Key Insights
The Inhaled COVID-19 Vaccine market is poised for substantial expansion, projected to reach an estimated USD 5.87 billion in 2025, driven by a compelling Compound Annual Growth Rate (CAGR) of 13.96% through 2033. This robust growth is primarily fueled by the inherent advantages of inhaled vaccines, including ease of administration, reduced needle-phobia, and the potential for enhanced mucosal immunity directly at the entry point of the virus. The increasing global demand for more convenient and effective vaccination strategies, coupled with ongoing research and development efforts, are significant drivers. Furthermore, the evolving nature of the COVID-19 virus, necessitating regular booster shots and variant-specific vaccines, creates a continuous market opportunity. The market is segmenting across various applications, with Medical and Research sectors being key beneficiaries. The 30 Years and Above age demographic is expected to represent a larger share due to higher susceptibility and the established vaccination infrastructure, while the 18-30 Years Old segment will also contribute significantly as public health initiatives promote broader accessibility and preventive care. Companies like CanSino Biologics are at the forefront of this innovation, showcasing the competitive landscape and the potential for significant market penetration.
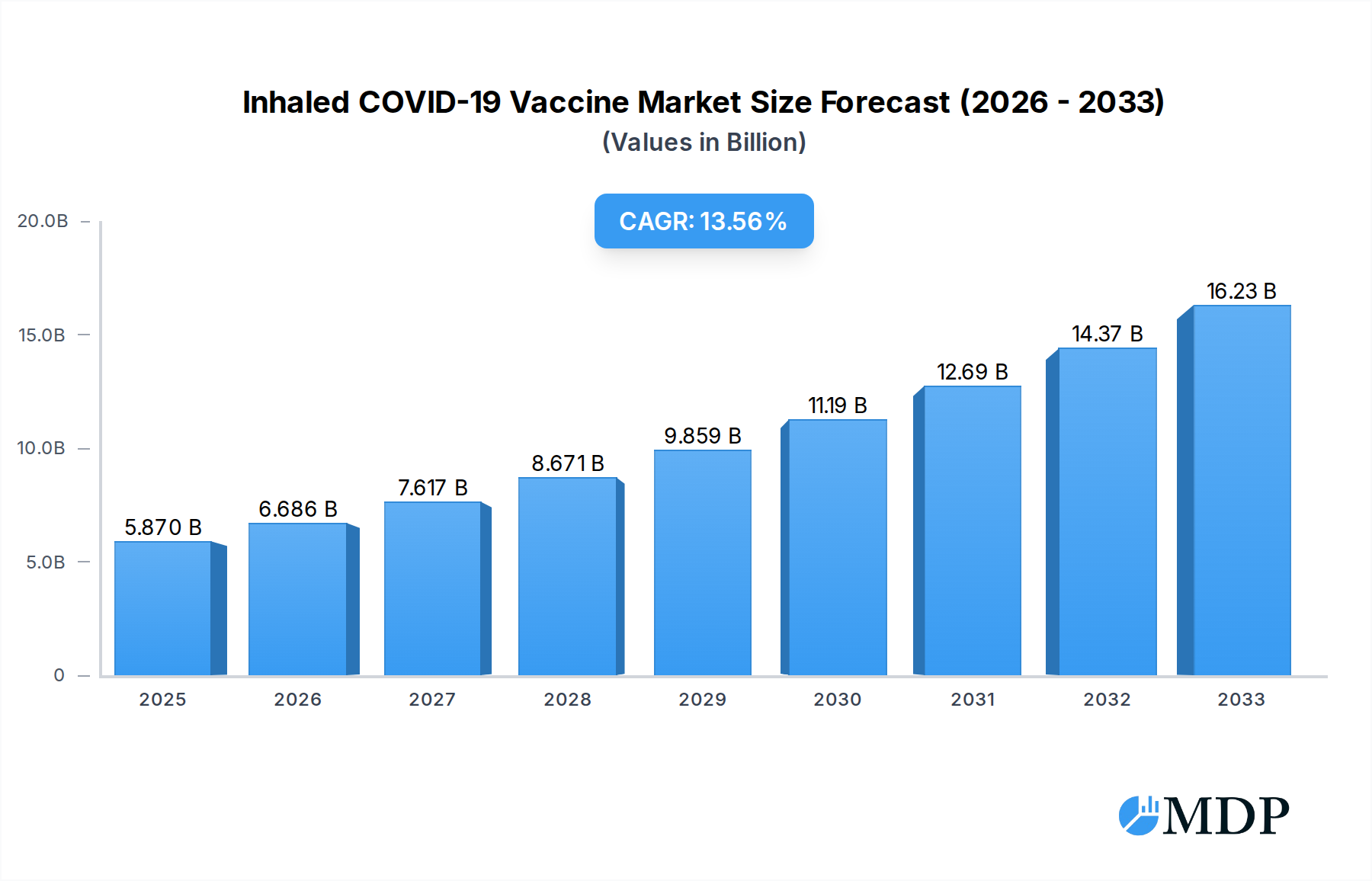
Inhaled COVID-19 Vaccine Market Size (In Billion)

The market's trajectory is also shaped by emerging trends such as the development of multi-valent inhaled vaccines capable of protecting against multiple respiratory pathogens simultaneously, thereby increasing their appeal and utility. The focus on cost-effectiveness and efficient delivery systems will be crucial for widespread adoption, especially in resource-limited regions. While the market exhibits strong growth potential, certain restraints need to be addressed. These include regulatory hurdles for novel delivery methods, the need for extensive clinical trials to establish non-inferiority and safety profiles compared to traditional injectable vaccines, and public perception and acceptance of inhaled vaccine technologies. Overcoming these challenges through robust scientific validation and effective public health communication will be paramount for realizing the full potential of this burgeoning market. The geographical distribution indicates a strong presence and anticipated growth in regions like North America and Europe, driven by advanced healthcare infrastructure and significant R&D investments, with Asia Pacific emerging as a key growth engine due to its large population and increasing focus on public health solutions.
Inhaled COVID-19 Vaccine Company Market Share

Inhaled COVID-19 Vaccine Market Report: Strategic Insights and Future Outlook (2019-2033)
This comprehensive report delves into the rapidly evolving Inhaled COVID-19 Vaccine market, providing in-depth analysis and actionable intelligence for industry stakeholders. With a study period spanning from 2019 to 2033, including a base year of 2025 and a forecast period of 2025–2033, this report offers a granular view of market dynamics, key trends, leading segments, and strategic opportunities. The analysis covers historical performance from 2019 to 2024, highlighting pivotal developments that have shaped the current landscape. Leveraging high-traffic keywords and a structured format, this report aims to maximize search visibility and attract decision-makers across the global healthcare and pharmaceutical sectors. We project the global inhaled COVID-19 vaccine market to reach a value of $50 billion by 2033, with a Compound Annual Growth Rate (CAGR) of 15.5% during the forecast period.
