Key Insights
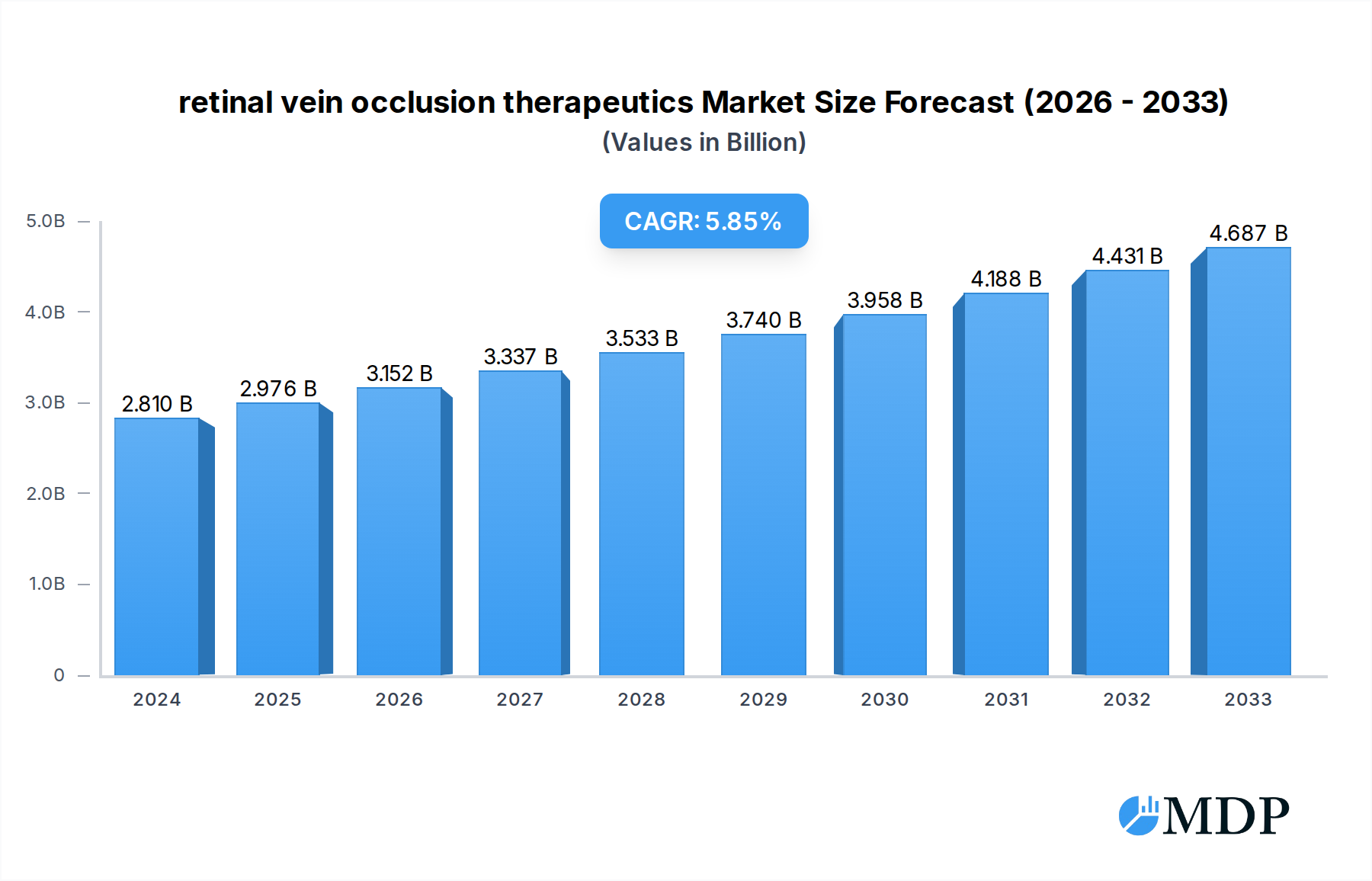
The global retinal vein occlusion (RVO) therapeutics market is poised for significant expansion, driven by an increasing prevalence of risk factors such as diabetes, hypertension, and an aging population. These conditions directly contribute to the incidence of both central retinal vein occlusion (CRVO) and branch retinal vein occlusion (BRVO), creating a growing patient pool necessitating effective treatment options. The market is currently valued at approximately $2,809.7 million in 2024, demonstrating a robust demand for advanced therapies. The projected compound annual growth rate (CAGR) of 5.81% from 2025 to 2033 underscores the sustained upward trajectory of this market. Key drivers include advancements in therapeutic modalities, such as anti-VEGF injections and intravitreal steroid implants, which offer improved efficacy and patient outcomes. Furthermore, increasing healthcare expenditure and rising awareness among patients and healthcare professionals about RVO and its management are contributing factors to market growth.
