Key Insights
The global pharmaceutical track and trace system market is poised for substantial growth, estimated at USD 8.33 billion in 2025. This expansion is driven by an impressive Compound Annual Growth Rate (CAGR) of 18.95% anticipated between 2025 and 2033. The increasing imperative for regulatory compliance, particularly concerning drug counterfeiting and diversion, stands as a primary catalyst for this market's upward trajectory. Governments worldwide are mandating serialization and aggregation solutions to ensure the integrity of the pharmaceutical supply chain, pushing pharmaceutical companies to adopt sophisticated track and trace technologies. Furthermore, the growing complexity of global pharmaceutical distribution networks and the rising demand for patient safety are significant drivers, encouraging investments in systems that provide real-time visibility and accountability from manufacturing to the end consumer. The market's expansion is also fueled by advancements in technology, such as AI and blockchain integration, which are enhancing the accuracy, efficiency, and security of track and trace solutions.
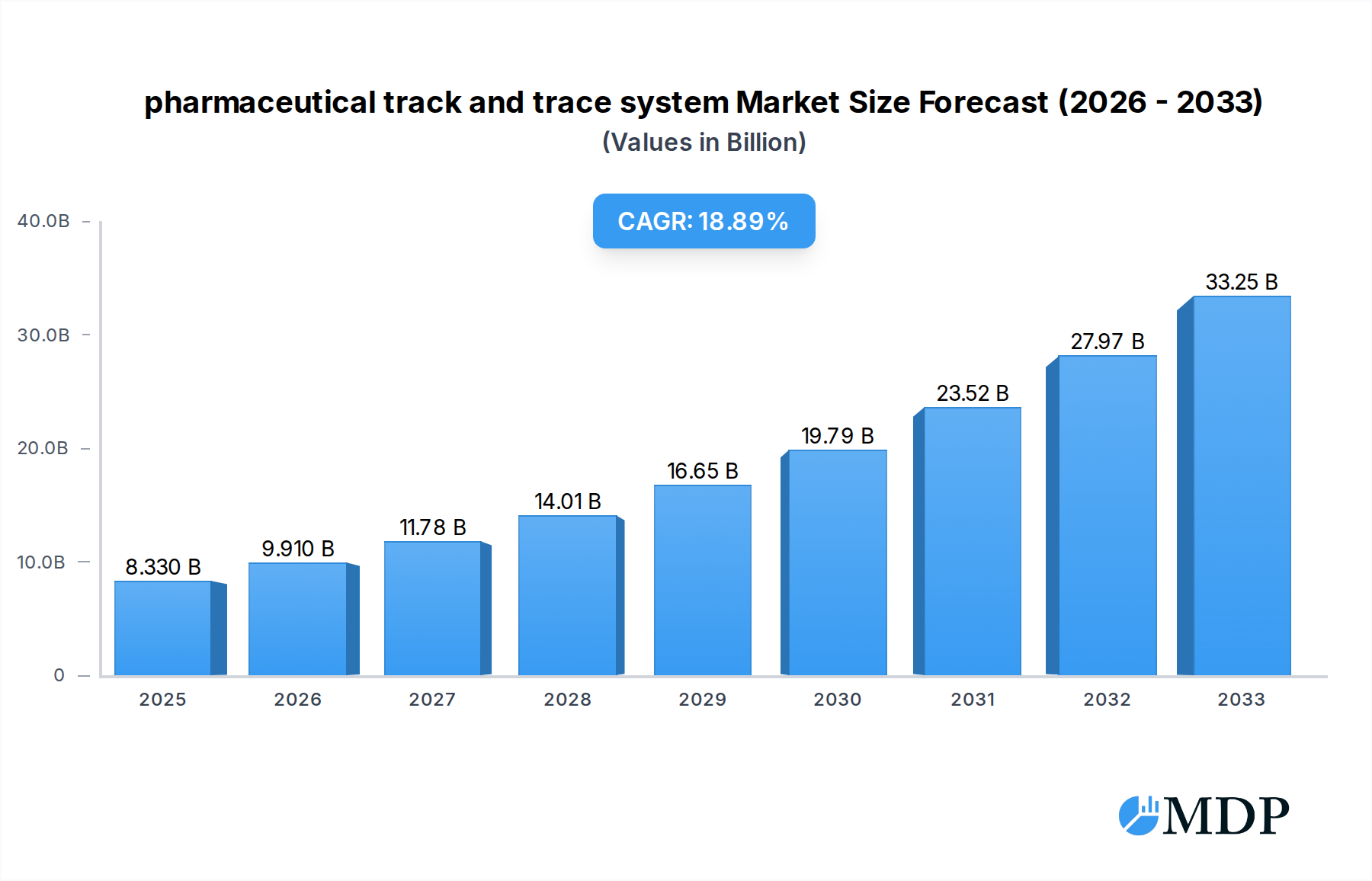
pharmaceutical track and trace system Market Size (In Billion)

The market is segmented across various applications, including hospital pharmacies, retail pharmacies, and online pharmacies, reflecting the widespread need for product traceability throughout the distribution chain. Technologically, the market is characterized by the dominance of Barcodes Pharmaceutical Track and Trace Systems, alongside the growing adoption of Real-time Locating System (RTLS) and Radio-frequency Identification (RFID) Tags Pharmaceutical Track and Trace Systems, which offer enhanced data capture and real-time monitoring capabilities. Leading companies like Marchesini Group, Weber Marking Systems, Pester Pac Automation, Siemens, Bosch, Körber, and Uhlmann Packaging System are actively investing in research and development to offer comprehensive solutions that meet evolving regulatory demands and industry best practices. The robust CAGR suggests that the market will continue to expand significantly, offering substantial opportunities for innovation and market penetration in the coming years.
pharmaceutical track and trace system Company Market Share

This comprehensive report delves into the dynamic pharmaceutical track and trace system market, providing in-depth analysis and actionable insights for industry stakeholders. Covering a study period from 2019 to 2033, with a base and estimated year of 2025, this report analyzes market dynamics, key trends, leading segments, product developments, growth drivers, challenges, emerging opportunities, and strategic outlook. We meticulously examine the competitive landscape featuring major players like Marchesini Group, Weber Marking