Inhaled COVID-19 Vaccine Market Dynamics & Concentration
The Inhaled COVID-19 Vaccine market, estimated at $20 billion in 2025, is characterized by a dynamic interplay of innovation, evolving regulatory landscapes, and shifting end-user preferences. Market concentration is moderate, with several key players vying for market share. Innovation drivers are primarily focused on improving vaccine efficacy, reducing side effects, and enhancing ease of administration. Regulatory frameworks, while stringent, are adapting to facilitate faster approval pathways for novel vaccine technologies, spurred by ongoing public health concerns. Product substitutes, though present in traditional injectable vaccines, are being challenged by the unique advantages offered by inhaled delivery. End-user trends indicate a growing preference for needle-free administration, particularly among pediatric populations and individuals with needle phobia, a segment representing an estimated 30% of potential users. Mergers and acquisitions (M&A) activity is anticipated to increase, with an estimated 5 significant deals projected between 2025 and 2033, aiming to consolidate market share and accelerate product development. The market share of inhaled vaccines is projected to grow from 5% in 2025 to 25% by 2033.
Inhaled COVID-19 Vaccine Industry Trends & Analysis
The Inhaled COVID-19 Vaccine industry is poised for substantial growth, driven by a confluence of factors including increasing demand for advanced vaccination methods, robust research and development initiatives, and a growing recognition of the advantages of non-invasive drug delivery. The market penetration of inhaled vaccines, currently at a nascent stage, is projected to skyrocket. Key growth drivers include the inherent advantages of inhaled vaccines, such as rapid systemic and mucosal immunity induction, reduced risk of needle-stick injuries, and potential for self-administration, thereby improving vaccine accessibility and uptake, especially in remote or underserved regions. Technological disruptions are at the forefront, with advancements in nebulizer technology, nanoparticle delivery systems, and mRNA and viral vector platforms enabling more efficient and targeted vaccine delivery. Consumer preferences are increasingly leaning towards comfort and convenience, making inhaled options highly attractive. Competitive dynamics are intensifying as pharmaceutical giants and innovative biotech firms invest heavily in clinical trials and manufacturing capabilities. The market is projected to grow at a CAGR of 15.5%, reaching an estimated $50 billion by 2033. We anticipate that by 2033, the market penetration for inhaled COVID-19 vaccines will reach 25% of the total COVID-19 vaccine market. The development of single-dose inhaled vaccines for booster shots is also a significant trend, further enhancing their appeal.
