Key Insights
The global RFID in Pharmaceuticals market is poised for significant expansion, projected to reach an estimated USD 6.08 billion in 2025 with a robust Compound Annual Growth Rate (CAGR) of 10.9%. This impressive growth trajectory is primarily fueled by the escalating demand for enhanced drug traceability, counterfeit prevention, and improved supply chain efficiency within the pharmaceutical industry. The increasing regulatory pressure to ensure drug authenticity and patient safety, coupled with the inherent benefits of RFID technology in providing real-time visibility and accurate inventory management, are acting as powerful catalysts for market adoption. Key drivers include the need for cold chain monitoring to maintain the integrity of temperature-sensitive pharmaceuticals, streamlined recall processes, and the optimization of manufacturing operations. The technology’s ability to offer granular data insights further empowers pharmaceutical companies to make informed decisions, reduce operational costs, and enhance overall product lifecycle management.
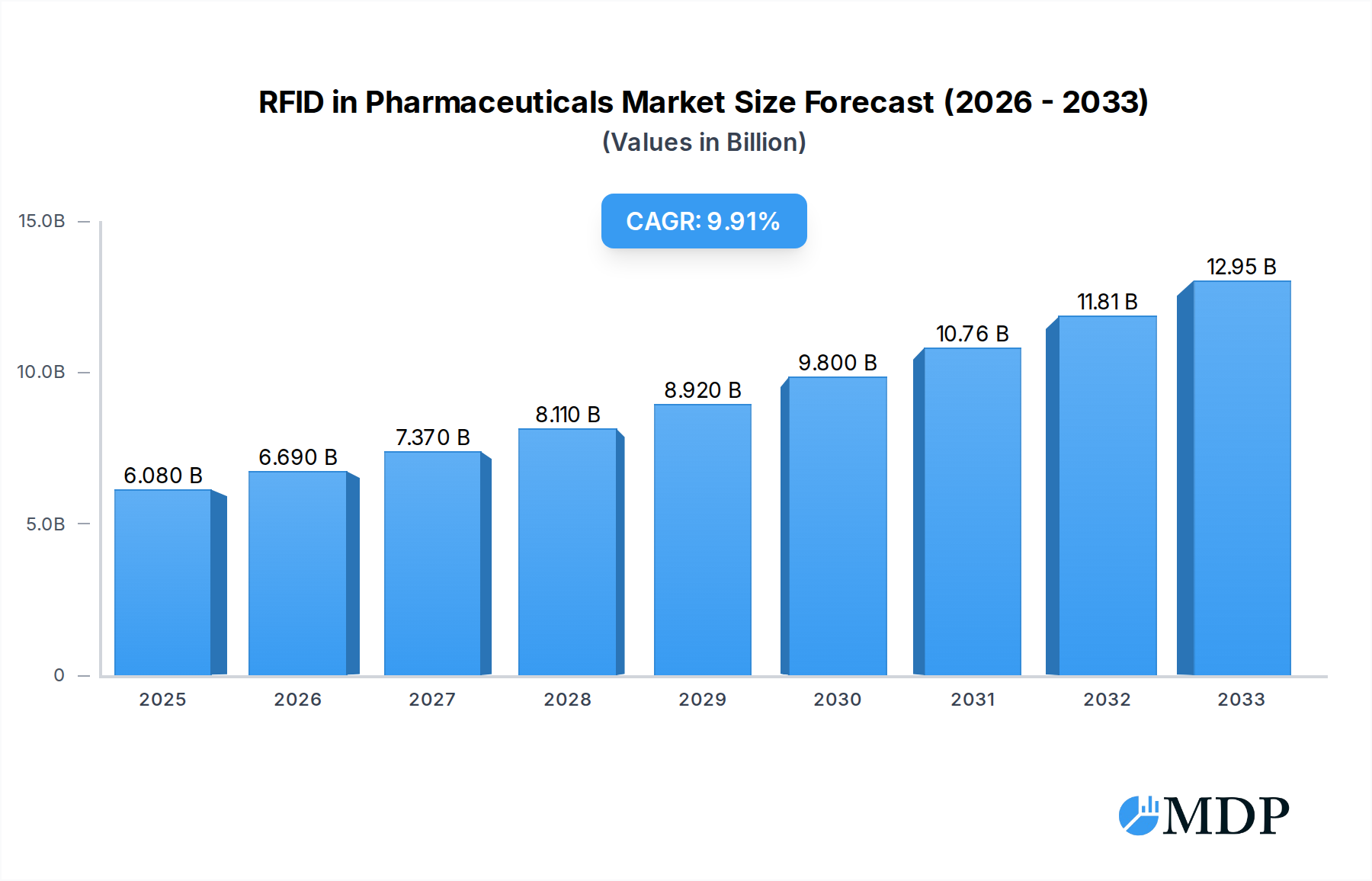
RFID in Pharmaceuticals Market Size (In Billion)

The market is segmented into various applications, with Hospitals & Clinics and Drug Manufacturers representing the largest and fastest-growing segments, respectively. Hospitals and clinics leverage RFID for inventory management, patient identification, and tracking of medical devices, while drug manufacturers are adopting it for supply chain security and serialization compliance. On the technology front, RFID Tags are foundational, but the integration of RFID Readers and sophisticated RFID Middleware is crucial for enabling seamless data capture and analysis, further driving market expansion. Geographically, Asia Pacific is emerging as a key growth region, driven by rapid industrialization, increasing healthcare expenditure, and a growing focus on pharmaceutical supply chain integrity in countries like China and India. North America and Europe continue to be significant markets due to established regulatory frameworks and high adoption rates of advanced technologies in their mature pharmaceutical sectors. Despite the strong growth outlook, challenges such as initial implementation costs and data security concerns may present some restraints, but the overarching benefits of enhanced security, efficiency, and compliance are expected to outweigh these limitations, paving the way for sustained market dominance.
