Key Insights
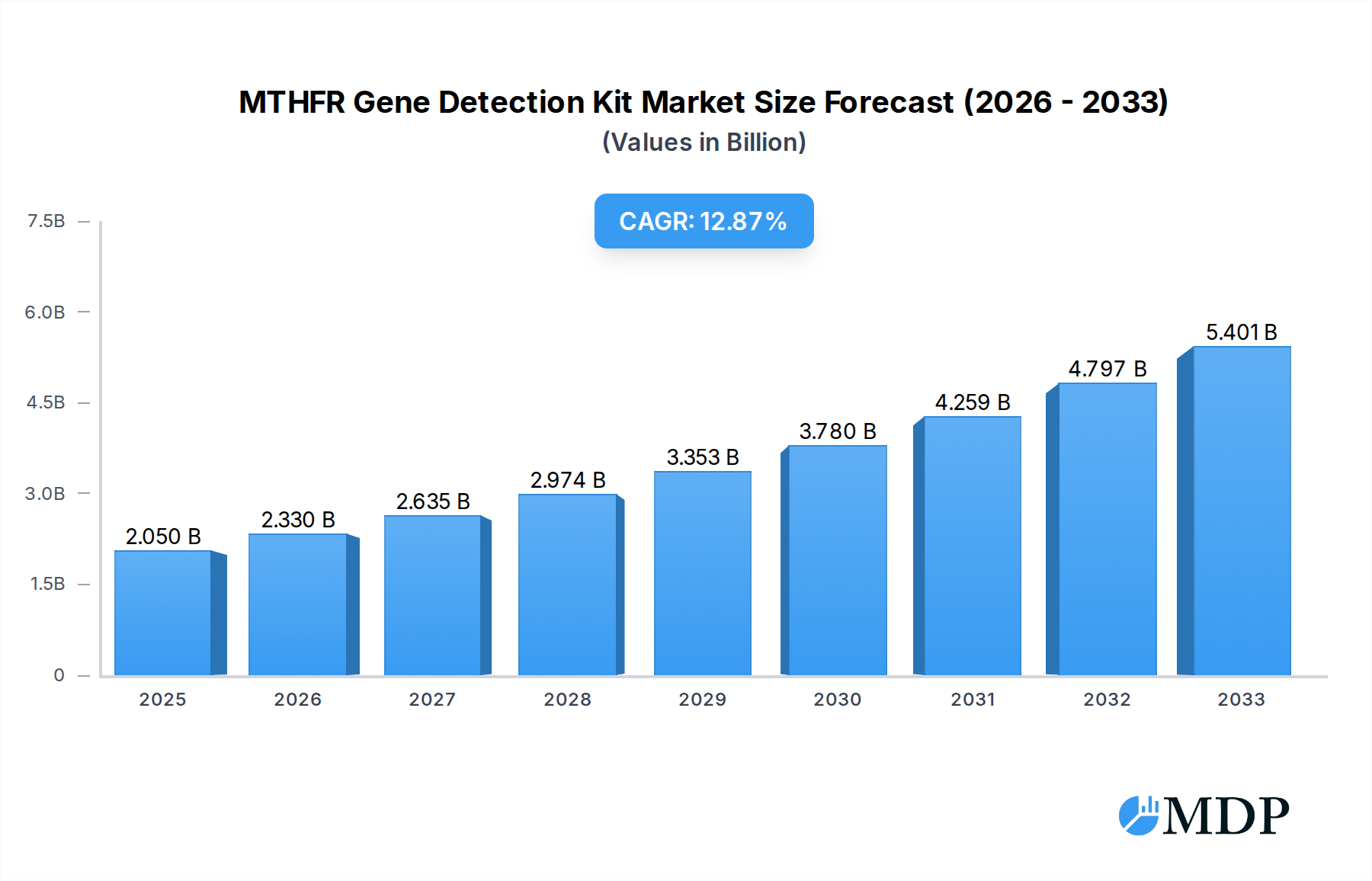
The MTHFR Gene Detection Kit market is poised for significant expansion, projected to reach $2.05 billion in 2025 with a robust CAGR of 13.64% through 2033. This growth is fueled by a heightened awareness of the MTHFR gene's role in various health conditions, including cardiovascular diseases, neurological disorders, and pregnancy complications. The increasing prevalence of genetic testing, coupled with advancements in diagnostic technologies like PCR-fluorescent probe methods and fluorescent PCR melting curve analysis, is making MTHFR gene detection more accessible and accurate. Key drivers include the rising demand for personalized medicine, where understanding an individual's genetic predisposition allows for tailored preventative strategies and treatment plans. Furthermore, the growing focus on reproductive health and the management of infertility is contributing to the increased adoption of MTHFR gene testing, particularly in hospital obstetrics and gynecology departments and genetic reproductive centers. The expanding healthcare infrastructure in emerging economies and increased investment in genetic research and development are also expected to propel market growth.
