Key Insights
The global medical biomedical materials market is poised for significant expansion, projected to reach USD 3.5 billion in 2024, with a robust Compound Annual Growth Rate (CAGR) of 8.6% from 2025 to 2033. This impressive growth is primarily fueled by an increasing demand for advanced biomaterials in a wide array of medical applications, including sophisticated surgical sutures, advanced fracture fixation devices, innovative oral implants, and targeted drug delivery microspheres. The rising prevalence of chronic diseases, an aging global population, and continuous technological advancements in healthcare are collectively driving the adoption of these high-performance materials. Furthermore, the expanding scope of regenerative medicine and minimally invasive surgical techniques are creating new avenues for material innovation and market penetration.
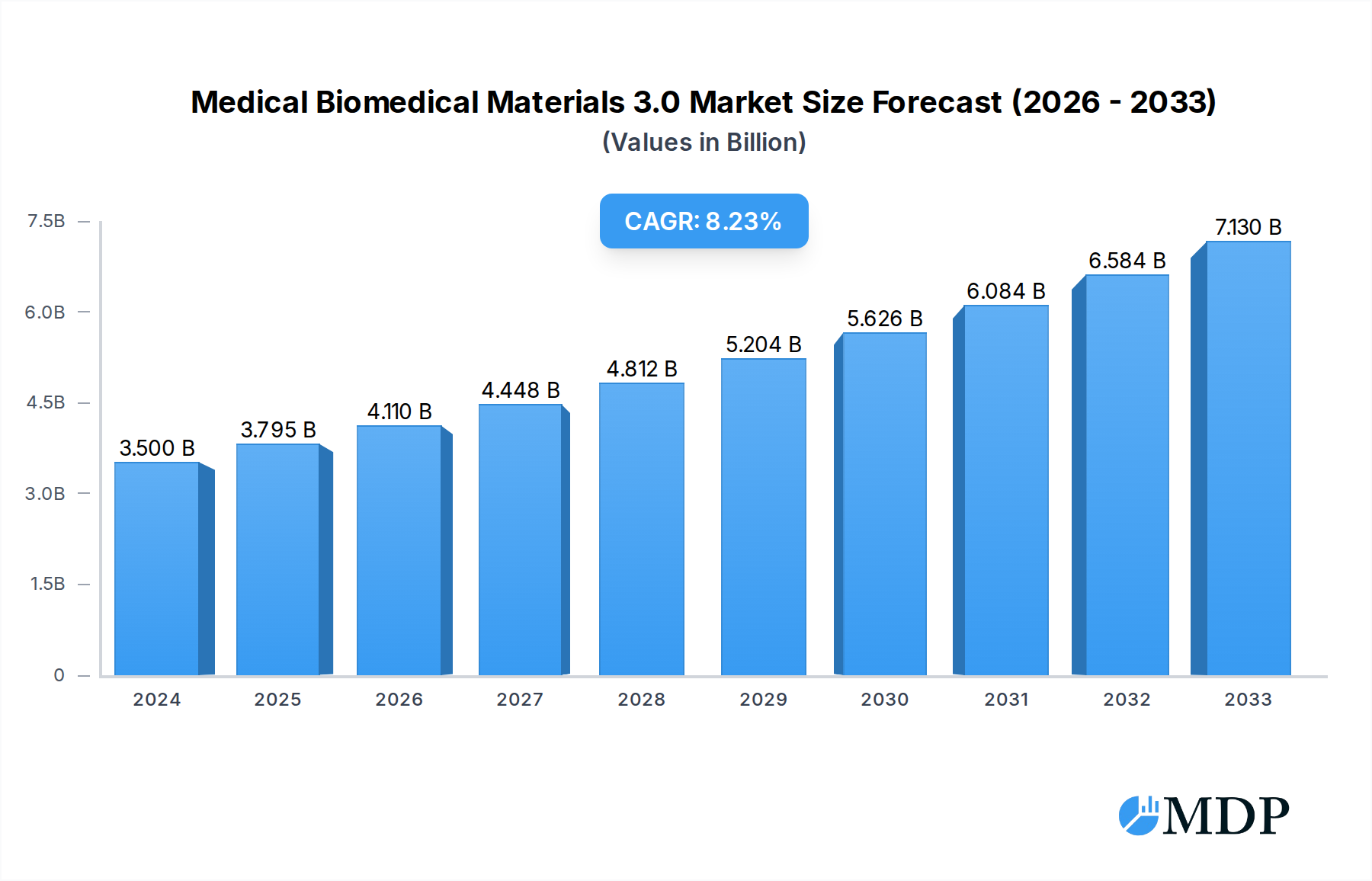
Medical Biomedical Materials 3.0 Market Size (In Billion)

Key market drivers include the growing need for biocompatible and biodegradable materials that can reduce adverse reactions and improve patient outcomes. Innovations in polymer science, such as the development of novel polylactic acid (PLA), polyglycolic acid (PGA), and their copolymers (PLGA, PLCG), are enabling the creation of materials with tailored degradation rates and mechanical properties. While the market presents substantial opportunities, it also faces certain restraints. High research and development costs, stringent regulatory approvals, and the availability of traditional materials in certain cost-sensitive applications could pose challenges. However, the persistent pursuit of enhanced therapeutic efficacy and improved patient quality of life, coupled with increasing healthcare expenditure globally, are expected to outweigh these restraints, solidifying the upward trajectory of the medical biomedical materials market.