Systems, Pester Pac Automation, Siemens, Bosch, Körber, and Uhlmann Packaging System. The report also explores critical applications within Hospital Pharmacy, Retail Pharmacy, and Online Pharmacy, alongside prevalent types such as Barcodes Pharmaceutical Track And Trace System, Real-time Locating System (RTLS) Pharmaceutical Track And Trace System, and Radio-frequency Identification (RFID) Tags Pharmaceutical Track And Trace System. With billions in projected market value and significant CAGR, this research is indispensable for navigating the evolving pharmaceutical supply chain.
pharmaceutical track and trace system Market Dynamics & Concentration
The pharmaceutical track and trace system market is characterized by a moderate to high concentration, with key players investing heavily in technological advancements and strategic acquisitions to bolster their market share, which is projected to reach over XX billion by 2025. Regulatory frameworks worldwide, such as the U.S. Drug Supply Chain Security Act (DSCSA) and the EU Falsified Medicines Directive (FMD), are the primary innovation drivers, mandating serialization and traceability for drug products to combat counterfeiting and ensure patient safety. Product substitutes are limited due to the stringent regulatory requirements and the inherent need for secure supply chain visibility. End-user trends highlight a growing demand for real-time data, enhanced supply chain efficiency, and robust recall management capabilities. Mergers and acquisitions (M&A) activities are a notable aspect, with an estimated XX number of deals occurring annually, aimed at consolidating market positions and expanding technological portfolios. For instance, the acquisition of smaller technology providers by larger conglomerates is a recurring theme, contributing to market consolidation.
pharmaceutical track and trace system Industry Trends & Analysis
The pharmaceutical track and trace system industry is experiencing robust growth, driven by a confluence of factors that are reshaping the global pharmaceutical supply chain. The escalating threat of counterfeit drugs, which has resulted in billions of dollars in economic losses and significant patient health risks, is a primary catalyst for the widespread adoption of sophisticated track and trace solutions. Governments and regulatory bodies worldwide are implementing increasingly stringent regulations, mandating serialization, aggregation, and real-time reporting of pharmaceutical products from manufacturing to the point of dispensing. This regulatory push, coupled with the inherent need for enhanced supply chain visibility and integrity, is propelling market expansion. The Compound Annual Growth Rate (CAGR) for this sector is projected to be a substantial XX% over the forecast period of 2025–2033, indicating a rapidly growing market.