RFID in Pharmaceuticals Company Market Share

Unlocking Pharmaceutical Supply Chain Integrity: RFID Market Report 2024-2033
This comprehensive report delves into the transformative power of Radio Frequency Identification (RFID) technology within the pharmaceutical sector. Explore the evolving market dynamics, key industry trends, and emerging opportunities that are shaping the future of drug traceability, counterfeit prevention, and operational efficiency. With a study period spanning from 2019 to 2033, this in-depth analysis, with a base year of 2025 and a forecast period from 2025 to 2033, provides actionable insights for stakeholders seeking to leverage RFID for enhanced pharmaceutical supply chain management.
RFID in Pharmaceuticals Market Dynamics & Concentration
The RFID in pharmaceuticals market is experiencing robust growth, driven by increasing demands for supply chain transparency and stringent regulatory mandates. Market concentration is moderate, with a few dominant players like Honeywell, Zebra Technologies, and Impinj holding significant market share, estimated at over 30% collectively. However, the landscape is dynamic, with emerging innovators like Invengo Technology and IBM actively pursuing niche applications and technological advancements. Innovation drivers include the perpetual quest to combat counterfeit drugs, a global issue with an estimated annual cost exceeding $200 billion, and the need for real-time inventory management, which can reduce stockouts and wastage by up to 15%. Regulatory frameworks, such as the Drug Supply Chain Security Act (DSCSA) in the US and similar initiatives in Europe and Asia, are acting as powerful catalysts, compelling manufacturers and distributors to adopt track-and-trace solutions. While direct product substitutes for the core functionality of RFID are limited, traditional barcode systems represent an indirect substitute, though they lack the real-time, automated data capture capabilities of RFID. End-user trends show a clear shift towards greater adoption by drug manufacturers and wholesalers, with hospitals and clinics increasingly recognizing the benefits for patient safety and operational efficiency. Mergers and acquisitions (M&A) activities are on the rise, with an estimated count of over 15 significant deals in the historical period (2019-2024), reflecting consolidation and strategic expansion within the market.