Medical Biomedical Materials 3.0 Company Market Share

Medical Biomedical Materials 3.0: Market Dynamics, Trends, and Strategic Outlook 2019-2033
This comprehensive report, "Medical Biomedical Materials 3.0," delves deep into the rapidly evolving landscape of advanced materials shaping the future of healthcare. With an estimated market value projected to reach billions by 2033, this study offers critical insights into market concentration, innovation drivers, regulatory frameworks, and the strategic maneuvers of key industry players. Covering a detailed study period from 2019 to 2033, with a base and estimated year of 2025 and a forecast period of 2025–2033, this report is an indispensable resource for stakeholders seeking to navigate and capitalize on the burgeoning opportunities within the medical biomedical materials sector.
Medical Biomedical Materials 3.0 Market Dynamics & Concentration
The Medical Biomedical Materials 3.0 market is characterized by a dynamic interplay of established giants and agile innovators, with market concentration influenced by patent landscapes and proprietary manufacturing processes. Key innovation drivers include the relentless pursuit of biocompatibility, biodegradability, and tailored mechanical properties for a myriad of medical applications. Regulatory frameworks, while crucial for patient safety, also present significant hurdles, demanding extensive testing and validation. Product substitutes, often stemming from advancements in traditional materials or novel composite structures, constantly challenge existing market shares. End-user trends are overwhelmingly driven by minimally invasive procedures, personalized medicine, and the increasing demand for implantable devices with extended lifespans and improved integration. Mergers and acquisitions (M&A) activities, with an estimated count of over billion deals historically and projected to grow by billion in the forecast period, are a testament to the strategic consolidation and pursuit of synergistic capabilities within the industry. The market share of leading entities is expected to fluctuate as technological breakthroughs redefine product efficacy and market demand. The increasing emphasis on patient outcomes and reduced healthcare costs continues to shape the strategic decisions of companies operating within this vital sector.
Medical Biomedical Materials 3.0 Industry Trends & Analysis
The Medical Biomedical Materials 3.0 industry is poised for substantial growth, driven by a convergence of technological advancements, escalating healthcare needs, and a growing global aging population. The compound annual growth rate (CAGR) is projected to be robust, estimated at billions% over the forecast period. Market penetration is steadily increasing across diverse applications, from advanced drug delivery systems to sophisticated orthopedic implants. Technological disruptions, such as the development of smart biomaterials capable of responding to physiological cues and the integration of additive manufacturing (3D printing) for patient-specific implants, are revolutionizing product design and functionality. Consumer preferences are leaning towards materials that offer superior biocompatibility, reduce the risk of rejection or inflammation, and promote faster healing and tissue regeneration. The competitive dynamics are intensifying, with companies investing heavily in research and development to secure a competitive edge. The increasing prevalence of chronic diseases and the expanding healthcare infrastructure in emerging economies are significant market growth drivers. Furthermore, the demand for biodegradable and bioresorbable materials is on the rise, aligning with global sustainability initiatives and reducing the need for secondary removal surgeries. The pursuit of cost-effective yet high-performance materials also plays a crucial role in shaping market trends. The integration of artificial intelligence and machine learning in material design and discovery is further accelerating innovation. The global expenditure on healthcare materials is projected to exceed billions annually by the end of the forecast period, underscoring the immense market potential.