Technological disruptions are playing a pivotal role in this evolution. Advancements in barcode technology, such as 2D data matrix codes, coupled with the increasing adoption of Radio-frequency Identification (RFID) tags and Real-time Locating Systems (RTLS), are offering more granular and accurate tracking capabilities. These technologies enable pharmaceutical companies to monitor the movement and condition of their products throughout the entire supply chain, from manufacturing facilities to the hands of patients. The integration of IoT devices and blockchain technology further enhances data security, transparency, and immutability, providing an unparalleled level of trust and traceability.
Consumer preferences are also subtly influencing the market. Patients and healthcare providers are increasingly demanding assurance of drug authenticity and safety, a sentiment amplified by high-profile recalls and concerns about drug diversion. This awareness is driving demand for systems that can provide verifiable proof of a product's journey and origin. The market penetration of track and trace solutions is steadily increasing across all segments of the pharmaceutical industry, including large-scale manufacturers, distributors, and even smaller retail pharmacies, as the benefits of improved inventory management, reduced losses, and enhanced compliance become more apparent. The competitive dynamics within the industry are intensifying, with established players and emerging innovators vying for market share through product differentiation, strategic partnerships, and robust service offerings.
Leading Markets & Segments in pharmaceutical track and trace system
The global pharmaceutical track and trace system market exhibits distinct regional and segment-specific dominance, driven by a combination of regulatory imperatives, infrastructure development, and market maturity.
Dominant Region: North America currently leads the market, primarily due to the early and stringent implementation of regulations like the DSCSA. The United States, with its vast pharmaceutical manufacturing and distribution network, presents a substantial market for track and trace solutions.
- Key Drivers in North America:
- Proactive regulatory enforcement and a strong focus on patient safety.
- Significant investment in pharmaceutical research and development.
- Well-established healthcare infrastructure with a high adoption rate of advanced technologies.
Dominant Country: The United States stands out as the leading country, owing to its comprehensive serialization mandates and a highly developed pharmaceutical ecosystem.
Dominant Application Segments:
- Hospital Pharmacy: This segment is a major contributor to market growth. Hospitals are increasingly implementing track and trace systems to manage inventory, prevent medication errors, and ensure the authenticity of drugs dispensed to inpatients.
- Key Drivers in Hospital Pharmacy:
- Need for precise medication administration and reduced risk of diversion.
- Compliance with evolving hospital accreditation standards.
- Integration with Electronic Health Records (EHR) for seamless data flow.
- Key Drivers in Hospital Pharmacy:
- Retail Pharmacy: The retail pharmacy segment is also a significant area of growth, driven by regulatory requirements for dispensing serialized prescription drugs and the need to combat illicit drug trade.
- Key Drivers in Retail Pharmacy:
- Mandatory serialization compliance for dispensing prescription medications.
- Improving inventory accuracy and reducing losses due to expiry or theft.
- Enhanced customer trust through verified product authenticity.
- Key Drivers in Retail Pharmacy:
- Online Pharmacy: With the burgeoning growth of e-pharmacies, track and trace systems are becoming crucial for ensuring the integrity of medications delivered directly to consumers, mitigating the risk of counterfeit products entering the online supply chain.
- Key Drivers in Online Pharmacy:
- Combating counterfeit and substandard medicines in the digital marketplace.
- Providing consumers with verifiable proof of origin and authenticity.
- Meeting the increasing demand for convenient and secure pharmaceutical procurement.
- Key Drivers in Online Pharmacy:
Dominant Types of Track and Trace Systems:
- Barcodes Pharmaceutical Track And Trace System: Currently, barcode-based systems, particularly 2D data matrix codes, represent the most widely adopted type due to their cost-effectiveness and compatibility with existing infrastructure.
- Key Drivers for Barcodes:
- Cost-efficiency and widespread adoption across the supply chain.
- Established infrastructure and ease of implementation.
- Adequate for basic serialization and aggregation needs.
- Key Drivers for Barcodes:
- Radio-frequency Identification (RFID) Tags Pharmaceutical Track And Trace System: RFID technology is gaining traction for its ability to offer faster read times, greater data capacity, and the ability to read multiple items simultaneously without direct line-of-sight, crucial for high-volume operations and item-level security.
- Key Drivers for RFID:
- Enhanced efficiency in inventory management and high-speed scanning.
- Greater data storage capacity for comprehensive product information.
- Improved security through unique identification and tamper-evident features.
