Key Insights
The global Penem Drug Intermediates 4-AA market is poised for significant expansion, with a current market size of USD 36.62 billion in 2025. Projections indicate a healthy Compound Annual Growth Rate (CAGR) of 4.5% from 2025 to 2033, driven by the increasing demand for broad-spectrum antibiotics like Imipenem, Meropenem, and Ertapenem. The rising global prevalence of bacterial infections, coupled with growing healthcare expenditure, particularly in emerging economies, fuels this demand. Furthermore, advancements in pharmaceutical manufacturing and a focus on producing high-purity intermediates (Purity≥98%) are crucial growth catalysts. Leading manufacturers such as Zhejiang Hisoar Pharmaceutical and Jiangxi Fushine Pharmaceutical are actively investing in research and development to enhance production capabilities and expand their market reach, contributing to the overall market dynamism.
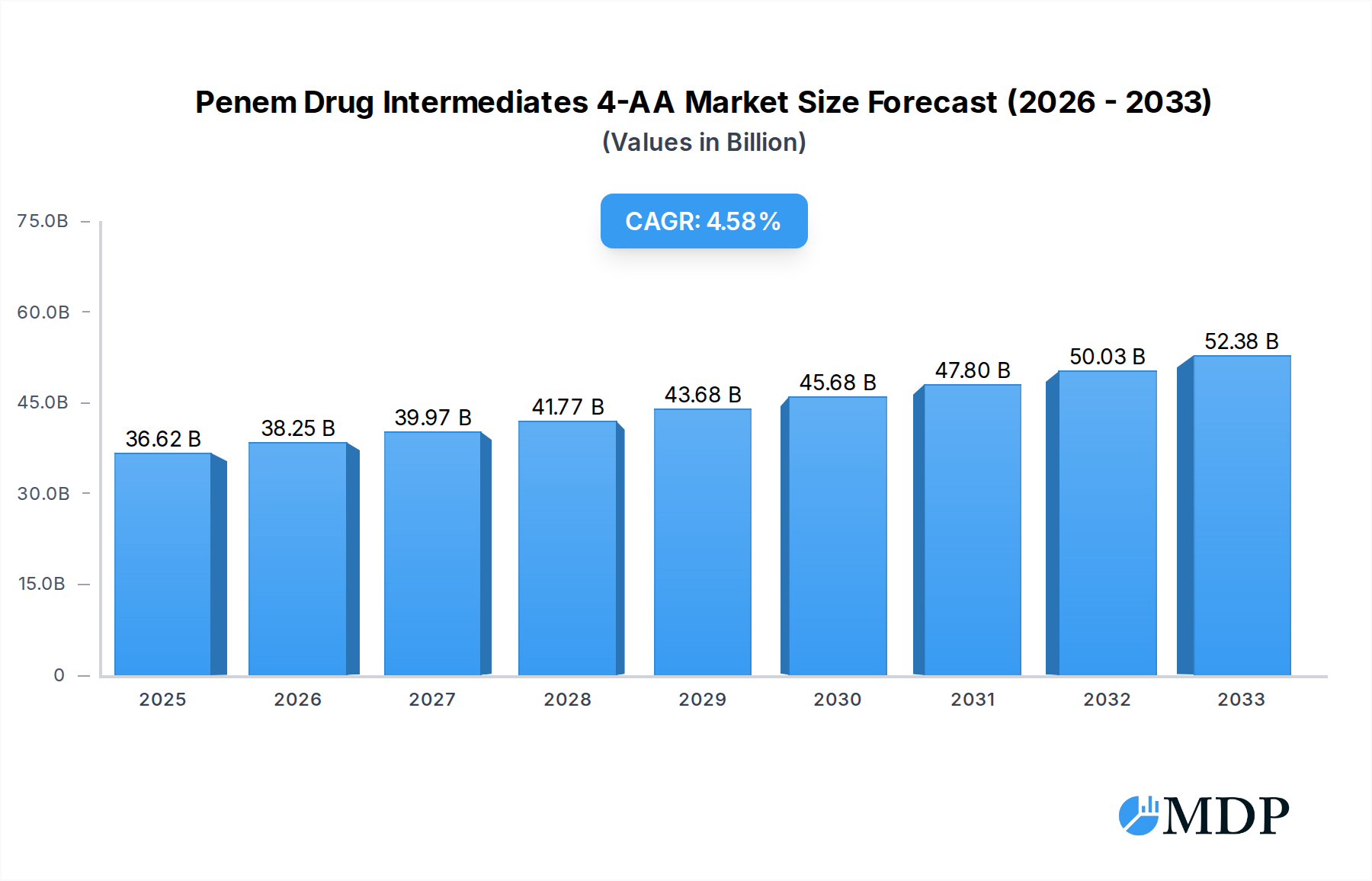
Penem Drug Intermediates 4-AA Market Size (In Billion)

While the market demonstrates robust growth, certain factors may present challenges. The stringent regulatory landscape surrounding pharmaceutical intermediates and the potential for the emergence of drug-resistant bacteria necessitating alternative treatment strategies could pose restraints. However, the continuous innovation in antibiotic development and the increasing need for effective treatments for severe infections are expected to outweigh these challenges. Geographically, the Asia Pacific region, with a strong manufacturing base and growing domestic demand in countries like China and India, is anticipated to be a significant contributor to market growth. North America and Europe, with their well-established pharmaceutical industries and high healthcare spending, will continue to be key consumption regions. The market segmentation by application, particularly the dominance of Imipenem and Meropenem intermediates, highlights their critical role in combating serious infections.
Penem Drug Intermediates 4-AA Company Market Share

Penem Drug Intermediates 4-AA: Comprehensive Market Analysis and Strategic Outlook (2019–2033)