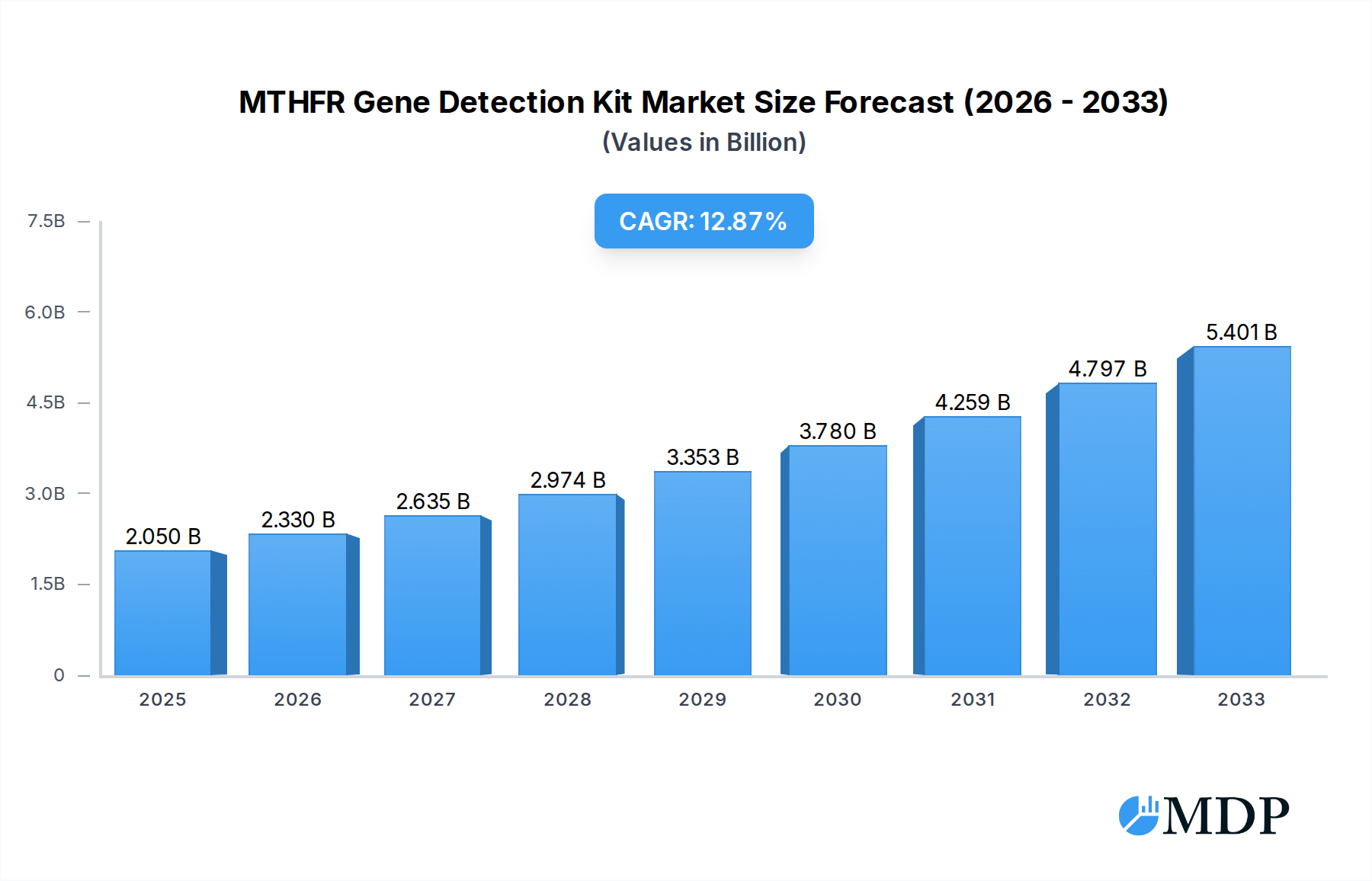
MTHFR Gene Detection Kit Market Size (In Billion)

The MTHFR Gene Detection Kit market's trajectory is also influenced by evolving trends such as the integration of artificial intelligence and machine learning in genetic data analysis, enhancing the interpretation of test results and their clinical implications. Home-based testing kits are emerging as a significant trend, offering convenience and wider accessibility to consumers. However, the market faces certain restraints, including stringent regulatory frameworks for diagnostic tests, concerns regarding data privacy and security, and the initial cost of advanced testing equipment for smaller healthcare facilities. Despite these challenges, the inherent value of MTHFR gene testing in proactive health management and disease prevention is expected to outweigh these limitations. The market is characterized by intense competition among established players and emerging innovators, driving continuous product development and market penetration across diverse geographical regions, with Asia Pacific and North America expected to be leading markets.
MTHFR Gene Detection Kit Company Market Share

MTHFR Gene Detection Kit Market Report Description
Unlock comprehensive insights into the burgeoning MTHFR Gene Detection Kit market with our in-depth report. Covering the period from 2019–2033, with a base year of 2025, this report provides a detailed analysis of market dynamics, key trends, leading segments, and strategic outlooks. We leverage high-traffic keywords to ensure maximum search visibility for industry stakeholders including genetic testing companies, diagnostic laboratories, healthcare providers, and investors. This report is essential for understanding the billion-dollar MTHFR gene detection market's current landscape and future trajectory.
MTHFR Gene Detection Kit Market Dynamics & Concentration
The MTHFR Gene Detection Kit market exhibits moderate to high concentration, with a significant portion of market share held by established players like LabCorp and 23andMe, alongside emerging innovators such as Empower DX and Mega Genomics. Innovation drivers are primarily fueled by advancements in PCR technology and a growing understanding of the MTHFR gene's implications in various health conditions, from cardiovascular disease risk to adverse drug reactions and pregnancy complications. Regulatory frameworks, while generally supportive of diagnostic tool development, vary by region, influencing market entry and product approval timelines. Product substitutes are limited in direct scope, as specific MTHFR detection kits offer targeted genotyping, but broader genetic screening panels can indirectly compete. End-user trends highlight a rising demand for personalized medicine and proactive health management, driving adoption in both clinical and direct-to-consumer settings. Merger and acquisition (M&A) activities are anticipated to increase as larger diagnostic companies seek to expand their genetic testing portfolios. We estimate approximately 20 billion in M&A deal volume over the forecast period, with a projected 15 M&A transactions.