- Key Drivers for RFID:
- Real-time Locating System (RTLS) Pharmaceutical Track And Trace System: RTLS offers unparalleled visibility into the precise location and movement of pharmaceutical products within a facility or across a broader network, enabling enhanced security, optimized logistics, and immediate response to any deviations.
- Key Drivers for RTLS:
- Real-time monitoring and precise location tracking for high-value or temperature-sensitive pharmaceuticals.
- Enhanced security against theft and diversion within manufacturing and storage facilities.
- Optimized logistics and operational efficiency through detailed movement analysis.
- Key Drivers for RTLS:
pharmaceutical track and trace system Product Developments
Recent product developments in the pharmaceutical track and trace system market center on enhancing data accuracy, speed, and integration. Companies are focusing on advanced serialization solutions, incorporating AI and machine learning for predictive analytics within supply chains, and developing more robust RFID tags and RTLS solutions for real-time, item-level tracking. Innovations include tamper-evident packaging with integrated authentication features and cloud-based platforms that offer seamless data sharing and compliance management across global supply chains. These advancements are crucial for meeting evolving regulatory demands and combating the ever-present threat of counterfeit drugs, thereby strengthening the overall security and integrity of pharmaceutical distribution.
Key Drivers of pharmaceutical track and trace system Growth
The growth of the pharmaceutical track and trace system market is propelled by several critical factors. Foremost among these are stringent government regulations mandating serialization and traceability to combat counterfeit drugs and ensure patient safety, with over XX billion worth of counterfeit drugs seized annually. Technological advancements, including the increasing adoption of IoT, AI, and blockchain, are enabling more sophisticated and efficient tracking solutions. Growing awareness among consumers and healthcare providers about the risks associated with counterfeit medicines is further driving demand for authenticated pharmaceuticals. Economic factors, such as the increasing global pharmaceutical market size, estimated at over XX trillion USD, directly correlate with the need for scaled track and trace solutions.
Challenges in the pharmaceutical track and trace system Market
Despite its robust growth, the pharmaceutical track and trace system market faces several significant challenges. High implementation costs, particularly for smaller manufacturers and pharmacies, can be a substantial barrier. The complexity of integrating new systems with existing legacy infrastructure and ensuring interoperability across diverse supply chain partners presents technical hurdles. Evolving regulatory landscapes across different regions necessitate continuous adaptation and compliance updates, adding to operational burdens. Furthermore, the threat of cyberattacks and data breaches in interconnected systems poses a constant risk to sensitive supply chain information, with an estimated XX% of pharmaceutical companies experiencing data breaches in recent years.
Emerging Opportunities in pharmaceutical track and trace system
Emerging opportunities in the pharmaceutical track and trace system market are vast and driven by technological innovation and evolving global health needs. The increasing demand for personalized medicine and advanced biologics, which often require specialized handling and stringent temperature monitoring, presents a significant opportunity for advanced RTLS and IoT-enabled track and trace solutions. The expansion of e-commerce in the pharmaceutical sector globally, projected to reach over XX billion by 2026, necessitates robust digital track and trace capabilities to ensure product authenticity and consumer safety. Furthermore, the development of interoperable global serialization standards and the integration of blockchain technology for enhanced data security and transparency offer lucrative avenues for solution providers. Strategic partnerships between technology developers, pharmaceutical manufacturers, and regulatory bodies are crucial for unlocking these opportunities.
Leading Players in the pharmaceutical track and trace system Sector
- Marchesini Group
- Weber Marking Systems
- Pester Pac Automation
- Siemens
- Bosch
- Körber
- Uhlmann Packaging System
Key Milestones in pharmaceutical track and trace system Industry
- 2019: Implementation of stricter serialization requirements in several key emerging markets.
- 2020: Increased focus on digital serialization and data management solutions post-pandemic.
- 2021: Significant investments in blockchain technology for pharmaceutical supply chain security.
- 2022: Expansion of RTLS solutions for temperature-sensitive drug tracking.
- 2023: Global harmonization efforts for track and trace data standards gain momentum.
- 2024: Anticipated widespread adoption of AI for predictive analytics in supply chain visibility.