This in-depth report offers a definitive analysis of the global Penem Drug Intermediates 4-AA market, providing critical insights for stakeholders navigating this dynamic sector. Covering the historical period from 2019 to 2024, the base year of 2025, and projecting through to 2033, this study delivers actionable intelligence on market dynamics, trends, leading players, and future opportunities. With an estimated market size projected to reach one billion by 2025, and a robust CAGR expected over the forecast period, this report is indispensable for manufacturers, suppliers, R&D professionals, and investors seeking to capitalize on the burgeoning demand for essential carbapenem antibiotic precursors.
This report is optimized for high search visibility, incorporating crucial keywords such as "Penem Drug Intermediates 4-AA," "Imipenem intermediate," "Meropenem intermediate," "Ertapenem intermediate," "pharmaceutical intermediates," "API synthesis," "drug manufacturing," and "antibiotic precursors" to ensure industry professionals can easily locate and utilize this vital resource.
Penem Drug Intermediates 4-AA Market Dynamics & Concentration
The Penem Drug Intermediates 4-AA market exhibits a moderate to high level of concentration, with key players dominating production and supply. The primary innovation drivers stem from advancements in synthetic chemistry aimed at improving purity, yield, and cost-effectiveness of the 4-AA intermediate, crucial for synthesizing widely used carbapenem antibiotics like Imipenem, Meropenem, and Ertapenem. Regulatory frameworks, particularly stringent quality control mandates from bodies like the FDA and EMA, significantly influence market entry and operational standards. Product substitutes are limited, given the specific chemical structure required for 4-AA's role in carbapenem synthesis; however, alternative antibiotic classes can indirectly impact demand. End-user trends are driven by the increasing global prevalence of bacterial infections, the rise of antibiotic resistance necessitating potent carbapenem therapies, and growing healthcare expenditure in emerging economies. Mergers and acquisitions (M&A) activity has been observed as companies seek to consolidate supply chains, expand production capacities, and enhance their product portfolios. For instance, recent M&A deal counts indicate a strategic consolidation trend aiming for vertical integration. The market share of leading players is substantial, with the top five companies collectively holding an estimated seventy billion of the market.