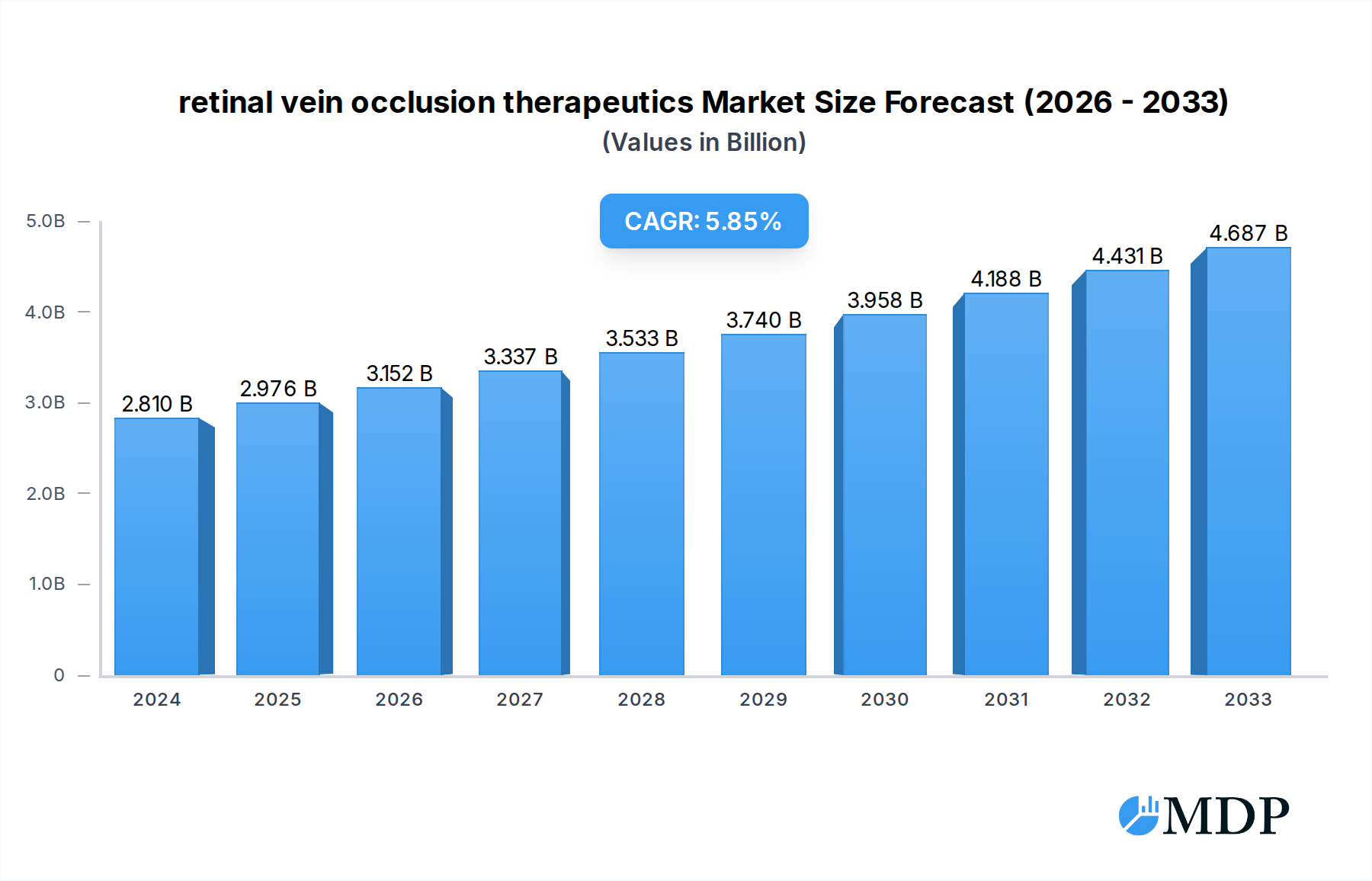
retinal vein occlusion therapeutics Market Size (In Billion)

The therapeutic landscape for RVO is characterized by a dynamic interplay between established treatments and emerging innovations. While anti-VEGF agents and corticosteroids remain cornerstones of treatment, ongoing research and development are exploring novel drug targets and delivery systems to enhance therapeutic benefits and address unmet needs, particularly in managing vision loss associated with RVO. The market is segmented by application into hospitals, ambulatory surgical centers, and others, with hospitals and ASCs representing the predominant segments due to the need for specialized medical procedures. Geographically, North America and Europe are expected to maintain significant market shares, driven by high healthcare spending, advanced research infrastructure, and established reimbursement policies. However, the Asia-Pacific region is anticipated to witness the fastest growth, fueled by a rising RVO incidence, improving healthcare access, and increasing investments in the pharmaceutical sector.