Leading Markets & Segments in Medical Biomedical Materials 3.0
The Medical Biomedical Materials 3.0 market exhibits significant regional dominance, with North America currently leading due to robust R&D investments, advanced healthcare infrastructure, and a high prevalence of target diseases. The United States, in particular, remains a critical hub for innovation and market penetration. Within this leading region, the Drug Delivery Microsphere application segment is experiencing exceptional growth, driven by the increasing demand for targeted and controlled release of therapeutics, particularly in oncology and chronic disease management. The material type dominating this segment and the broader market is PLGA, owing to its excellent biocompatibility, tunable degradation rates, and established safety profile.
- Dominant Region: North America
- Key Drivers:
- Significant government and private funding for biomedical research and development.
- High adoption rate of advanced medical technologies and devices.
- Strong presence of leading pharmaceutical and medical device companies.
- Favorable regulatory pathways for innovative medical materials.
- Aging population with increasing healthcare needs.
- Key Drivers:
- Dominant Country: United States
- Detailed Dominance Analysis: The US market benefits from a mature healthcare system, extensive clinical trial capabilities, and a large patient pool. Strategic collaborations between academic institutions and industry players foster rapid innovation. The growing demand for personalized medicine further propels the adoption of advanced biomedical materials.
- Dominant Application Segment: Drug Delivery Microsphere
- Detailed Dominance Analysis: The development of novel drug delivery systems, including biodegradable microspheres for sustained drug release and targeted therapy, is a major growth catalyst. This segment is driven by advancements in polymer science and nanotechnology, enabling more effective and safer drug administration.
- Dominant Material Type: PLGA (Poly(lactic-co-glycolic acid))
- Detailed Dominance Analysis: PLGA's versatility in terms of biodegradability and its ability to encapsulate a wide range of active pharmaceutical ingredients make it a cornerstone material. Its application spans from controlled drug release to tissue engineering scaffolds, further solidifying its market leadership. The established safety and efficacy profiles of PLGA-based products contribute significantly to its widespread adoption.
Medical Biomedical Materials 3.0 Product Developments
Product development in Medical Biomedical Materials 3.0 is heavily focused on enhanced biocompatibility, biodegradability, and tailored mechanical properties. Innovations include novel PLA and PLGA copolymers with precisely controlled degradation kinetics for improved long-term implant performance and reduced inflammatory responses. Advancements in PLCG materials are enabling the creation of more robust and flexible scaffolds for tissue regeneration. Companies are also developing specialized drug delivery microspheres with targeted release mechanisms, improving therapeutic efficacy and minimizing side effects. Competitive advantages are derived from proprietary synthesis methods, advanced material characterization, and successful clinical validation, ensuring market fit and superior patient outcomes.
Key Drivers of Medical Biomedical Materials 3.0 Growth
The growth of the Medical Biomedical Materials 3.0 market is propelled by several interconnected factors. Technological advancements in polymer science and nanotechnology are enabling the development of materials with unprecedented biocompatibility and functionality. The increasing global prevalence of chronic diseases and the aging population are creating sustained demand for advanced medical devices and implants. Supportive government initiatives and funding for biomedical research, coupled with favorable regulatory pathways for innovative biomaterials, further accelerate market expansion. The rising healthcare expenditure worldwide, particularly in emerging economies, also plays a crucial role in driving market penetration.
Challenges in the Medical Biomedical Materials 3.0 Market
Despite its promising growth, the Medical Biomedical Materials 3.0 market faces significant challenges. Stringent and evolving regulatory hurdles necessitate extensive and costly preclinical and clinical trials, potentially delaying market entry for new innovations. Supply chain complexities and the need for specialized manufacturing processes can lead to higher production costs and potential material shortages. Intense competition from both established players and emerging companies, particularly those offering lower-cost alternatives, exerts downward pressure on pricing. Furthermore, the risk of material degradation issues or adverse biological reactions, even with advanced materials, remains a critical concern for patient safety and market acceptance, impacting market share by an estimated billions.
