Key Insights
The global General Influenza Diagnostics market is poised for robust expansion, projected to reach $11.24 billion by 2025, with an impressive Compound Annual Growth Rate (CAGR) of 11.37%. This significant growth is underpinned by several key drivers, including the increasing prevalence of influenza outbreaks, a growing awareness of the importance of early and accurate diagnosis for effective treatment and public health management, and advancements in diagnostic technologies. The market is segmented by type into rapid influenza diagnostic tests (RIDTs), viral culture, direct fluorescent antibody (DFA) assays, and serological assays, with RIDTs and advanced molecular diagnostic methods likely to see substantial adoption due to their speed and accuracy. By application, hospitals and clinical laboratories represent the largest end-user segments, driven by the need for immediate patient management and public health surveillance.
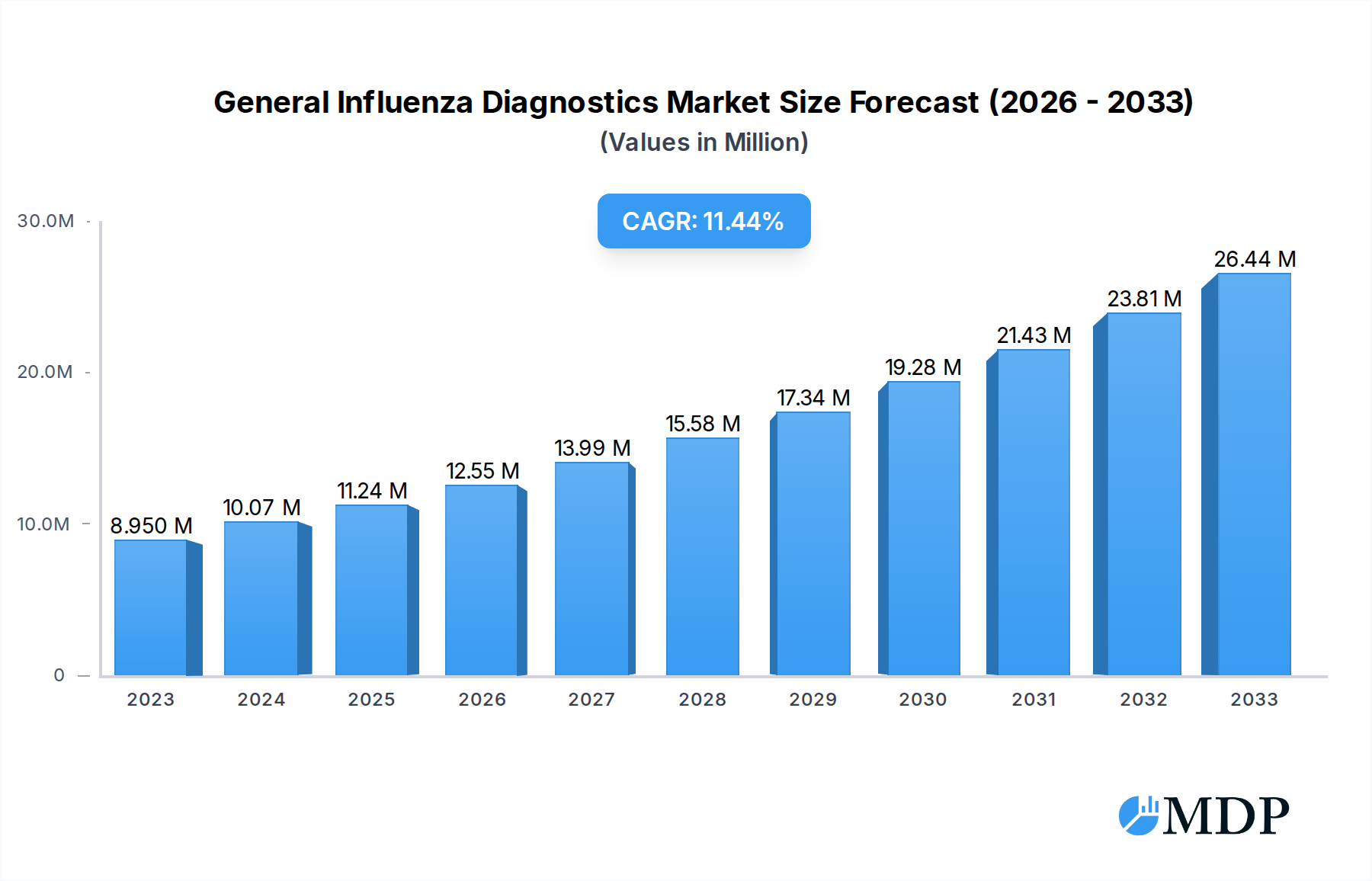
General Influenza Diagnostics Market Size (In Million)

The market's trajectory is further shaped by evolving trends such as the development of multiplex assays capable of detecting multiple respiratory pathogens simultaneously, including influenza and other viruses like RSV and COVID-19, enhancing diagnostic efficiency. The growing demand for point-of-care testing (POCT) solutions is also a significant trend, enabling faster diagnoses in diverse settings. While the market is largely driven by these positive factors, potential restraints include the cost of advanced diagnostic equipment, reimbursement policies, and the availability of skilled personnel to operate sophisticated instruments. Geographically, North America and Europe are expected to lead the market in the near term, owing to well-established healthcare infrastructures and high adoption rates of advanced diagnostics. However, the Asia Pacific region is anticipated to exhibit the highest growth rate, fueled by a large population, increasing healthcare expenditure, and a rising incidence of infectious diseases. Leading companies such as F. Hoffmann-La Roche, Quidel Corporation, Thermo Fisher Scientific, and Abbott Laboratories are actively investing in research and development to introduce innovative solutions and expand their market presence.
General Influenza Diagnostics Company Market Share

General Influenza Diagnostics Market Report: Comprehensive Analysis & Future Outlook (2019-2033)
Unlock critical insights into the global General Influenza Diagnostics market with this in-depth report. Covering the historical period from 2019 to 2024 and projecting robust growth through 2033, this study provides a definitive roadmap for stakeholders, investors, and industry professionals. Delve into market dynamics, technological advancements, regional dominance, and emerging opportunities within this vital sector. The report leverages high-traffic keywords for maximum visibility, ensuring you access the most relevant and actionable information.