MTHFR Gene Detection Kit Industry Trends & Analysis
The MTHFR Gene Detection Kit industry is poised for substantial growth, driven by an escalating awareness of genetic predispositions to health issues and the increasing integration of genetic testing into routine healthcare. The market is projected to expand at a Compound Annual Growth Rate (CAGR) of approximately 12.5 billion, reaching an estimated market size of 45 billion by 2033. Key market growth drivers include the rising prevalence of folate-related health concerns, such as neural tube defects and cardiovascular disease, which are linked to MTHFR gene variations. Technological disruptions, particularly the refinement of PCR-fluorescent probe and melting curve methodologies, are enhancing sensitivity, specificity, and turnaround times for MTHFR gene detection, making these kits more accessible and cost-effective. Consumer preferences are shifting towards proactive health management and personalized nutrition plans, with individuals increasingly seeking to understand their genetic makeup to optimize their well-being. The competitive landscape is characterized by both established diagnostic giants and agile biotech startups, all vying for market share through product innovation and strategic partnerships. Market penetration is expected to deepen as reimbursement policies for genetic testing become more favorable, further driving adoption across various healthcare settings.
Leading Markets & Segments in MTHFR Gene Detection Kit
The Hospital Obstetrics and Gynecology segment is projected to be the dominant force in the MTHFR Gene Detection Kit market, driven by the critical role of MTHFR gene variations in pregnancy outcomes, including risks of miscarriage, preeclampsia, and neural tube defects. This segment's dominance is supported by robust economic policies promoting prenatal care and genetic screening initiatives in developed nations, translating to an estimated market share of 30 billion.
- Key Drivers in Hospital Obstetrics and Gynecology:
- High Incidence of Pregnancy Complications: MTHFR gene mutations are strongly associated with adverse pregnancy outcomes, making routine screening a clinical imperative.
- Governmental Health Initiatives: Proactive policies encouraging prenatal genetic testing and folic acid supplementation directly boost demand for MTHFR kits.
- Advancements in Reproductive Medicine: The growth of assisted reproductive technologies (ART) necessitates comprehensive genetic screening for optimal patient management.
- Increasing Awareness Among Healthcare Providers: Medical professionals are increasingly recognizing the clinical utility of MTHFR testing in managing reproductive health.
The Genetic Reproductive Center segment also represents a significant and growing market, estimated at 25 billion, due to its specialized focus on fertility and genetic counseling. These centers utilize MTHFR gene detection kits to assess risks and guide treatment decisions for couples facing infertility or recurrent pregnancy loss.
The PCR-fluorescent Probe Method type is expected to capture the largest market share, estimated at 35 billion, owing to its superior sensitivity, specificity, and rapid detection capabilities, which are crucial for clinical diagnostics. The Fluorescent PCR Melting Curve Method is also gaining traction, offering a cost-effective and efficient alternative for high-throughput screening, contributing an estimated 20 billion to the market.
MTHFR Gene Detection Kit Product Developments
Recent product developments in the MTHFR Gene Detection Kit sector are characterized by enhanced multiplexing capabilities, improved assay sensitivity, and user-friendly designs for both laboratory and point-of-care settings. Innovations are focusing on reducing assay times to mere hours and increasing the precision of mutation detection for C677T and A1298C variants. Companies are also developing kits compatible with existing laboratory equipment, minimizing capital investment for adopters. These advancements aim to broaden the application of MTHFR testing beyond specialized genetic labs into primary care and obstetrics, facilitating earlier diagnosis and personalized interventions. The competitive advantage lies in kits offering higher throughput, lower cost per test, and seamless integration into clinical workflows.
Key Drivers of MTHFR Gene Detection Kit Growth
The MTHFR Gene Detection Kit market is propelled by several key growth drivers. Firstly, the increasing global awareness of personalized medicine and the role of genetics in health outcomes is paramount. Secondly, a rise in chronic diseases and pregnancy complications linked to MTHFR gene variants, such as cardiovascular disease and neural tube defects, fuels demand for accurate diagnostic tools. Thirdly, technological advancements in molecular diagnostics, including the refinement of PCR-based methods, are leading to more accurate, faster, and cost-effective testing solutions. Finally, favorable regulatory landscapes in many regions and growing reimbursement for genetic testing further incentivize market expansion.
