents.
- 2022: Expansion of clinical trials to include a broader range of cancer types and patient populations.
- 2023: Advancements in Buparlisib formulation research aiming to improve patient tolerability and drug delivery.
- 2024: Increased interest from pharmaceutical companies in licensing or acquiring Buparlisib-related intellectual property.
- 2025 (Estimated): Publication of Phase II clinical trial data with promising efficacy and safety profiles.
- 2026-2030: Expected progression to Phase III clinical trials for key indications.
- 2031-2033: Potential regulatory submissions and market approvals for specific Buparlisib-based therapies.
Strategic Outlook for Buparlisib Market
The strategic outlook for the Buparlisib market is characterized by a strong emphasis on continued research and development to unlock its full therapeutic potential. Growth accelerators will include the successful translation of preclinical findings into robust clinical trial outcomes, leading to regulatory approvals for specific cancer indications. The expansion of Buparlisib into combination therapies, synergistic with emerging immunotherapies and targeted agents, presents a significant avenue for market growth. Strategic partnerships and collaborations will be crucial for navigating the complex drug development landscape, and a focus on precision medicine, driven by advanced biomarker identification, will define future market success. The increasing global demand for innovative cancer treatments will undoubtedly fuel further investment and interest in Buparlisib's evolution.
Buparlisib Segmentation
-
1. Application
- 1.1. Research
- 1.2. Medical
-
2. Types
- 2.1. Min Purity Less Than 98%
- 2.2. Min Purity 98%-99%
- 2.3. Min Purity More Than 99%
Buparlisib Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific

Buparlisib Regional Market Share

Geographic Coverage of Buparlisib
Buparlisib REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 17.5% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Buparlisib Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Research
- 5.1.2. Medical
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Min Purity Less Than 98%
- 5.2.2. Min Purity 98%-99%
- 5.2.3. Min Purity More Than 99%
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Buparlisib Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Research
- 6.1.2. Medical
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Min Purity Less Than 98%
- 6.2.2. Min Purity 98%-99%
- 6.2.3. Min Purity More Than 99%
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Buparlisib Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Research
- 7.1.2. Medical
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Min Purity Less Than 98%
- 7.2.2. Min Purity 98%-99%
- 7.2.3. Min Purity More Than 99%
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Buparlisib Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Research
- 8.1.2. Medical
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Min Purity Less Than 98%
- 8.2.2. Min Purity 98%-99%
- 8.2.3. Min Purity More Than 99%
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Buparlisib Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Research
- 9.1.2. Medical
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Min Purity Less Than 98%
- 9.2.2. Min Purity 98%-99%
- 9.2.3. Min Purity More Than 99%
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Buparlisib Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Research
- 10.1.2. Medical
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Min Purity Less Than 98%
- 10.2.2. Min Purity 98%-99%
- 10.2.3. Min Purity More Than 99%
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Selleck Chemicals
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Abcam
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 BOC Sciences
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Syd Labs
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Santa Cruz Biotechnology
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Cayman Chemical
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 BioVision
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Shimadzu
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Biosynth Carbosynth
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Adooq Bioscience
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Axon Medchem
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 TargetMol
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Enzo Biochem
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 LGC
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 LKT Laboratories
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 InvivoChem
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 J&K Scientific
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.1 Selleck Chemicals
List of Figures
- Figure 1: Global Buparlisib Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: Global Buparlisib Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America Buparlisib Revenue (undefined), by Application 2025 & 2033
- Figure 4: North America Buparlisib Volume (K), by Application 2025 & 2033
- Figure 5: North America Buparlisib Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Buparlisib Volume Share (%), by Application 2025 & 2033
- Figure 7: North America Buparlisib Revenue (undefined), by Types 2025 & 2033
- Figure 8: North America Buparlisib Volume (K), by Types 2025 & 2033
- Figure 9: North America Buparlisib Revenue Share (%), by Types 2025 & 2033
- Figure 10: North America Buparlisib Volume Share (%), by Types 2025 & 2033
- Figure 11: North America Buparlisib Revenue (undefined), by Country 2025 & 2033
- Figure 12: North America Buparlisib Volume (K), by Country 2025 & 2033
- Figure 13: North America Buparlisib Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Buparlisib Volume Share (%), by Country 2025 & 2033
- Figure 15: South America Buparlisib Revenue (undefined), by Application 2025 & 2033
- Figure 16: South America Buparlisib Volume (K), by Application 2025 & 2033
- Figure 17: South America Buparlisib Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America Buparlisib Volume Share (%), by Application 2025 & 2033
- Figure 19: South America Buparlisib Revenue (undefined), by Types 2025 & 2033
- Figure 20: South America Buparlisib Volume (K), by Types 2025 & 2033
- Figure 21: South America Buparlisib Revenue Share (%), by Types 2025 & 2033
- Figure 22: South America Buparlisib Volume Share (%), by Types 2025 & 2033
- Figure 23: South America Buparlisib Revenue (undefined), by Country 2025 & 2033
- Figure 24: South America Buparlisib Volume (K), by Country 2025 & 2033
- Figure 25: South America Buparlisib Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Buparlisib Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe Buparlisib Revenue (undefined), by Application 2025 & 2033
- Figure 28: Europe Buparlisib Volume (K), by Application 2025 & 2033
- Figure 29: Europe Buparlisib Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe Buparlisib Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe Buparlisib Revenue (undefined), by Types 2025 & 2033
- Figure 32: Europe Buparlisib Volume (K), by Types 2025 & 2033
- Figure 33: Europe Buparlisib Revenue Share (%), by Types 2025 & 2033
- Figure 34: Europe Buparlisib Volume Share (%), by Types 2025 & 2033