Strategic Outlook for pharmaceutical track and trace system Market
The strategic outlook for the pharmaceutical track and trace system market remains exceptionally positive, driven by an unwavering commitment to supply chain security and patient safety. The continuous evolution of regulatory frameworks across the globe, coupled with the increasing sophistication of counterfeit operations, will continue to fuel the demand for advanced serialization, aggregation, and authentication technologies. Key growth accelerators will include the deeper integration of AI and machine learning for predictive supply chain management, the wider adoption of blockchain for enhanced data integrity and transparency, and the expansion of IoT-enabled solutions for real-time monitoring of critical parameters. Strategic partnerships and collaborations will be vital for navigating the complexities of global supply chains and ensuring seamless interoperability, positioning the market for sustained expansion and innovation in the years to come.
pharmaceutical track and trace system Segmentation
-
1. Application
- 1.1. Hospital Pharmacy
- 1.2. Retail Pharmacy
- 1.3. Online Pharmacy
-
2. Types
- 2.1. Barcodes Pharmaceutical Track And Trace System
- 2.2. Real-time Locating System (RTLS) Pharmaceutical Track And Trace System
- 2.3. Radio-frequency Identification (RFID) Tags Pharmaceutical Track And Trace System
pharmaceutical track and trace system Segmentation By Geography
- 1. CA
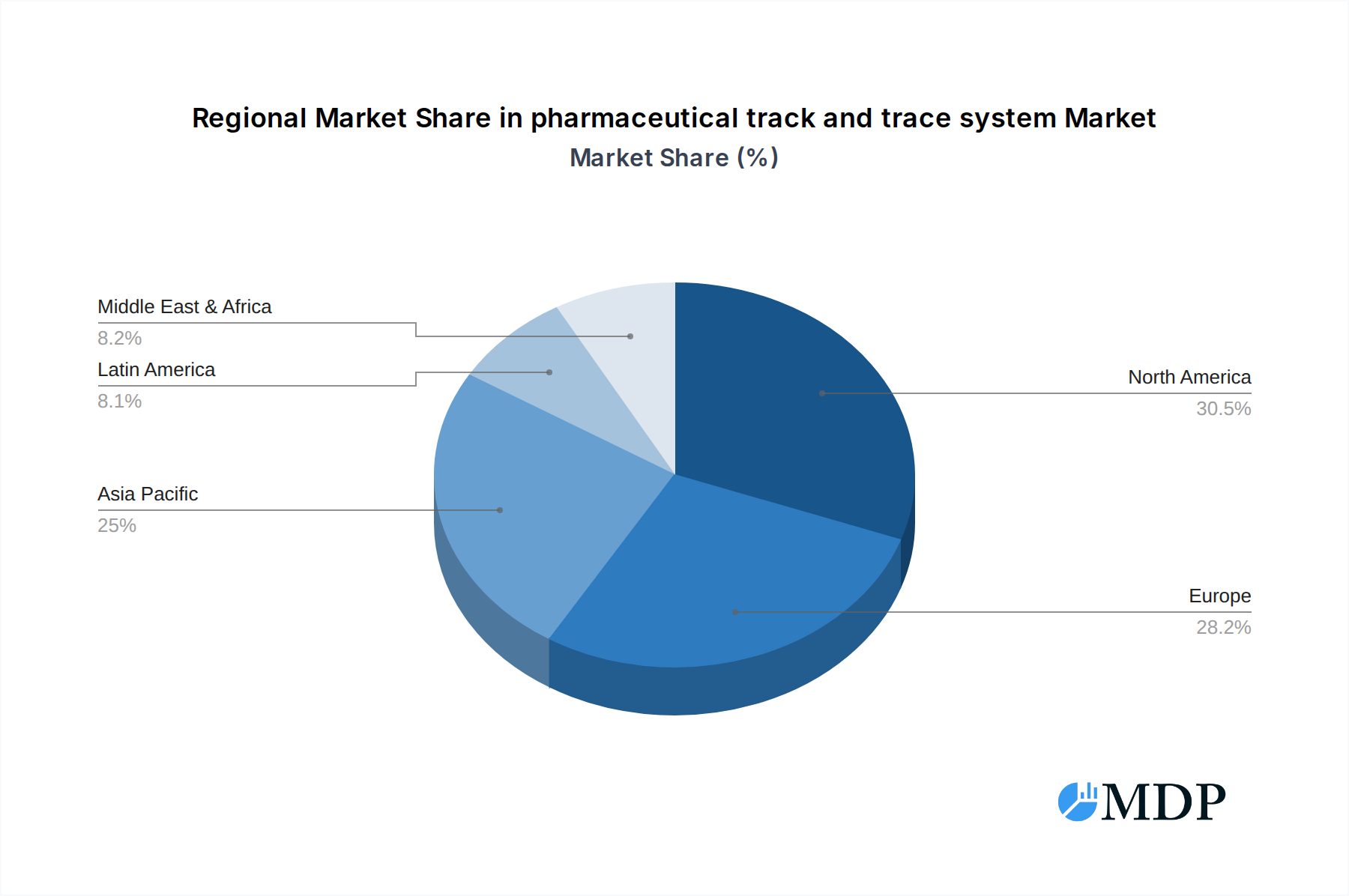
pharmaceutical track and trace system Regional Market Share

Geographic Coverage of pharmaceutical track and trace system
pharmaceutical track and trace system REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 18.95% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. pharmaceutical track and trace system Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital Pharmacy
- 5.1.2. Retail Pharmacy
- 5.1.3. Online Pharmacy
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Barcodes Pharmaceutical Track And Trace System
- 5.2.2. Real-time Locating System (RTLS) Pharmaceutical Track And Trace System
- 5.2.3. Radio-frequency Identification (RFID) Tags Pharmaceutical Track And Trace System
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. CA
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Competitive Analysis
- 6.1. Market Share Analysis 2025
- 6.2. Company Profiles
- 6.2.1 Marchesini Group
- 6.2.1.1. Overview
- 6.2.1.2. Products
- 6.2.1.3. SWOT Analysis
- 6.2.1.4. Recent Developments
- 6.2.1.5. Financials (Based on Availability)
- 6.2.2 Weber Marking Systems
- 6.2.2.1. Overview
- 6.2.2.2. Products
- 6.2.2.3. SWOT Analysis
- 6.2.2.4. Recent Developments