RFID in Pharmaceuticals Industry Trends & Analysis
The pharmaceutical RFID market is projected to witness a Compound Annual Growth Rate (CAGR) of approximately 18% during the forecast period (2025-2033), signaling a significant expansion trajectory. This growth is fueled by a confluence of technological advancements, evolving consumer preferences, and intensifying competitive dynamics. The primary market growth driver is the escalating global concern over pharmaceutical counterfeiting, a menace that impacts hundreds of billions of dollars annually and poses severe health risks to patients. RFID technology offers a robust defense mechanism by enabling unique item-level identification and authentication throughout the entire supply chain, from manufacturing to dispensing. Technological disruptions are continuously enhancing RFID capabilities, with advancements in miniaturization of tags, improved read ranges, and the integration of AI and blockchain for enhanced data security and analytics. These innovations are making RFID solutions more cost-effective and versatile, leading to increased market penetration, which is expected to reach over 40% for critical drug products by 2030.
Consumer preferences are also indirectly influencing the adoption of RFID. Patients and healthcare providers are increasingly demanding greater transparency and assurance regarding the authenticity and provenance of medications. This demand translates into a need for sophisticated track-and-trace systems, where RFID plays a pivotal role. Furthermore, the drive for operational efficiency within pharmaceutical companies is a significant motivator. Real-time visibility of inventory, automated data capture for compliance reporting, and optimized recall management can lead to substantial cost savings, estimated to be in the billions annually for major pharmaceutical corporations.
Competitive dynamics are characterized by a blend of established technology giants and agile specialized RFID providers. Companies like Smartrac and Aucxis are focusing on developing high-performance tags and reader solutions tailored for the pharmaceutical environment, while Turck Korea and Datalogic are expanding their product portfolios to address specific needs within the supply chain. The intense competition is spurring further innovation and driving down costs, making RFID technology more accessible to a wider range of pharmaceutical businesses. The increasing emphasis on serialization and aggregation requirements, mandated by regulatory bodies globally, further solidifies RFID's position as an indispensable technology for ensuring drug integrity and patient safety. The overall market penetration is projected to climb steadily, with certain high-value and high-risk drug segments seeing near-universal adoption.
Leading Markets & Segments in RFID in Pharmaceuticals
The RFID in Pharmaceuticals market showcases distinct leadership across various regions and segments, driven by a combination of regulatory mandates, economic factors, and specific application needs. North America, particularly the United States, stands as the dominant market, primarily due to the proactive implementation of the DSCSA and a strong emphasis on patient safety. Economic policies favoring technological adoption and robust healthcare infrastructure contribute significantly to this leadership. The Drug Manufacturer segment represents the largest application within the pharmaceutical RFID landscape. This is a direct consequence of manufacturers being at the forefront of serialization and track-and-trace mandates, requiring them to implement RFID solutions for individual unit identification and supply chain visibility. The estimated market share for RFID solutions within drug manufacturing alone is projected to exceed 50% of the total market by 2027.
In terms of RFID types, RFID Tags constitute the largest segment, accounting for over 60% of the market revenue. This dominance is attributed to the sheer volume of tags required for item-level serialization of pharmaceuticals, from vials and bottles to cartons and pallets. The demand for high-frequency (HF) and ultra-high-frequency (UHF) tags specifically designed for pharmaceutical applications, offering features like tamper-evidence and resistance to environmental factors, is a key driver.
Key Drivers for Drug Manufacturer Dominance:
- Strict regulatory compliance requirements for serialization and track-and-trace.
- Need to combat counterfeit drugs and ensure product authenticity.
- Desire for real-time inventory management and supply chain optimization.
- Facilitation of efficient recalls and returns management.
Key Drivers for RFID Tag Dominance:
- Item-level serialization mandates necessitate a tag for every pharmaceutical unit.
- Advancements in tag technology offering enhanced durability, security, and data storage.
- Cost-effectiveness of tags at high volumes, making them viable for mass drug production.
Hospitals & Clinics: While a smaller segment currently, Hospitals & Clinics are demonstrating significant growth potential. The drivers here include improving patient safety through medication verification, reducing medication errors, and optimizing inventory management to prevent stockouts of critical drugs. Infrastructure investments in healthcare systems are enabling greater adoption of RFID readers and middleware solutions.
Drug Wholesalers: This segment plays a crucial role in the pharmaceutical supply chain and is increasingly adopting RFID for improved inventory accuracy, efficient handling of large volumes, and seamless integration with the serialization data provided by manufacturers. Their adoption is driven by the need to maintain the integrity of drugs as they move through the distribution network.