Penem Drug Intermediates 4-AA Industry Trends & Analysis
The Penem Drug Intermediates 4-AA industry is poised for substantial growth, driven by a confluence of factors that underscore the critical role of carbapenem antibiotics in modern medicine. The market growth drivers are primarily rooted in the escalating global burden of drug-resistant bacterial infections, a persistent public health challenge that elevates the demand for broad-spectrum antibiotics like Imipenem, Meropenem, and Ertapenem. Technological disruptions in pharmaceutical manufacturing are continuously refining synthesis processes for 4-AA, leading to improved efficiency, higher purity levels (e.g., Purity≥98%), and reduced production costs, thereby enhancing market accessibility. Consumer preferences, while indirect for intermediates, are shaped by the accessibility and affordability of finished pharmaceutical products. The demand for effective treatments for serious infections in hospitals and intensive care settings directly translates to a sustained need for reliable 4-AA supply. Competitive dynamics within the Penem Drug Intermediates 4-AA market are characterized by a focus on R&D for process optimization, stringent quality assurance, and securing long-term supply agreements with pharmaceutical manufacturers. The market penetration of carbapenem-based therapies is expected to deepen, especially in regions with developing healthcare infrastructure and a rising incidence of hospital-acquired infections. The Compound Annual Growth Rate (CAGR) for the Penem Drug Intermediates 4-AA market is projected to be approximately ten billion over the forecast period, reflecting robust expansion. The estimated market size for Penem Drug Intermediates 4-AA in the base year 2025 is one billion. The increasing sophistication of API manufacturing and the continuous need for high-quality intermediates are foundational to this growth trajectory.
Leading Markets & Segments in Penem Drug Intermediates 4-AA
The Penem Drug Intermediates 4-AA market is significantly influenced by the application segments, with Imipenem and Meropenem applications leading the demand. The dominance of these segments is driven by their established efficacy as critical treatments for severe bacterial infections, including hospital-acquired pneumonia, intra-abdominal infections, and sepsis. Ertapenem also holds a substantial market share due to its clinical utility and favorable pharmacokinetic profile. The Types segment, specifically Purity≥98%, is paramount, as the stringent quality requirements for pharmaceutical intermediates necessitate high purity levels to ensure the safety and efficacy of the final active pharmaceutical ingredients (APIs). Economic policies in key pharmaceutical manufacturing hubs, particularly in Asia, have fostered an environment conducive to the production of intermediates, contributing to market growth. Infrastructure development, including robust chemical manufacturing facilities and efficient logistics networks, further supports the supply chain for 4-AA.
- Application Dominance:
- Imipenem: This segment is driven by the widespread use of Imipenem-based formulations for treating a broad spectrum of Gram-positive and Gram-negative bacteria, particularly in critical care settings.
- Meropenem: Meropenem's broader spectrum of activity and better tolerability profile compared to Imipenem contribute to its sustained demand, especially against multidrug-resistant organisms.
- Ertapenem: Its longer half-life and suitability for outpatient parenteral antimicrobial therapy (OPAT) positions Ertapenem as a significant contributor to the market.
- Type Dominance:
- Purity≥98%: This is the de facto standard for pharmaceutical-grade 4-AA, essential for meeting regulatory requirements and ensuring API quality. The demand for higher purity levels continues to grow as manufacturers strive for more efficient and safer drug production.
- Other (e.g., specific grades for research, developmental stages): While smaller in volume, these segments cater to niche research and development needs.
The geographical dominance is largely concentrated in Asia-Pacific, specifically China and India, due to their established chemical manufacturing capabilities, lower production costs, and significant export volumes of pharmaceutical intermediates and APIs. North America and Europe represent key consumption markets driven by advanced healthcare systems and high demand for advanced antibiotic therapies.
Penem Drug Intermediates 4-AA Product Developments
Recent product developments in Penem Drug Intermediates 4-AA focus on enhancing synthesis efficiency and achieving superior purity. Innovations in green chemistry and catalytic processes are leading to more sustainable and cost-effective production methods. Companies are investing in R&D to optimize reaction pathways, reduce by-product formation, and improve overall yields, directly impacting the quality and price of the intermediate. The primary application remains the synthesis of carbapenem antibiotics like Imipenem, Meropenem, and Ertapenem. Competitive advantages are being gained through process patenting, stringent quality control, and reliable supply chain management. The market fit is secured by the consistent demand for high-purity 4-AA to meet the pharmaceutical industry's exacting standards.