Emerging Opportunities in Medical Biomedical Materials 3.0
Emerging opportunities in the Medical Biomedical Materials 3.0 sector are abundant and driven by groundbreaking scientific discoveries and strategic market expansion. The burgeoning field of regenerative medicine, with its focus on tissue engineering and advanced scaffolds, presents a significant growth catalyst. Technological breakthroughs in nanotechnology are paving the way for "smart" biomaterials capable of real-time monitoring and response within the body. Strategic partnerships between material manufacturers, medical device companies, and research institutions are fostering synergistic development and accelerating product commercialization. Furthermore, the untapped potential of emerging economies, with their rapidly developing healthcare infrastructures and growing patient populations, offers substantial market expansion opportunities for innovative biomedical materials. The increasing demand for personalized medicine solutions also opens new avenues for custom-designed biomaterials.
Leading Players in the Medical Biomedical Materials 3.0 Sector
- Merck
- PCAS
- Fujifilm
- Evonik Health Care
- Nomisma Healthcare
- Corbion
- DURECT
- Mitsui Chemicals
- Phosphorex Inc.
- Lattice Medical
- Jinan Daigang Biomaterial
- Shenzhen Polymtek Biomaterial Co.,Ltd.
- Changchun Foliaplast Bio-tech Co.,Ltd
Key Milestones in Medical Biomedical Materials 3.0 Industry
- 2019: Launch of novel bioresorbable PLGA sutures with enhanced tensile strength, improving surgical outcomes.
- 2020: FDA approval for a new PLGA-based drug delivery system for chronic pain management, expanding market reach.
- 2021: Merck acquires a leading biomaterial startup, strengthening its portfolio in advanced implantable devices.
- 2022: Evonik Health Care expands its production capacity for high-purity PLA and PLGA for pharmaceutical applications.
- 2023: Corbion announces a significant investment in R&D for biodegradable polymers in oral implant applications.
- 2024: Phosphorex Inc. introduces a new generation of drug delivery microspheres with improved encapsulation efficiency.
- 2025: Nomisma Healthcare partners with a leading research institution to develop next-generation fracture fixation materials.
- 2026: Lattice Medical receives CE mark for its innovative scaffold for regenerative medicine applications.
- 2027: DURECT advances clinical trials for its long-acting injectable PLGA-based drug delivery system.
- 2028: Mitsui Chemicals develops a new class of biocompatible polymers for advanced medical device coatings.
- 2029: Jinan Daigang Biomaterial showcases its expanded range of biodegradable films for wound healing applications.
- 2030: Shenzhen Polymtek Biomaterial Co.,Ltd. launches a new series of PLGA microparticles for targeted cancer therapy.
- 2031: Changchun Foliaplast Bio-tech Co.,Ltd. announces significant advancements in the sustainable production of medical-grade PLA.
- 2032: PCAS invests in a new facility dedicated to the synthesis of advanced biomedical polymers.
- 2033: Fujifilm demonstrates a breakthrough in drug delivery microsphere technology with enhanced drug loading capacity.
Strategic Outlook for Medical Biomedical Materials 3.0 Market
The strategic outlook for the Medical Biomedical Materials 3.0 market is exceptionally positive, driven by sustained innovation and increasing global healthcare demands. Growth accelerators include the continued development of personalized medicine, the integration of smart materials with diagnostic and therapeutic capabilities, and the expanding use of additive manufacturing for bespoke implants. Companies are expected to focus on strategic collaborations, mergers, and acquisitions to gain access to new technologies and expand their market reach. The increasing emphasis on sustainability and bioresorbability will further shape product development and market penetration. The projected growth trajectory indicates significant opportunities for stakeholders who can effectively navigate the regulatory landscape, leverage technological advancements, and cater to the evolving needs of patients and healthcare providers. The market is ripe for expansion, with an estimated future market value reaching billions.