Challenges in the MTHFR Gene Detection Kit Market
Despite its promising growth, the MTHFR Gene Detection Kit market faces several challenges. Regulatory hurdles and the complex approval processes in different countries can slow down market entry and expansion. The high cost of advanced genetic testing equipment and reagents can be a barrier for smaller laboratories and healthcare facilities, particularly in developing economies, impacting market penetration. Moreover, competition from broader genetic screening panels, which may include MTHFR analysis as part of a larger offering, presents a competitive pressure. Supply chain disruptions and the need for specialized personnel to operate and interpret results can also pose significant challenges.
Emerging Opportunities in MTHFR Gene Detection Kit
Emerging opportunities in the MTHFR Gene Detection Kit market are abundant and driven by several catalysts. The expanding application of MTHFR testing in pharmacogenomics, predicting drug efficacy and adverse reactions, presents a significant growth avenue. Furthermore, the increasing adoption of direct-to-consumer (DTC) genetic testing offers a burgeoning market for home-based MTHFR detection kits. Strategic partnerships between kit manufacturers and healthcare providers, as well as the development of novel companion diagnostics for gene-editing therapies, are poised to unlock substantial market potential. Technological breakthroughs in CRISPR-based diagnostics and liquid biopsy techniques could also revolutionize MTHFR gene detection, creating new market niches.
Leading Players in the MTHFR Gene Detection Kit Sector
- 23andMe
- LabCorp
- Empower DX
- Mega Genomics
- Blueprint Genetics
- Generi-Biotech
- RxHomeTest
- Kashi Labs
- SolGent
- CapitalBio
- Genex Diagnostics
- Agena Bioscience
- Sansure
- Coyote Bio
- BaiO
Key Milestones in MTHFR Gene Detection Kit Industry
- 2019: Increased research highlighting MTHFR gene links to chronic disease risk.
- 2020: Launch of new, more sensitive PCR-fluorescent probe kits by several key players.
- 2021: Regulatory approvals for expanded use of MTHFR testing in prenatal care in select regions.
- 2022: Rise in direct-to-consumer genetic testing companies offering MTHFR analysis.
- 2023: Significant investment in MTHFR gene detection technology R&D by major diagnostic firms.
- 2024: Introduction of faster assay turnaround times for MTHFR kits.
Strategic Outlook for MTHFR Gene Detection Kit Market
The strategic outlook for the MTHFR Gene Detection Kit market is exceptionally positive, driven by converging trends in personalized medicine, preventative healthcare, and technological innovation. Growth accelerators will include the expanding role of MTHFR testing in pharmacogenomics and the increasing integration into routine prenatal and general health screenings. The market will witness a surge in demand for user-friendly, high-throughput, and cost-effective solutions. Companies that focus on strategic partnerships with healthcare providers, expand their global reach through regulatory approvals, and invest in next-generation diagnostic technologies will be best positioned to capitalize on the substantial future market potential, estimated to grow by over 50 billion in the next decade.