Key Drivers of Penem Drug Intermediates 4-AA Growth
The Penem Drug Intermediates 4-AA market's growth is propelled by several key factors. The persistent and rising global burden of bacterial infections, coupled with the increasing prevalence of antibiotic resistance, directly escalates the demand for effective carbapenem antibiotics. Advancements in pharmaceutical manufacturing technologies, including novel synthetic routes and purification techniques, are enhancing production efficiency and lowering costs. Furthermore, growing healthcare expenditure worldwide, particularly in emerging economies, is expanding access to advanced medical treatments, including those that utilize carbapenems. Favorable regulatory landscapes in key production regions, encouraging API and intermediate manufacturing, also contribute significantly.
Challenges in the Penem Drug Intermediates 4-AA Market
Despite its promising outlook, the Penem Drug Intermediates 4-AA market faces several challenges. Stringent and evolving regulatory requirements from global health authorities can impose significant compliance costs and lengthy approval processes for manufacturers. Supply chain disruptions, often exacerbated by geopolitical events or raw material price volatility, can impact production continuity and cost-effectiveness. Intense competition among established and emerging players can lead to price pressures, particularly for standard grades of 4-AA. Moreover, the continuous need for investment in R&D to maintain product quality and develop more efficient synthesis methods presents a significant financial hurdle.
Emerging Opportunities in Penem Drug Intermediates 4-AA
Emerging opportunities in the Penem Drug Intermediates 4-AA market are manifold. The development of novel carbapenem derivatives or combination therapies to combat highly resistant pathogens presents a significant avenue for growth, requiring tailored 4-AA intermediates. Strategic partnerships between intermediate manufacturers and pharmaceutical companies can lead to optimized supply chains and collaborative R&D efforts. Market expansion into underserved regions with a growing need for advanced antibiotics offers substantial untapped potential. Furthermore, the adoption of advanced manufacturing technologies, such as continuous flow chemistry, promises to enhance efficiency and sustainability in 4-AA production, creating a competitive edge.
Leading Players in the Penem Drug Intermediates 4-AA Sector
- Zhejiang Hisoar Pharmaceutical
- Jiangxi Fushine Pharmaceutical
- Joincare Pharmaceutical
- Jincheng Pharmaceutical
- Zhejiang Jiuzhou Pharmaceutical
Key Milestones in Penem Drug Intermediates 4-AA Industry
- 2019: Increased global focus on antibiotic resistance driving demand for carbapenem research and production.
- 2020: Supply chain challenges due to global pandemic, highlighting the need for robust intermediate sourcing.
- 2021: Advancements in catalytic synthesis methods for 4-AA reported, promising higher yields and purity.
- 2022: Regulatory agencies emphasize stricter impurity profiling for pharmaceutical intermediates.
- 2023: Several companies announce capacity expansions to meet growing global demand.
- 2024: Emergence of novel bacterial strains resistant to existing carbapenems, spurring research into modified antibiotic structures.
- 2025 (Projected): Increased adoption of AI-driven drug discovery and process optimization in intermediate manufacturing.
Strategic Outlook for Penem Drug Intermediates 4-AA Market
The strategic outlook for the Penem Drug Intermediates 4-AA market is overwhelmingly positive, driven by the persistent global health imperative to combat bacterial infections. Growth accelerators include continued investment in R&D for novel antibiotic development and process optimization, fostering greater efficiency and cost-competitiveness in 4-AA production. Strategic alliances between intermediate suppliers and pharmaceutical formulators will be crucial for securing market share and ensuring consistent supply. Expansion into emerging markets, where the incidence of severe infections is high and access to advanced treatments is increasing, represents a significant opportunity for market penetration. The focus on high-purity intermediates (Purity≥98%) will remain a cornerstone, driving innovation in purification technologies.