Leading Markets & Segments in Inhaled COVID-19 Vaccine
North America is expected to emerge as the dominant region in the Inhaled COVID-19 Vaccine market, driven by robust government funding for R&D, a high prevalence of respiratory diseases, and a well-established healthcare infrastructure that readily adopts new medical technologies. The United States, in particular, leads due to significant investments in biopharmaceutical innovation and a proactive regulatory environment.
Within the Application segment, the Medical application is anticipated to dominate, accounting for an estimated 85% of the market share by 2033. This is attributed to the widespread need for effective COVID-19 vaccination strategies in healthcare settings, including hospitals, clinics, and public health initiatives.
Analyzing the Types segmentation, the 30 Years Old and Above demographic is projected to be the largest consumer base, representing approximately 70% of the market. This segment includes a higher proportion of individuals at risk for severe COVID-19 outcomes, making them prime candidates for advanced vaccine technologies.
Key Drivers for North American Dominance:
- Economic Policies: Favorable government grants and tax incentives for biopharmaceutical research and development.
- Infrastructure: Advanced healthcare systems with widespread accessibility to vaccination centers and advanced medical equipment.
- Technological Adoption: A receptive market for cutting-edge medical technologies and needle-free delivery systems.
- Regulatory Support: Streamlined approval processes for innovative vaccines and therapeutics.
Dominance Analysis for Medical Application: The continuous need for widespread vaccination campaigns, booster shots, and potential future variants necessitates scalable and efficient delivery methods. Inhaled vaccines offer a significant advantage in mass vaccination drives and in settings where patient compliance with traditional injections may be a challenge.
Dominance Analysis for 30 Years Old and Above Segment: This demographic often exhibits a greater awareness of health risks and a stronger desire for preventative measures. The convenience and reduced anxiety associated with inhaled vaccines make them particularly appealing to this broad age group, including elderly populations and those with pre-existing health conditions. The 18-30 Years Old segment is expected to contribute 30% of the market share, driven by a preference for modern and less intrusive healthcare solutions.
Inhaled COVID-19 Vaccine Product Developments
Product developments in the Inhaled COVID-19 Vaccine sector are rapidly advancing, focusing on optimizing delivery mechanisms and enhancing immunogenicity. Innovations include the development of novel aerosol formulations designed for efficient deposition in the respiratory tract and improved stability. Companies are leveraging advanced platform technologies like mRNA and viral vectors to create highly effective inhaled vaccines. The competitive advantage lies in the potential for mucosal immunity at the point of entry of the virus, offering a potentially superior protective barrier. These advancements aim to address unmet needs for convenient, needle-free vaccination options, positioning inhaled vaccines as a significant future component of global public health strategies.
Key Drivers of Inhaled COVID-19 Vaccine Growth
The growth of the Inhaled COVID-19 Vaccine market is propelled by several key factors. Technological advancements in drug delivery systems, particularly in nebulization and aerosolization, are making inhaled vaccines more efficient and viable. The global demand for convenient and needle-free vaccination alternatives, driven by patient preference and the desire to overcome injection-related anxieties, is a significant catalyst. Furthermore, the ongoing need for adaptable vaccine solutions to combat emerging COVID-19 variants and the potential for inhaled vaccines to induce superior mucosal immunity are crucial growth drivers. Favorable regulatory pathways and increased government funding for research and development further support market expansion.
Challenges in the Inhaled COVID-19 Vaccine Market
Despite its promising outlook, the Inhaled COVID-19 Vaccine market faces several challenges. Regulatory hurdles, though easing, still require extensive clinical validation to ensure safety and efficacy, which can be time-consuming and costly. Establishing robust supply chains for specialized nebulizer devices and ensuring equitable distribution globally present significant logistical challenges. Competitive pressures from established injectable vaccine manufacturers and the need for extensive public education campaigns to promote adoption are also key restraints. The estimated cost of developing and scaling up manufacturing for inhaled vaccines could be 10-15% higher than traditional vaccines initially.