Medical Biomedical Materials 3.0 Segmentation
-
1. Application
- 1.1. Suture
- 1.2. Fracture Fixation
- 1.3. Oral Implant
- 1.4. Drug Delivery Microsphere
- 1.5. Others
-
2. Types
- 2.1. PLA
- 2.2. PGA
- 2.3. PLGA
- 2.4. PLCG
- 2.5. Other
Medical Biomedical Materials 3.0 Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
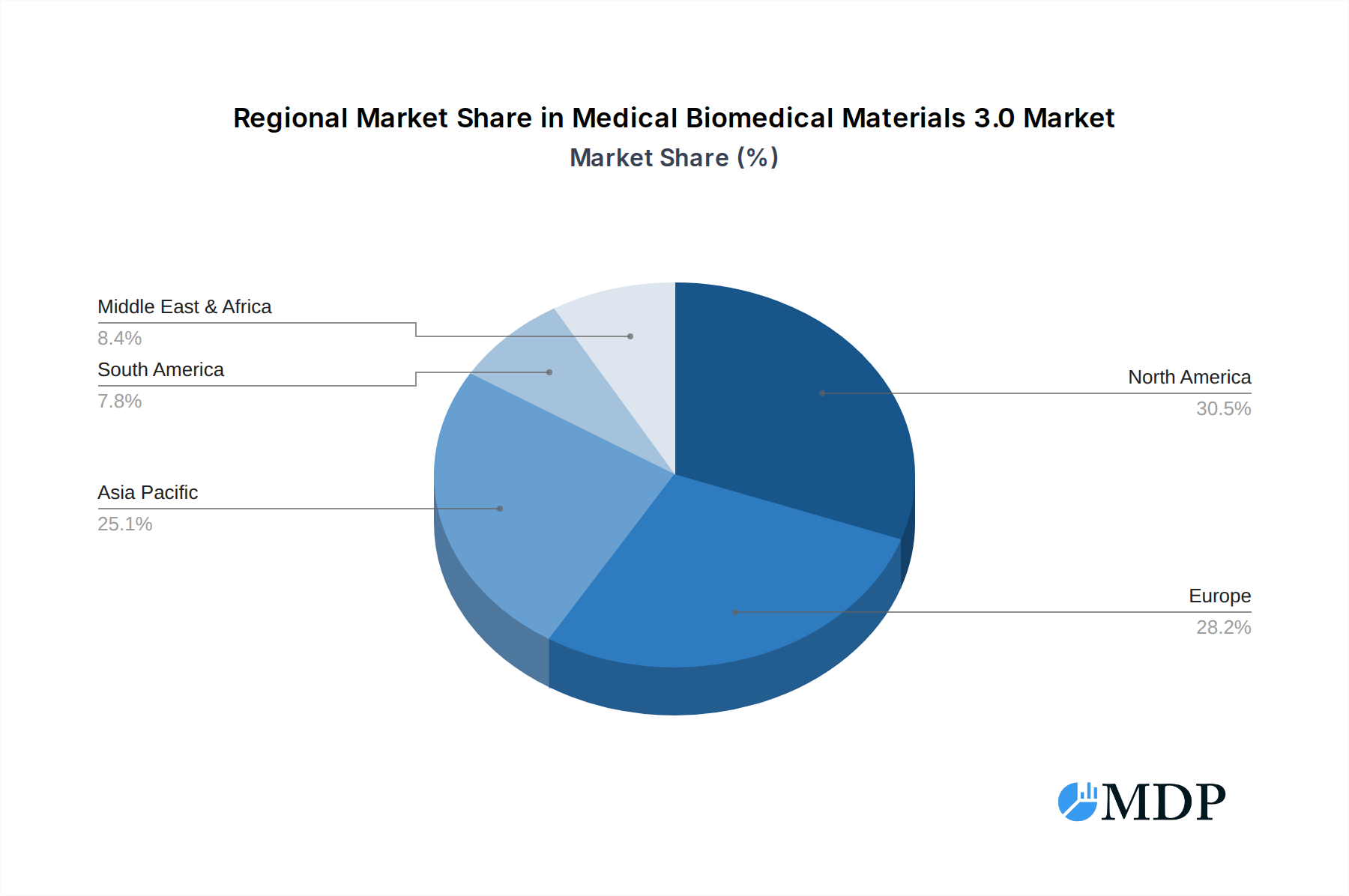
Medical Biomedical Materials 3.0 Regional Market Share

Geographic Coverage of Medical Biomedical Materials 3.0
Medical Biomedical Materials 3.0 REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.6% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Medical Biomedical Materials 3.0 Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Suture
- 5.1.2. Fracture Fixation
- 5.1.3. Oral Implant
- 5.1.4. Drug Delivery Microsphere
- 5.1.5. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. PLA
- 5.2.2. PGA
- 5.2.3. PLGA
- 5.2.4. PLCG
- 5.2.5. Other
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Medical Biomedical Materials 3.0 Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Suture
- 6.1.2. Fracture Fixation
- 6.1.3. Oral Implant
- 6.1.4. Drug Delivery Microsphere
- 6.1.5. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. PLA
- 6.2.2. PGA
- 6.2.3. PLGA
- 6.2.4. PLCG
- 6.2.5. Other
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Medical Biomedical Materials 3.0 Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Suture
- 7.1.2. Fracture Fixation
- 7.1.3. Oral Implant
- 7.1.4. Drug Delivery Microsphere
- 7.1.5. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. PLA
- 7.2.2. PGA
- 7.2.3. PLGA
- 7.2.4. PLCG
- 7.2.5. Other
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Medical Biomedical Materials 3.0 Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Suture
- 8.1.2. Fracture Fixation
- 8.1.3. Oral Implant
- 8.1.4. Drug Delivery Microsphere
- 8.1.5. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. PLA
- 8.2.2. PGA
- 8.2.3. PLGA
- 8.2.4. PLCG
- 8.2.5. Other
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Medical Biomedical Materials 3.0 Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Suture
- 9.1.2. Fracture Fixation
- 9.1.3. Oral Implant
- 9.1.4. Drug Delivery Microsphere
- 9.1.5. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. PLA
- 9.2.2. PGA
- 9.2.3. PLGA
- 9.2.4. PLCG
- 9.2.5. Other
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Medical Biomedical Materials 3.0 Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Suture
- 10.1.2. Fracture Fixation
- 10.1.3. Oral Implant
- 10.1.4. Drug Delivery Microsphere
- 10.1.5. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. PLA
- 10.2.2. PGA
- 10.2.3. PLGA
- 10.2.4. PLCG
- 10.2.5. Other
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Merck
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 PCAS
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Fujifilm
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Evonik Health Care
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Nomisma Healthcare
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Corbion
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 DURECT
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Mitsui Chemicals
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Phosphorex Inc.
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Lattice Medical
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Jinan Daigang Biomaterial
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Shenzhen Polymtek Biomaterial Co.
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Ltd.
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Changchun Foliaplast Bio-tech Co.
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Ltd
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.1 Merck
List of Figures