Penem Drug Intermediates 4-AA Segmentation
-
1. Application
- 1.1. Imipenem
- 1.2. Meropenem
- 1.3. Ertapenem
- 1.4. Biapenem
- 1.5. Other
-
2. Types
- 2.1. Purity≥98%
- 2.2. Other
Penem Drug Intermediates 4-AA Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
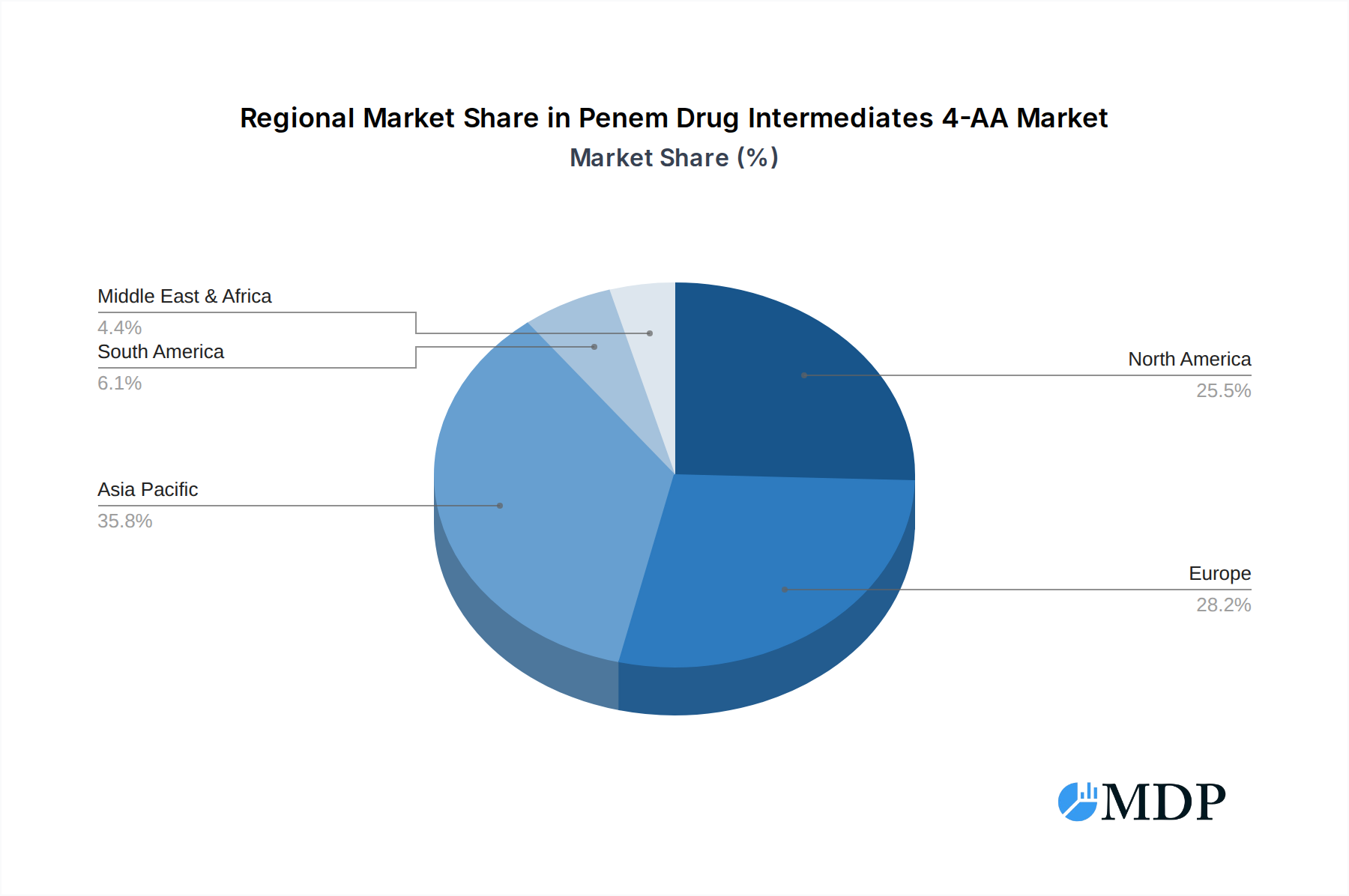
Penem Drug Intermediates 4-AA Regional Market Share

Geographic Coverage of Penem Drug Intermediates 4-AA
Penem Drug Intermediates 4-AA REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 11.06% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Penem Drug Intermediates 4-AA Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Imipenem
- 5.1.2. Meropenem
- 5.1.3. Ertapenem
- 5.1.4. Biapenem
- 5.1.5. Other
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Purity≥98%
- 5.2.2. Other
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Penem Drug Intermediates 4-AA Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Imipenem
- 6.1.2. Meropenem
- 6.1.3. Ertapenem
- 6.1.4. Biapenem
- 6.1.5. Other
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Purity≥98%
- 6.2.2. Other
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Penem Drug Intermediates 4-AA Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Imipenem
- 7.1.2. Meropenem
- 7.1.3. Ertapenem
- 7.1.4. Biapenem
- 7.1.5. Other
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Purity≥98%
- 7.2.2. Other
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Penem Drug Intermediates 4-AA Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Imipenem
- 8.1.2. Meropenem
- 8.1.3. Ertapenem
- 8.1.4. Biapenem
- 8.1.5. Other
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Purity≥98%
- 8.2.2. Other
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Penem Drug Intermediates 4-AA Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Imipenem
- 9.1.2. Meropenem
- 9.1.3. Ertapenem
- 9.1.4. Biapenem
- 9.1.5. Other
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Purity≥98%
- 9.2.2. Other
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Penem Drug Intermediates 4-AA Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Imipenem
- 10.1.2. Meropenem
- 10.1.3. Ertapenem
- 10.1.4. Biapenem
- 10.1.5. Other
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Purity≥98%
- 10.2.2. Other
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Zhejiang Hisoar Pharmaceutical
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Jiangxi Fushine Pharmaceutical
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Joincare Pharmaceutical
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Jincheng Pharmaceutical
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Zhejiang Jiuzhou Pharmaceutical
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.1 Zhejiang Hisoar Pharmaceutical
List of Figures