Emerging Opportunities in Inhaled COVID-19 Vaccine
Emerging opportunities in the Inhaled COVID-19 Vaccine market are vast and diverse. Technological breakthroughs in nanoparticle delivery and formulation science are paving the way for more potent and stable inhaled vaccines. Strategic partnerships between pharmaceutical companies and device manufacturers are crucial for developing integrated delivery solutions. Market expansion into developing economies, where healthcare infrastructure may be less developed, presents a significant opportunity, as inhaled vaccines could bypass the need for cold-chain refrigeration and skilled needle-handling personnel. The development of multi-purpose inhaled vaccines targeting other respiratory pathogens alongside COVID-19 also represents a substantial growth avenue.
Leading Players in the Inhaled COVID-19 Vaccine Sector
- CanSino Biologics
- Pfizer Inc.
- Moderna, Inc.
- AstraZeneca PLC
- Johnson & Johnson
Key Milestones in Inhaled COVID-19 Vaccine Industry
- 2020 August: Initiation of early-stage clinical trials for inhaled COVID-19 vaccine candidates.
- 2021 Q2: Announcement of promising preclinical data for several inhaled vaccine platforms.
- 2022 Q4: Commencement of Phase 1/2 clinical trials for CanSino Biologics' inhaled COVID-19 vaccine.
- 2023 Q3: Publication of research detailing the potential for enhanced mucosal immunity with inhaled vaccines.
- 2024 Q1: Expansion of clinical trials for inhaled COVID-19 vaccines to larger patient cohorts across multiple regions.
Strategic Outlook for Inhaled COVID-19 Vaccine Market
The strategic outlook for the Inhaled COVID-19 Vaccine market is highly optimistic, fueled by ongoing innovation and a clear demand for advanced vaccination technologies. Future market growth will be accelerated by the continued development of needle-free delivery systems, the expansion of vaccine indications beyond primary immunization to include booster doses and broad-spectrum respiratory pathogen protection, and strategic collaborations to enhance manufacturing capacity and global distribution networks. The increasing focus on patient convenience and public health preparedness will further solidify the position of inhaled COVID-19 vaccines as a critical component of future pandemic response strategies, projecting sustained high growth and significant market value.
Inhaled COVID-19 Vaccine Segmentation
-
1. Application
- 1.1. Medical
- 1.2. Research
-
2. Types
- 2.1. 18-30 Years Old
- 2.2. 30 Years Old and Above
Inhaled COVID-19 Vaccine Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
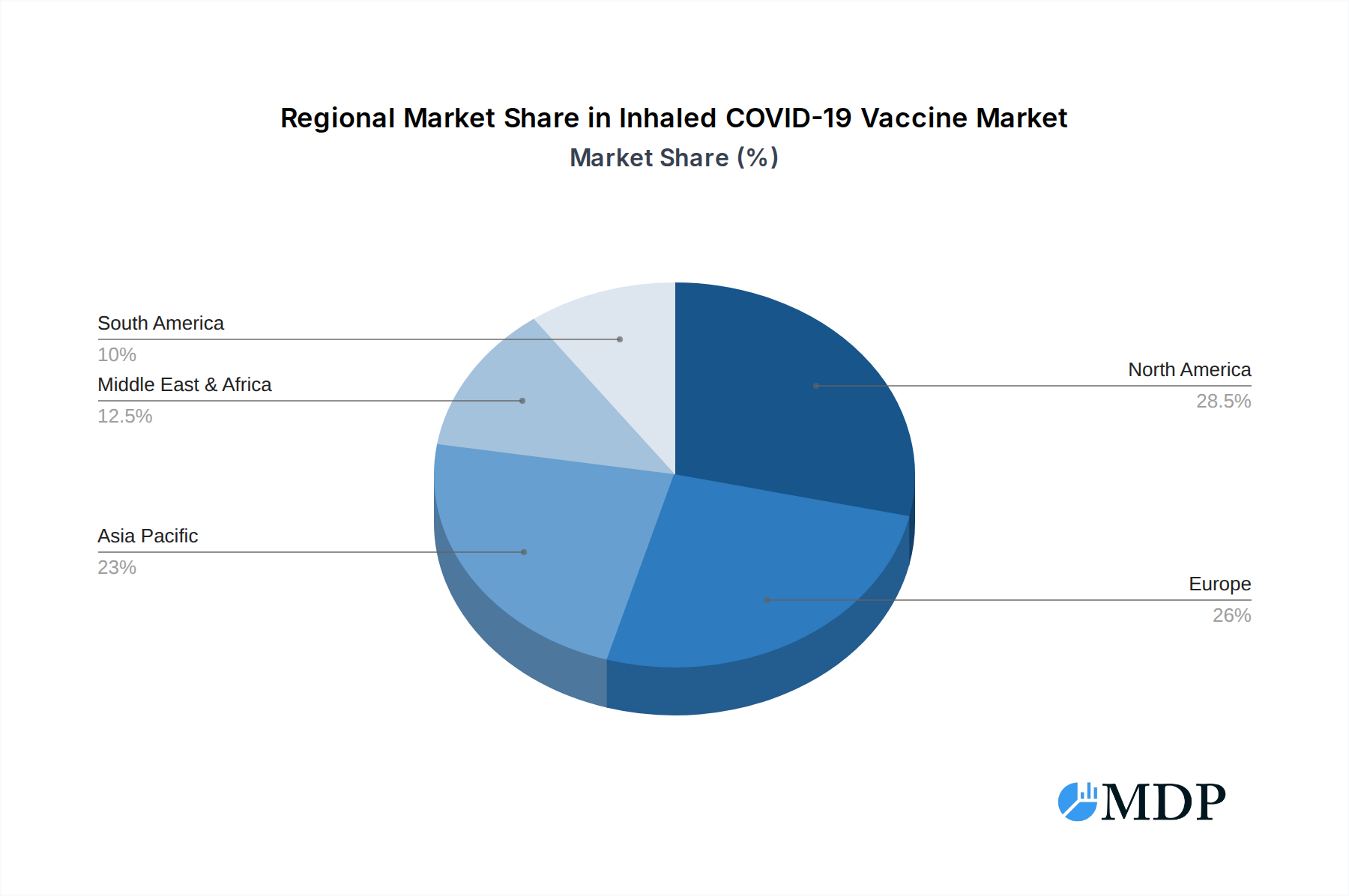
Inhaled COVID-19 Vaccine Regional Market Share

Geographic Coverage of Inhaled COVID-19 Vaccine
Inhaled COVID-19 Vaccine REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 13.96% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Inhaled COVID-19 Vaccine Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Medical
- 5.1.2. Research
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. 18-30 Years Old
- 5.2.2. 30 Years Old and Above
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Inhaled COVID-19 Vaccine Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Medical
- 6.1.2. Research
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. 18-30 Years Old
- 6.2.2. 30 Years Old and Above
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Inhaled COVID-19 Vaccine Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Medical
- 7.1.2. Research
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. 18-30 Years Old
- 7.2.2. 30 Years Old and Above
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Inhaled COVID-19 Vaccine Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Medical