- Figure 1: Global Medical Biomedical Materials 3.0 Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Medical Biomedical Materials 3.0 Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Medical Biomedical Materials 3.0 Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Medical Biomedical Materials 3.0 Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Medical Biomedical Materials 3.0 Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Medical Biomedical Materials 3.0 Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Medical Biomedical Materials 3.0 Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Medical Biomedical Materials 3.0 Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Medical Biomedical Materials 3.0 Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Medical Biomedical Materials 3.0 Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Medical Biomedical Materials 3.0 Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Medical Biomedical Materials 3.0 Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Medical Biomedical Materials 3.0 Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Medical Biomedical Materials 3.0 Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Medical Biomedical Materials 3.0 Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Medical Biomedical Materials 3.0 Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Medical Biomedical Materials 3.0 Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Medical Biomedical Materials 3.0 Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Medical Biomedical Materials 3.0 Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Medical Biomedical Materials 3.0 Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Medical Biomedical Materials 3.0 Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Medical Biomedical Materials 3.0 Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Medical Biomedical Materials 3.0 Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Medical Biomedical Materials 3.0 Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Medical Biomedical Materials 3.0 Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Medical Biomedical Materials 3.0 Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Medical Biomedical Materials 3.0 Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Medical Biomedical Materials 3.0 Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Medical Biomedical Materials 3.0 Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Medical Biomedical Materials 3.0 Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Medical Biomedical Materials 3.0 Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Medical Biomedical Materials 3.0 Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Medical Biomedical Materials 3.0 Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Medical Biomedical Materials 3.0?
The projected CAGR is approximately 8.6%.
2. Which companies are prominent players in the Medical Biomedical Materials 3.0?
Key companies in the market include Merck, PCAS, Fujifilm, Evonik Health Care, Nomisma Healthcare, Corbion, DURECT, Mitsui Chemicals, Phosphorex Inc., Lattice Medical, Jinan Daigang Biomaterial, Shenzhen Polymtek Biomaterial Co., Ltd., Changchun Foliaplast Bio-tech Co., Ltd.
3. What are the main segments of the Medical Biomedical Materials 3.0?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Medical Biomedical Materials 3.0," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Medical Biomedical Materials 3.0 report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Medical Biomedical Materials 3.0?
To stay informed about further developments, trends, and reports in the Medical Biomedical Materials 3.0, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