- 6.2.2.5. Financials (Based on Availability)
- 6.2.3 Pester Pac Automation
- 6.2.3.1. Overview
- 6.2.3.2. Products
- 6.2.3.3. SWOT Analysis
- 6.2.3.4. Recent Developments
- 6.2.3.5. Financials (Based on Availability)
- 6.2.4 Siemens
- 6.2.4.1. Overview
- 6.2.4.2. Products
- 6.2.4.3. SWOT Analysis
- 6.2.4.4. Recent Developments
- 6.2.4.5. Financials (Based on Availability)
- 6.2.5 Bosch
- 6.2.5.1. Overview
- 6.2.5.2. Products
- 6.2.5.3. SWOT Analysis
- 6.2.5.4. Recent Developments
- 6.2.5.5. Financials (Based on Availability)
- 6.2.6 Körber
- 6.2.6.1. Overview
- 6.2.6.2. Products
- 6.2.6.3. SWOT Analysis
- 6.2.6.4. Recent Developments
- 6.2.6.5. Financials (Based on Availability)
- 6.2.7 Uhlmann Packaging System
- 6.2.7.1. Overview
- 6.2.7.2. Products
- 6.2.7.3. SWOT Analysis
- 6.2.7.4. Recent Developments
- 6.2.7.5. Financials (Based on Availability)
- 6.2.1 Marchesini Group
List of Figures
- Figure 1: pharmaceutical track and trace system Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: pharmaceutical track and trace system Share (%) by Company 2025
List of Tables
- Table 1: pharmaceutical track and trace system Revenue billion Forecast, by Application 2020 & 2033
- Table 2: pharmaceutical track and trace system Revenue billion Forecast, by Types 2020 & 2033
- Table 3: pharmaceutical track and trace system Revenue billion Forecast, by Region 2020 & 2033
- Table 4: pharmaceutical track and trace system Revenue billion Forecast, by Application 2020 & 2033
- Table 5: pharmaceutical track and trace system Revenue billion Forecast, by Types 2020 & 2033
- Table 6: pharmaceutical track and trace system Revenue billion Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the pharmaceutical track and trace system?
The projected CAGR is approximately 18.95%.
2. Which companies are prominent players in the pharmaceutical track and trace system?
Key companies in the market include Marchesini Group, Weber Marking Systems, Pester Pac Automation, Siemens, Bosch, Körber, Uhlmann Packaging System.
3. What are the main segments of the pharmaceutical track and trace system?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 8.33 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3400.00, USD 5100.00, and USD 6800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "pharmaceutical track and trace system," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the pharmaceutical track and trace system report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the pharmaceutical track and trace system?
To stay informed about further developments, trends, and reports in the pharmaceutical track and trace system, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