retinal vein occlusion therapeutics Company Market Share

Retinal Vein Occlusion Therapeutics Market: Comprehensive Analysis and Future Outlook (2019-2033)
This in-depth report provides a detailed analysis of the global Retinal Vein Occlusion (RVO) therapeutics market, a critical segment within ophthalmology. Covering the historical period of 2019–2024, the base and estimated year of 2025, and a comprehensive forecast period extending to 2033, this report offers actionable insights for pharmaceutical companies, healthcare providers, investors, and regulatory bodies. We delve into market dynamics, key trends, leading segments, product developments, growth drivers, challenges, and emerging opportunities, along with an exhaustive overview of leading players and their strategic milestones. This report is essential for stakeholders seeking to understand and capitalize on the evolving RVO therapeutics landscape, which is projected to reach multi-million dollar valuations.
Retinal Vein Occlusion Therapeutics Market Dynamics & Concentration
The Retinal Vein Occlusion (RVO) therapeutics market is characterized by a moderate concentration, with key players like Sanofi Aventis, Bausch Health, and Allergan holding significant market shares. Innovation drivers are primarily focused on developing novel anti-vascular endothelial growth factor (anti-VEGF) therapies, sustained-release drug delivery systems, and gene therapies to improve treatment efficacy and patient compliance. The regulatory framework is stringent, with agencies like the FDA and EMA meticulously evaluating new drug applications based on safety and efficacy data. Product substitutes, while limited for severe RVO, include laser photocoagulation and vitrectomy for specific cases. End-user trends indicate a growing preference for less invasive treatments and therapies that offer long-term vision preservation. Mergers and acquisitions (M&A) activity, while not pervasive, has seen strategic collaborations and smaller acquisitions aimed at acquiring promising pipeline assets or expanding geographical reach. The M&A deal count has remained steady in recent years, reflecting a cautious yet strategic approach to market consolidation. Market share distribution is influenced by patent expirations of established drugs and the successful introduction of innovative therapies.
Retinal Vein Occlusion Therapeutics Industry Trends & Analysis
The global Retinal Vein Occlusion (RVO) therapeutics market is poised for significant expansion, driven by an increasing prevalence of RVO, rising healthcare expenditures, and advancements in therapeutic modalities. The compound annual growth rate (CAGR) is expected to be robust, fueled by a growing aging population, a significant risk factor for vascular diseases affecting the retina. Technological disruptions are revolutionizing RVO treatment. The introduction of biosimil versions of established anti-VEGF drugs is expected to increase market penetration by offering more affordable treatment options. Furthermore, ongoing research into novel drug targets beyond VEGF, such as angiopoietin-2 inhibitors and gene therapies, promises to offer new avenues for treating RVO and its associated complications. Consumer preferences are shifting towards treatments that minimize the frequency of intravitreal injections and offer improved visual outcomes with fewer side effects. This trend is stimulating the development of sustained-release implants and more targeted therapeutic approaches. Competitive dynamics within the RVO therapeutics market are intense, with pharmaceutical giants and emerging biotech firms vying for market dominance. The pipeline for RVO therapeutics is dynamic, featuring a range of innovative molecules and delivery systems that aim to address unmet clinical needs. Market penetration is expected to increase as awareness about RVO and its treatment options grows among both patients and healthcare professionals. The economic policies supporting healthcare infrastructure development and reimbursement for advanced RVO therapies will play a crucial role in shaping market growth.