The dominance of North America, the leading application segment of Drug Manufacturers, and the preeminence of RFID Tags underscore the current landscape. However, the growing adoption in Hospitals & Clinics and the continuous innovation across all RFID types indicate a dynamic and expanding market future.
RFID in Pharmaceuticals Product Developments
Product developments in RFID for pharmaceuticals are characterized by a strong focus on enhanced security, miniaturization, and specialized functionalities. Companies like HID Global and CCL Label are leading the charge in developing tamper-evident and secure RFID tags that are integral to preventing counterfeiting and ensuring product integrity. Innovations include passive UHF tags with advanced encryption capabilities and integrated sensors for temperature and humidity monitoring, crucial for sensitive biologics and vaccines. Xerox Corporation and Fieg Electronics are contributing by developing robust RFID readers and antennas capable of operating in challenging pharmaceutical manufacturing environments, ensuring high read rates and accuracy. The trend towards smaller, more discreet tags that can be embedded directly into packaging or even drug delivery devices is a key technological advancement, enhancing user experience and minimizing impact on product aesthetics. These developments offer competitive advantages by providing pharmaceutical companies with more reliable, secure, and user-friendly solutions for track-and-trace, supply chain visibility, and regulatory compliance.
Key Drivers of RFID in Pharmaceuticals Growth
The growth of the RFID in Pharmaceuticals market is propelled by several critical factors. Firstly, regulatory mandates such as the DSCSA and EU FMD are creating an unavoidable demand for track-and-trace solutions, pushing pharmaceutical companies towards RFID adoption for serialization and authentication. Secondly, the escalating global threat of counterfeit drugs, which results in billions of dollars in losses and poses significant patient safety risks, is a primary motivator for implementing robust anti-counterfeiting technologies like RFID. Thirdly, the pursuit of operational efficiency within the pharmaceutical supply chain, including improved inventory management, reduced stockouts, and streamlined recall processes, offers substantial cost savings, estimated in the billions annually. Finally, technological advancements in RFID, such as miniaturization, enhanced read range, and integration with IoT and blockchain, are making the technology more accessible, reliable, and cost-effective.
Challenges in the RFID in Pharmaceuticals Market
Despite its immense potential, the RFID in Pharmaceuticals market faces several significant challenges that can impede its widespread adoption. High initial investment costs for RFID infrastructure, including readers, tags, and middleware, can be a considerable barrier for smaller pharmaceutical companies and healthcare providers, potentially running into millions for large-scale deployments. Interoperability issues between different RFID systems and existing IT infrastructure can create complexity and hinder seamless data integration, requiring extensive customization. Regulatory hurdles and variations in compliance standards across different regions can also add to the complexity and cost of implementing global RFID solutions. Furthermore, data security and privacy concerns related to the sensitive information captured by RFID tags necessitate robust cybersecurity measures, adding another layer of complexity and expense. Finally, resistance to change within established supply chain processes and a lack of awareness regarding the full benefits of RFID technology can slow down adoption rates.
Emerging Opportunities in RFID in Pharmaceuticals
The RFID in Pharmaceuticals market is ripe with emerging opportunities driven by technological innovation and evolving industry needs. The integration of AI and machine learning with RFID data analytics presents a significant opportunity for predictive insights into supply chain performance, demand forecasting, and proactive identification of potential disruptions, offering potential billions in efficiency gains. The expansion of blockchain technology alongside RFID provides an immutable and transparent ledger for drug provenance, further bolstering anti-counterfeiting efforts and enhancing trust across the supply chain. Strategic partnerships between RFID technology providers and pharmaceutical giants are creating tailor-made solutions, addressing specific pain points and accelerating adoption. Furthermore, the growing focus on cold chain management for vaccines and biologics is creating a substantial market for temperature-monitoring RFID tags, opening up new revenue streams estimated in the hundreds of millions. The increasing demand for personalized medicine also presents an opportunity for unique item-level tracking of patient-specific treatments.