MTHFR Gene Detection Kit Segmentation
-
1. Application
- 1.1. Hospital Obstetrics and Gynecology
- 1.2. Genetic Reproductive Center
- 1.3. Medical Center
- 1.4. Others
-
2. Types
- 2.1. PCR-fluorescent Probe Method
- 2.2. Fluorescent PCR Melting Curve Method
MTHFR Gene Detection Kit Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
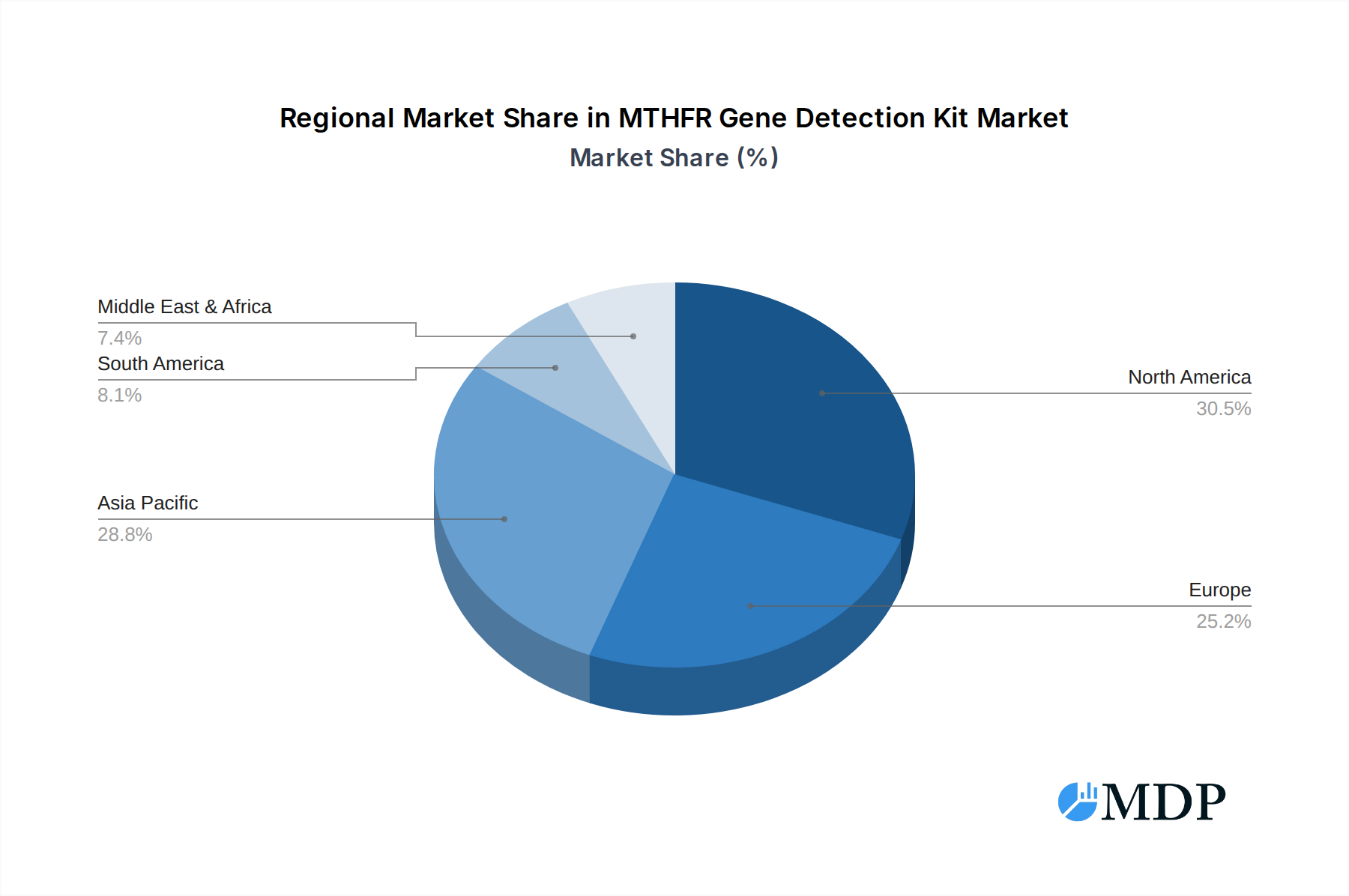
MTHFR Gene Detection Kit Regional Market Share

Geographic Coverage of MTHFR Gene Detection Kit
MTHFR Gene Detection Kit REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 13.64% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global MTHFR Gene Detection Kit Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital Obstetrics and Gynecology
- 5.1.2. Genetic Reproductive Center
- 5.1.3. Medical Center
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. PCR-fluorescent Probe Method
- 5.2.2. Fluorescent PCR Melting Curve Method
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America MTHFR Gene Detection Kit Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital Obstetrics and Gynecology
- 6.1.2. Genetic Reproductive Center
- 6.1.3. Medical Center
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. PCR-fluorescent Probe Method
- 6.2.2. Fluorescent PCR Melting Curve Method
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America MTHFR Gene Detection Kit Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital Obstetrics and Gynecology
- 7.1.2. Genetic Reproductive Center
- 7.1.3. Medical Center
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. PCR-fluorescent Probe Method
- 7.2.2. Fluorescent PCR Melting Curve Method
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe MTHFR Gene Detection Kit Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital Obstetrics and Gynecology
- 8.1.2. Genetic Reproductive Center
- 8.1.3. Medical Center
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. PCR-fluorescent Probe Method
- 8.2.2. Fluorescent PCR Melting Curve Method
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa MTHFR Gene Detection Kit Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital Obstetrics and Gynecology
- 9.1.2. Genetic Reproductive Center
- 9.1.3. Medical Center
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. PCR-fluorescent Probe Method
- 9.2.2. Fluorescent PCR Melting Curve Method
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific MTHFR Gene Detection Kit Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital Obstetrics and Gynecology
- 10.1.2. Genetic Reproductive Center
- 10.1.3. Medical Center
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. PCR-fluorescent Probe Method
- 10.2.2. Fluorescent PCR Melting Curve Method
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 23andMe
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 LabCorp
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Empower DX
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Mega Genomics
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Blueprint Genetics
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Generi-Biotech
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 RxHomeTest
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Kashi Labs
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 SolGent
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 CapitalBio
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Genex Diagnostics
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Agena Bioscience
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Sansure
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Coyote Bio
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 BaiO