Leading Markets & Segments in Retinal Vein Occlusion Therapeutics
The Retinal Vein Occlusion (RVO) therapeutics market exhibits distinct regional and segmental dominance. Geographically, North America currently leads the market, driven by high healthcare spending, advanced medical infrastructure, and a strong emphasis on research and development. Within North America, the United States represents the largest single market due to its substantial patient population and a well-established reimbursement framework for advanced ophthalmic treatments. Asia Pacific is emerging as a rapidly growing region, fueled by increasing RVO prevalence, expanding healthcare access, and rising disposable incomes.
Application Segments:
- Hospitals: Hospitals remain the primary treatment setting for RVO, particularly for managing complex cases and delivering intravitreal injections. The presence of specialized ophthalmology departments and trained medical professionals makes hospitals the go-to destination for acute RVO management. Economic policies supporting hospital infrastructure and the availability of advanced diagnostic and treatment equipment further solidify this segment's dominance.
- Ambulatory Surgical Centers (ASCs): ASCs are increasingly gaining traction for RVO treatment, especially for routine intravitreal injections and follow-up care. Their cost-effectiveness and patient convenience make them an attractive alternative to hospitals for less complex cases. The growing trend of outpatient procedures and favorable reimbursement for ASCs are key drivers for this segment.
- Others: This category encompasses specialized eye clinics and private practices. While smaller in overall market share compared to hospitals, these settings play a crucial role in providing specialized care and managing chronic RVO patients, offering personalized treatment plans and convenient access to care.
Types of RVO:
- Central Retinal Vein Occlusion (CRVO): CRVO, being a more severe form of RVO, generally commands a larger share of the market for therapeutics. The potential for significant vision loss necessitates aggressive and often continuous treatment, leading to higher drug utilization and expenditure. Factors like an aging population and the prevalence of cardiovascular comorbidities contribute to the higher incidence and severity of CRVO.
- Branch Retinal Vein Occlusion (BRVO): BRVO, while more common, is often considered a less severe condition. However, its widespread occurrence still contributes significantly to the overall market for RVO therapeutics. Early detection and intervention in BRVO cases are crucial to prevent complications and preserve vision, driving demand for effective treatment options in this segment.
Retinal Vein Occlusion Therapeutics Product Developments
Product innovation in RVO therapeutics centers on enhancing efficacy, duration of action, and patient convenience. Leading pharmaceutical companies are actively developing next-generation anti-VEGF agents with improved pharmacokinetics and reduced systemic exposure. Furthermore, advancements in drug delivery systems, such as sustained-release implants, are gaining prominence, offering longer intervals between treatments and potentially improving patient adherence. Gene therapy approaches are also emerging as a promising frontier, aiming to provide a more durable and potentially curative treatment for RVO. These developments are driven by a need to address the limitations of current therapies, such as the frequent need for intravitreal injections and the variability in patient response.
Key Drivers of Retinal Vein Occlusion Therapeutics Growth
The growth of the Retinal Vein Occlusion (RVO) therapeutics market is propelled by several key factors. An increasing global prevalence of RVO, largely attributed to an aging population and the rising incidence of conditions like hypertension and diabetes, is a primary driver. Advancements in diagnostic technologies, enabling earlier and more accurate detection of RVO, contribute to a larger patient pool seeking treatment. Moreover, the continuous development of novel and more effective therapeutic agents, including sustained-release formulations and gene therapies, is expanding treatment options and improving patient outcomes. Favorable reimbursement policies for advanced RVO treatments in developed economies and the expanding healthcare infrastructure in emerging markets further accelerate market growth.
Challenges in the Retinal Vein Occlusion Therapeutics Market
Despite the promising growth trajectory, the Retinal Vein Occlusion (RVO) therapeutics market faces several challenges. The high cost of novel RVO therapies, particularly biologics and gene therapies, can limit accessibility, especially in resource-constrained regions. Stringent regulatory approval processes, requiring extensive clinical trials and post-market surveillance, can lead to lengthy development timelines and significant investment. Furthermore, the potential for adverse events associated with intravitreal injections, such as increased intraocular pressure and endophthalmitis, necessitates careful patient monitoring. Competitive pressures from existing treatments and the development of alternative therapies also pose a challenge to market expansion.