Leading Players in the RFID in Pharmaceuticals Sector
- Honeywell
- Zebra Technologies
- Impinj
- Invengo Technology
- IBM
- Smartrac
- Aucxis
- Turck Korea
- Datalogic
- Unitech
- HID Global
- CCL Label
- Xerox Corporation
- Fieg Electronics
- JADAK
- Texas Instrument
- TSL
- CSL
- GAO RFID
- Alien Technology
- Cipher Lab
- Sense Technology
- Chafon Group
Key Milestones in RFID in Pharmaceuticals Industry
- 2019: Increased global regulatory focus on serialization and track-and-trace mandates, driving early adoption of RFID by major drug manufacturers.
- 2020: Advancements in UHF RFID tag technology offering improved read accuracy and reduced form factors, making them more suitable for pharmaceutical packaging.
- 2021: The COVID-19 pandemic highlighted the critical need for supply chain visibility and security, accelerating interest and investment in RFID solutions for vaccines and essential medicines.
- 2022: Emergence of integrated RFID solutions combining hardware, software, and analytics platforms, simplifying implementation for pharmaceutical companies. Significant M&A activity as larger tech companies acquire specialized RFID firms to expand their pharmaceutical offerings.
- 2023: Growing exploration of blockchain integration with RFID for enhanced data security and provenance tracking, addressing counterfeit concerns more effectively.
- 2024: Increased adoption of RFID for cold chain monitoring of temperature-sensitive drugs, a segment with immense growth potential.
Strategic Outlook for RFID in Pharmaceuticals Market
The strategic outlook for the RFID in Pharmaceuticals market is overwhelmingly positive, driven by the indispensable role of RFID in ensuring drug integrity, patient safety, and supply chain efficiency. Growth accelerators will be primarily fueled by the continuous evolution of regulatory landscapes worldwide, demanding ever-greater levels of traceability and authentication. Technological advancements, particularly in areas like miniaturized, secure, and sensor-integrated RFID tags, coupled with sophisticated analytics platforms, will unlock new use cases and drive down costs, making the technology accessible to a broader spectrum of the pharmaceutical industry. The growing demand for real-time visibility, coupled with the increasing sophistication of anti-counterfeiting strategies, positions RFID as a cornerstone technology for pharmaceutical operations for the foreseeable future. Strategic partnerships and a focus on interoperability will be crucial for maximizing the return on investment and ensuring seamless integration into complex global supply chains.
RFID in Pharmaceuticals Segmentation
-
1. Application
- 1.1. Hospitals & Clinics
- 1.2. Drug Manufacturer
- 1.3. Drug Wholesalers
- 1.4. Others
-
2. Types
- 2.1. RFID Readers
- 2.2. RFID Tags
- 2.3. RFID Middleware
RFID in Pharmaceuticals Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
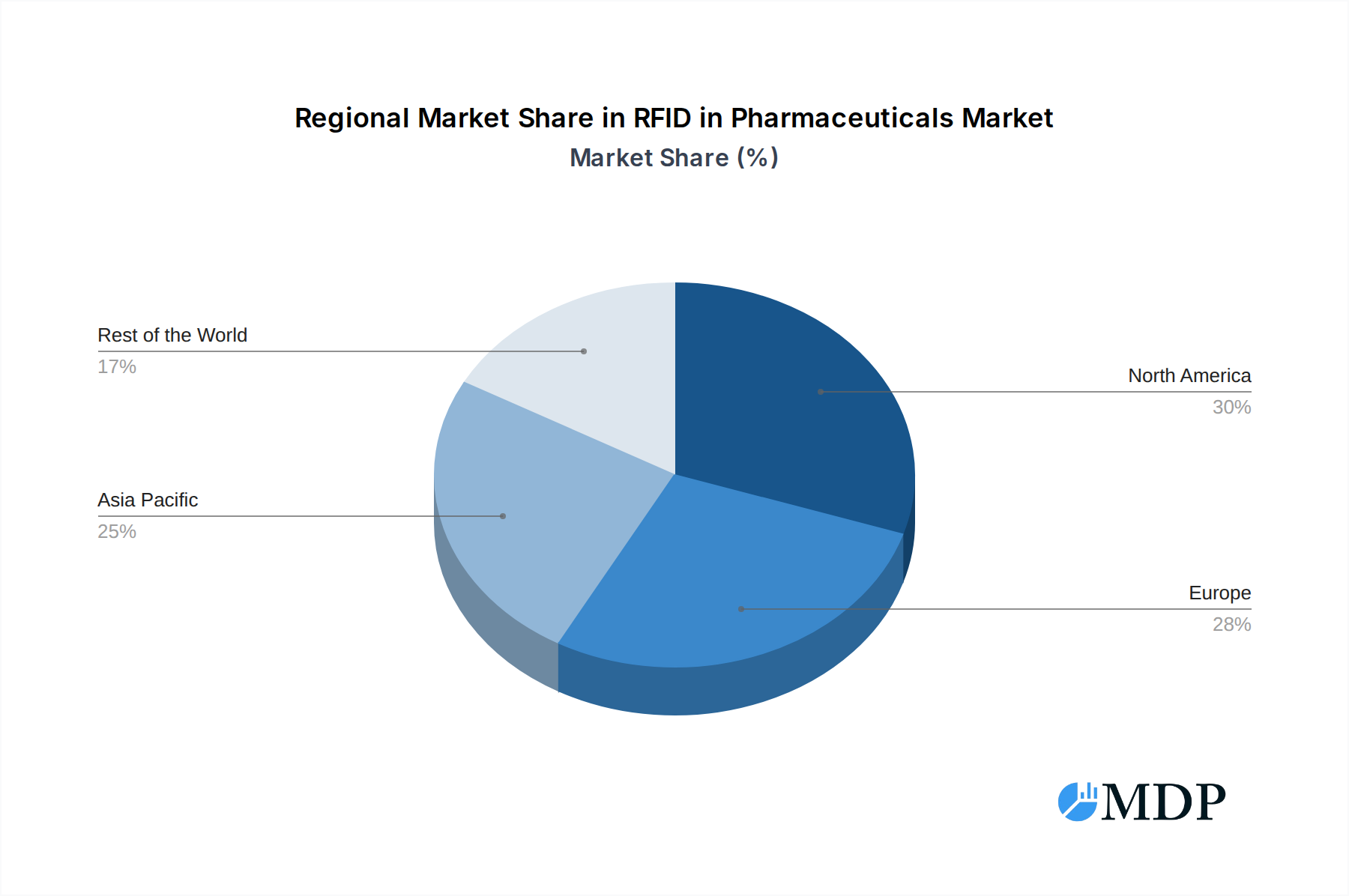
RFID in Pharmaceuticals Regional Market Share

Geographic Coverage of RFID in Pharmaceuticals
RFID in Pharmaceuticals REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 10.9% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global RFID in Pharmaceuticals Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals & Clinics
- 5.1.2. Drug Manufacturer
- 5.1.3. Drug Wholesalers
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. RFID Readers
- 5.2.2. RFID Tags
- 5.2.3. RFID Middleware
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America RFID in Pharmaceuticals Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospitals & Clinics
- 6.1.2. Drug Manufacturer