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.1 23andMe
List of Figures
- Figure 1: Global MTHFR Gene Detection Kit Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America MTHFR Gene Detection Kit Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America MTHFR Gene Detection Kit Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America MTHFR Gene Detection Kit Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America MTHFR Gene Detection Kit Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America MTHFR Gene Detection Kit Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America MTHFR Gene Detection Kit Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America MTHFR Gene Detection Kit Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America MTHFR Gene Detection Kit Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America MTHFR Gene Detection Kit Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America MTHFR Gene Detection Kit Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America MTHFR Gene Detection Kit Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America MTHFR Gene Detection Kit Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe MTHFR Gene Detection Kit Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe MTHFR Gene Detection Kit Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe MTHFR Gene Detection Kit Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe MTHFR Gene Detection Kit Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe MTHFR Gene Detection Kit Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe MTHFR Gene Detection Kit Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa MTHFR Gene Detection Kit Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa MTHFR Gene Detection Kit Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa MTHFR Gene Detection Kit Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa MTHFR Gene Detection Kit Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa MTHFR Gene Detection Kit Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa MTHFR Gene Detection Kit Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific MTHFR Gene Detection Kit Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific MTHFR Gene Detection Kit Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific MTHFR Gene Detection Kit Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific MTHFR Gene Detection Kit Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific MTHFR Gene Detection Kit Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific MTHFR Gene Detection Kit Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global MTHFR Gene Detection Kit Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific MTHFR Gene Detection Kit Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the MTHFR Gene Detection Kit?
The projected CAGR is approximately 13.64%.
2. Which companies are prominent players in the MTHFR Gene Detection Kit?
Key companies in the market include 23andMe, LabCorp, Empower DX, Mega Genomics, Blueprint Genetics, Generi-Biotech, RxHomeTest, Kashi Labs, SolGent, CapitalBio, Genex Diagnostics, Agena Bioscience, Sansure, Coyote Bio, BaiO.
3. What are the main segments of the MTHFR Gene Detection Kit?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "MTHFR Gene Detection Kit," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the MTHFR Gene Detection Kit report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the MTHFR Gene Detection Kit?
To stay informed about further developments, trends, and reports in the MTHFR Gene Detection Kit, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