Emerging Opportunities in Retinal Vein Occlusion Therapeutics
Emerging opportunities in the Retinal Vein Occlusion (RVO) therapeutics market are primarily driven by technological breakthroughs and unmet clinical needs. The development of targeted therapies that address specific molecular pathways involved in RVO pathogenesis, beyond VEGF, presents a significant opportunity. Furthermore, advancements in artificial intelligence (AI) and machine learning for early RVO detection and personalized treatment selection hold immense potential. Strategic partnerships between pharmaceutical companies, academic institutions, and device manufacturers can accelerate the development and commercialization of innovative RVO treatments. Expanding access to affordable RVO therapies in emerging markets and focusing on patient education to improve early diagnosis and adherence also represent substantial growth avenues.
Leading Players in the Retinal Vein Occlusion Therapeutics Sector
- Sanofi Aventis
- Bausch Health
- Allergan
Key Milestones in Retinal Vein Occlusion Therapeutics Industry
- 2019: Approval of new sustained-release implants for RVO treatment.
- 2020: Significant clinical trial results showcasing improved efficacy of novel anti-VEGF agents.
- 2021: Merger of key pharmaceutical entities to strengthen RVO therapeutic portfolios.
- 2022: Introduction of biosimilar RVO therapeutics, increasing market accessibility.
- 2023: Positive early-stage clinical data for gene therapy candidates targeting RVO.
- 2024: Expansion of RVO treatment guidelines to include emerging therapeutic modalities.
Strategic Outlook for Retinal Vein Occlusion Therapeutics Market
The strategic outlook for the Retinal Vein Occlusion (RVO) therapeutics market remains exceptionally positive. The sustained demand driven by demographic shifts and increasing disease awareness, coupled with ongoing innovation, will fuel market expansion. Pharmaceutical companies are expected to focus on developing combination therapies to address complex RVO cases and exploring novel delivery mechanisms to enhance patient compliance. Strategic collaborations and acquisitions will continue to play a vital role in consolidating market positions and accelerating pipeline development. The growing emphasis on personalized medicine and the integration of digital health tools for patient monitoring and management will further shape the future landscape of RVO therapeutics.
retinal vein occlusion therapeutics Segmentation
-
1. Application
- 1.1. Hospitals
- 1.2. Ambulatory Surgical Centers
- 1.3. Others
-
2. Types
- 2.1. Branch Retinal Vein Occlusion
- 2.2. Central Retinal Vein Occlusion
retinal vein occlusion therapeutics Segmentation By Geography
- 1. CA
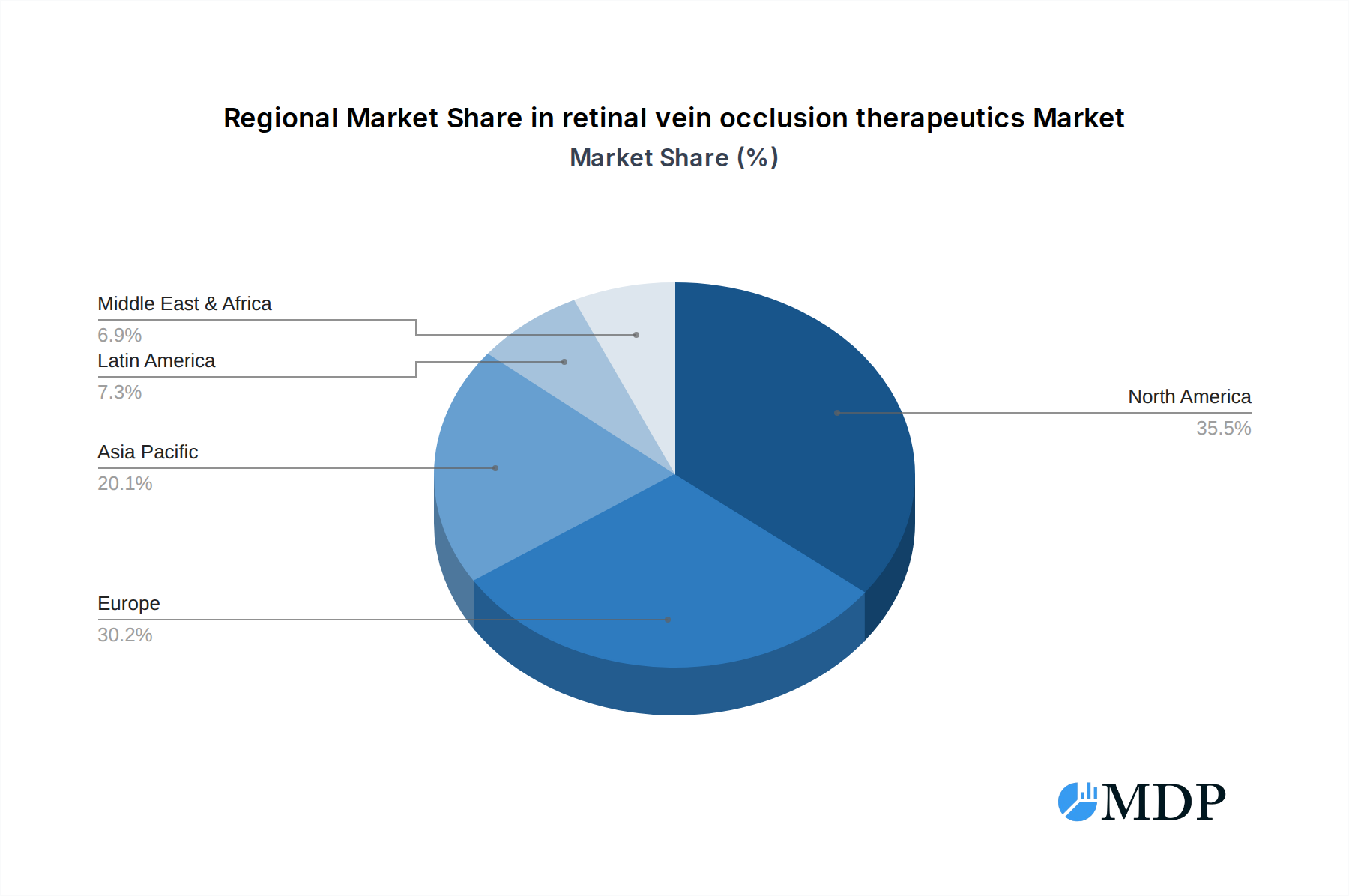
retinal vein occlusion therapeutics Regional Market Share