- 8.1.2. Research
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. 18-30 Years Old
- 8.2.2. 30 Years Old and Above
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Inhaled COVID-19 Vaccine Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Medical
- 9.1.2. Research
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. 18-30 Years Old
- 9.2.2. 30 Years Old and Above
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Inhaled COVID-19 Vaccine Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Medical
- 10.1.2. Research
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. 18-30 Years Old
- 10.2.2. 30 Years Old and Above
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1. CanSino Biologics
List of Figures
- Figure 1: Global Inhaled COVID-19 Vaccine Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Inhaled COVID-19 Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Inhaled COVID-19 Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Inhaled COVID-19 Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Inhaled COVID-19 Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Inhaled COVID-19 Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Inhaled COVID-19 Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Inhaled COVID-19 Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Inhaled COVID-19 Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Inhaled COVID-19 Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Inhaled COVID-19 Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Inhaled COVID-19 Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Inhaled COVID-19 Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Inhaled COVID-19 Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Inhaled COVID-19 Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Inhaled COVID-19 Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Inhaled COVID-19 Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Inhaled COVID-19 Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Inhaled COVID-19 Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Inhaled COVID-19 Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Inhaled COVID-19 Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Inhaled COVID-19 Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Inhaled COVID-19 Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Inhaled COVID-19 Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Inhaled COVID-19 Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Inhaled COVID-19 Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Inhaled COVID-19 Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Inhaled COVID-19 Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Inhaled COVID-19 Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Inhaled COVID-19 Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Inhaled COVID-19 Vaccine Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Inhaled COVID-19 Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Inhaled COVID-19 Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Inhaled COVID-19 Vaccine?
The projected CAGR is approximately 13.96%.
2. Which companies are prominent players in the Inhaled COVID-19 Vaccine?
Key companies in the market include CanSino Biologics.
3. What are the main segments of the Inhaled COVID-19 Vaccine?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Inhaled COVID-19 Vaccine," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Inhaled COVID-19 Vaccine report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Inhaled COVID-19 Vaccine?
To stay informed about further developments, trends, and reports in the Inhaled COVID-19 Vaccine, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