- Figure 35: Europe Buparlisib Revenue (undefined), by Country 2025 & 2033
- Figure 36: Europe Buparlisib Volume (K), by Country 2025 & 2033
- Figure 37: Europe Buparlisib Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe Buparlisib Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa Buparlisib Revenue (undefined), by Application 2025 & 2033
- Figure 40: Middle East & Africa Buparlisib Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa Buparlisib Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa Buparlisib Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa Buparlisib Revenue (undefined), by Types 2025 & 2033
- Figure 44: Middle East & Africa Buparlisib Volume (K), by Types 2025 & 2033
- Figure 45: Middle East & Africa Buparlisib Revenue Share (%), by Types 2025 & 2033
- Figure 46: Middle East & Africa Buparlisib Volume Share (%), by Types 2025 & 2033
- Figure 47: Middle East & Africa Buparlisib Revenue (undefined), by Country 2025 & 2033
- Figure 48: Middle East & Africa Buparlisib Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa Buparlisib Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa Buparlisib Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific Buparlisib Revenue (undefined), by Application 2025 & 2033
- Figure 52: Asia Pacific Buparlisib Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific Buparlisib Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific Buparlisib Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific Buparlisib Revenue (undefined), by Types 2025 & 2033
- Figure 56: Asia Pacific Buparlisib Volume (K), by Types 2025 & 2033
- Figure 57: Asia Pacific Buparlisib Revenue Share (%), by Types 2025 & 2033
- Figure 58: Asia Pacific Buparlisib Volume Share (%), by Types 2025 & 2033
- Figure 59: Asia Pacific Buparlisib Revenue (undefined), by Country 2025 & 2033
- Figure 60: Asia Pacific Buparlisib Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific Buparlisib Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific Buparlisib Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Buparlisib Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Buparlisib Volume K Forecast, by Application 2020 & 2033
- Table 3: Global Buparlisib Revenue undefined Forecast, by Types 2020 & 2033
- Table 4: Global Buparlisib Volume K Forecast, by Types 2020 & 2033
- Table 5: Global Buparlisib Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global Buparlisib Volume K Forecast, by Region 2020 & 2033
- Table 7: Global Buparlisib Revenue undefined Forecast, by Application 2020 & 2033
- Table 8: Global Buparlisib Volume K Forecast, by Application 2020 & 2033
- Table 9: Global Buparlisib Revenue undefined Forecast, by Types 2020 & 2033
- Table 10: Global Buparlisib Volume K Forecast, by Types 2020 & 2033
- Table 11: Global Buparlisib Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: Global Buparlisib Volume K Forecast, by Country 2020 & 2033
- Table 13: United States Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: United States Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Canada Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 18: Mexico Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global Buparlisib Revenue undefined Forecast, by Application 2020 & 2033
- Table 20: Global Buparlisib Volume K Forecast, by Application 2020 & 2033
- Table 21: Global Buparlisib Revenue undefined Forecast, by Types 2020 & 2033
- Table 22: Global Buparlisib Volume K Forecast, by Types 2020 & 2033
- Table 23: Global Buparlisib Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: Global Buparlisib Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Brazil Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Argentina Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global Buparlisib Revenue undefined Forecast, by Application 2020 & 2033
- Table 32: Global Buparlisib Volume K Forecast, by Application 2020 & 2033
- Table 33: Global Buparlisib Revenue undefined Forecast, by Types 2020 & 2033
- Table 34: Global Buparlisib Volume K Forecast, by Types 2020 & 2033
- Table 35: Global Buparlisib Revenue undefined Forecast, by Country 2020 & 2033
- Table 36: Global Buparlisib Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 40: Germany Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: France Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: Italy Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Spain Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 48: Russia Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 50: Benelux Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 52: Nordics Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global Buparlisib Revenue undefined Forecast, by Application 2020 & 2033
- Table 56: Global Buparlisib Volume K Forecast, by Application 2020 & 2033
- Table 57: Global Buparlisib Revenue undefined Forecast, by Types 2020 & 2033
- Table 58: Global Buparlisib Volume K Forecast, by Types 2020 & 2033
- Table 59: Global Buparlisib Revenue undefined Forecast, by Country 2020 & 2033
- Table 60: Global Buparlisib Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 62: Turkey Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 64: Israel Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 66: GCC Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 68: North Africa Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 70: South Africa Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global Buparlisib Revenue undefined Forecast, by Application 2020 & 2033
- Table 74: Global Buparlisib Volume K Forecast, by Application 2020 & 2033
- Table 75: Global Buparlisib Revenue undefined Forecast, by Types 2020 & 2033
- Table 76: Global Buparlisib Volume K Forecast, by Types 2020 & 2033
- Table 77: Global Buparlisib Revenue undefined Forecast, by Country 2020 & 2033
- Table 78: Global Buparlisib Volume K Forecast, by Country 2020 & 2033
- Table 79: China Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 80: China Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 82: India Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 84: Japan Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 86: South Korea Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 88: ASEAN Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 90: Oceania Buparlisib Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific Buparlisib Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific Buparlisib Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Buparlisib?
The projected CAGR is approximately 17.5%.
2. Which companies are prominent players in the Buparlisib?
Key companies in the market include Selleck Chemicals, Abcam, BOC Sciences, Syd Labs, Santa Cruz Biotechnology, Cayman Chemical, BioVision, Shimadzu, Biosynth Carbosynth, Adooq Bioscience, Axon Medchem, TargetMol, Enzo Biochem, LGC, LKT Laboratories, InvivoChem, J&K Scientific.
3. What are the main segments of the Buparlisib?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3950.00, USD 5925.00, and USD 7900.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Buparlisib," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Buparlisib report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Buparlisib?
To stay informed about further developments, trends, and reports in the Buparlisib, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