- 6.1.3. Drug Wholesalers
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. RFID Readers
- 6.2.2. RFID Tags
- 6.2.3. RFID Middleware
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America RFID in Pharmaceuticals Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospitals & Clinics
- 7.1.2. Drug Manufacturer
- 7.1.3. Drug Wholesalers
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. RFID Readers
- 7.2.2. RFID Tags
- 7.2.3. RFID Middleware
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe RFID in Pharmaceuticals Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospitals & Clinics
- 8.1.2. Drug Manufacturer
- 8.1.3. Drug Wholesalers
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. RFID Readers
- 8.2.2. RFID Tags
- 8.2.3. RFID Middleware
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa RFID in Pharmaceuticals Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospitals & Clinics
- 9.1.2. Drug Manufacturer
- 9.1.3. Drug Wholesalers
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. RFID Readers
- 9.2.2. RFID Tags
- 9.2.3. RFID Middleware
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific RFID in Pharmaceuticals Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospitals & Clinics
- 10.1.2. Drug Manufacturer
- 10.1.3. Drug Wholesalers
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. RFID Readers
- 10.2.2. RFID Tags
- 10.2.3. RFID Middleware
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Honeywell
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Zebra Technologies
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Impinj
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Invengo Technology
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 IBM
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Smartrac
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Aucxis
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Turck Korea
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Datalogic
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Unitech
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 HID Global
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 CCL Label
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Xerox Corporation
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Fieg Electronics
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 JADAK
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Texas Instrument
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 TSL
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.18 CSL
- 11.2.18.1. Overview
- 11.2.18.2. Products
- 11.2.18.3. SWOT Analysis
- 11.2.18.4. Recent Developments
- 11.2.18.5. Financials (Based on Availability)
- 11.2.19 GAO RFID
- 11.2.19.1. Overview
- 11.2.19.2. Products
- 11.2.19.3. SWOT Analysis
- 11.2.19.4. Recent Developments
- 11.2.19.5. Financials (Based on Availability)
- 11.2.20 Alien Technology
- 11.2.20.1. Overview
- 11.2.20.2. Products
- 11.2.20.3. SWOT Analysis
- 11.2.20.4. Recent Developments
- 11.2.20.5. Financials (Based on Availability)
- 11.2.21 Cipher Lab
- 11.2.21.1. Overview
- 11.2.21.2. Products
- 11.2.21.3. SWOT Analysis
- 11.2.21.4. Recent Developments
- 11.2.21.5. Financials (Based on Availability)
- 11.2.22 Sense Technology
- 11.2.22.1. Overview
- 11.2.22.2. Products
- 11.2.22.3. SWOT Analysis
- 11.2.22.4. Recent Developments
- 11.2.22.5. Financials (Based on Availability)
- 11.2.23 Chafon Group
- 11.2.23.1. Overview
- 11.2.23.2. Products
- 11.2.23.3. SWOT Analysis
- 11.2.23.4. Recent Developments
- 11.2.23.5. Financials (Based on Availability)
- 11.2.1 Honeywell