- Figure 1: Global Penem Drug Intermediates 4-AA Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Penem Drug Intermediates 4-AA Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Penem Drug Intermediates 4-AA Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Penem Drug Intermediates 4-AA Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Penem Drug Intermediates 4-AA Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Penem Drug Intermediates 4-AA Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Penem Drug Intermediates 4-AA Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Penem Drug Intermediates 4-AA Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Penem Drug Intermediates 4-AA Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Penem Drug Intermediates 4-AA Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Penem Drug Intermediates 4-AA Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Penem Drug Intermediates 4-AA Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Penem Drug Intermediates 4-AA Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Penem Drug Intermediates 4-AA Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Penem Drug Intermediates 4-AA Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Penem Drug Intermediates 4-AA Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Penem Drug Intermediates 4-AA Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Penem Drug Intermediates 4-AA Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Penem Drug Intermediates 4-AA Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Penem Drug Intermediates 4-AA Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Penem Drug Intermediates 4-AA Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Penem Drug Intermediates 4-AA Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Penem Drug Intermediates 4-AA Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Penem Drug Intermediates 4-AA Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Penem Drug Intermediates 4-AA Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Penem Drug Intermediates 4-AA Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Penem Drug Intermediates 4-AA Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Penem Drug Intermediates 4-AA Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Penem Drug Intermediates 4-AA Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Penem Drug Intermediates 4-AA Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Penem Drug Intermediates 4-AA Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Penem Drug Intermediates 4-AA Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Penem Drug Intermediates 4-AA Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Penem Drug Intermediates 4-AA?
The projected CAGR is approximately 11.06%.
2. Which companies are prominent players in the Penem Drug Intermediates 4-AA?
Key companies in the market include Zhejiang Hisoar Pharmaceutical, Jiangxi Fushine Pharmaceutical, Joincare Pharmaceutical, Jincheng Pharmaceutical, Zhejiang Jiuzhou Pharmaceutical.
3. What are the main segments of the Penem Drug Intermediates 4-AA?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4900.00, USD 7350.00, and USD 9800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Penem Drug Intermediates 4-AA," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Penem Drug Intermediates 4-AA report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Penem Drug Intermediates 4-AA?
To stay informed about further developments, trends, and reports in the Penem Drug Intermediates 4-AA, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