Geographic Coverage of retinal vein occlusion therapeutics
retinal vein occlusion therapeutics REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.81% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. retinal vein occlusion therapeutics Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals
- 5.1.2. Ambulatory Surgical Centers
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Branch Retinal Vein Occlusion
- 5.2.2. Central Retinal Vein Occlusion
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. CA
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Competitive Analysis
- 6.1. Market Share Analysis 2025
- 6.2. Company Profiles
- 6.2.1 Sanofi Aventis
- 6.2.1.1. Overview
- 6.2.1.2. Products
- 6.2.1.3. SWOT Analysis
- 6.2.1.4. Recent Developments
- 6.2.1.5. Financials (Based on Availability)
- 6.2.2 Bausch Health
- 6.2.2.1. Overview
- 6.2.2.2. Products
- 6.2.2.3. SWOT Analysis
- 6.2.2.4. Recent Developments
- 6.2.2.5. Financials (Based on Availability)
- 6.2.3 Allergan
- 6.2.3.1. Overview
- 6.2.3.2. Products
- 6.2.3.3. SWOT Analysis
- 6.2.3.4. Recent Developments
- 6.2.3.5. Financials (Based on Availability)
- 6.2.1 Sanofi Aventis
List of Figures
- Figure 1: retinal vein occlusion therapeutics Revenue Breakdown (undefined, %) by Product 2025 & 2033
- Figure 2: retinal vein occlusion therapeutics Share (%) by Company 2025
List of Tables
- Table 1: retinal vein occlusion therapeutics Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: retinal vein occlusion therapeutics Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: retinal vein occlusion therapeutics Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: retinal vein occlusion therapeutics Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: retinal vein occlusion therapeutics Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: retinal vein occlusion therapeutics Revenue undefined Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the retinal vein occlusion therapeutics?
The projected CAGR is approximately 5.81%.
2. Which companies are prominent players in the retinal vein occlusion therapeutics?
Key companies in the market include Sanofi Aventis, Bausch Health, Allergan.
3. What are the main segments of the retinal vein occlusion therapeutics?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3400.00, USD 5100.00, and USD 6800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "retinal vein occlusion therapeutics," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the retinal vein occlusion therapeutics report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the retinal vein occlusion therapeutics?
To stay informed about further developments, trends, and reports in the retinal vein occlusion therapeutics, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