List of Figures
- Figure 1: Global RFID in Pharmaceuticals Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America RFID in Pharmaceuticals Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America RFID in Pharmaceuticals Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America RFID in Pharmaceuticals Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America RFID in Pharmaceuticals Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America RFID in Pharmaceuticals Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America RFID in Pharmaceuticals Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America RFID in Pharmaceuticals Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America RFID in Pharmaceuticals Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America RFID in Pharmaceuticals Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America RFID in Pharmaceuticals Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America RFID in Pharmaceuticals Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America RFID in Pharmaceuticals Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe RFID in Pharmaceuticals Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe RFID in Pharmaceuticals Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe RFID in Pharmaceuticals Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe RFID in Pharmaceuticals Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe RFID in Pharmaceuticals Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe RFID in Pharmaceuticals Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa RFID in Pharmaceuticals Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa RFID in Pharmaceuticals Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa RFID in Pharmaceuticals Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa RFID in Pharmaceuticals Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa RFID in Pharmaceuticals Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa RFID in Pharmaceuticals Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific RFID in Pharmaceuticals Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific RFID in Pharmaceuticals Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific RFID in Pharmaceuticals Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific RFID in Pharmaceuticals Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific RFID in Pharmaceuticals Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific RFID in Pharmaceuticals Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global RFID in Pharmaceuticals Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific RFID in Pharmaceuticals Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the RFID in Pharmaceuticals?
The projected CAGR is approximately 10.9%.
2. Which companies are prominent players in the RFID in Pharmaceuticals?
Key companies in the market include Honeywell, Zebra Technologies, Impinj, Invengo Technology, IBM, Smartrac, Aucxis, Turck Korea, Datalogic, Unitech, HID Global, CCL Label, Xerox Corporation, Fieg Electronics, JADAK, Texas Instrument, TSL, CSL, GAO RFID, Alien Technology, Cipher Lab, Sense Technology, Chafon Group.
3. What are the main segments of the RFID in Pharmaceuticals?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "RFID in Pharmaceuticals," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the RFID in Pharmaceuticals report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the RFID in Pharmaceuticals?
To stay informed about further developments, trends, and reports in the RFID in Pharmaceuticals, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