General Influenza Diagnostics Market Dynamics & Concentration
The General Influenza Diagnostics market is characterized by a dynamic interplay of innovation, regulatory oversight, and evolving end-user needs. Market concentration remains moderately fragmented, with a few dominant players like F. Hoffmann-La Roche, Quidel Corporation, Thermo Fisher Scientific, Abbott Laboratories, and Becton, Dickinson, and Company holding significant market share, estimated to be in the billions. Innovation drivers are primarily fueled by the continuous need for faster, more accurate, and point-of-care diagnostic solutions, especially in response to pandemic preparedness. Regulatory frameworks, governed by bodies like the FDA and EMA, play a crucial role in ensuring product safety and efficacy, influencing product approvals and market entry strategies. Product substitutes, while present, are often less sensitive or specific than dedicated influenza diagnostic tests, limiting their impact. End-user trends indicate a growing preference for multiplex assays capable of detecting multiple respiratory pathogens simultaneously, alongside demand for home-use and rapid diagnostic tests. Mergers and Acquisitions (M&A) activities, with an estimated xx billion in deal value and xx number of deals recorded historically, are crucial for market consolidation, technology acquisition, and expanding geographical reach.
General Influenza Diagnostics Industry Trends & Analysis
The General Influenza Diagnostics industry is poised for significant expansion, driven by an escalating global incidence of influenza, heightened awareness of public health threats, and substantial investments in healthcare infrastructure. The Compound Annual Growth Rate (CAGR) is projected to be approximately X.XX% during the forecast period of 2025–2033, contributing to an estimated market valuation of over one hundred billion by 2033. Technological disruptions are at the forefront, with advancements in molecular diagnostics, such as Real-Time Polymerase Chain Reaction (RT-PCR), offering superior sensitivity and specificity compared to traditional methods. The increasing adoption of Rapid Influenza Diagnostic Tests (RIDTs) at point-of-care settings, including clinics and physician offices, is a key trend, enhancing accessibility and enabling timely treatment decisions. Consumer preferences are shifting towards user-friendly, cost-effective, and integrated diagnostic solutions that can provide rapid results. The competitive landscape is intense, with leading companies actively engaged in research and development to launch next-generation diagnostic platforms and expand their product portfolios. Market penetration is steadily increasing across both developed and emerging economies, fueled by government initiatives promoting early disease detection and the growing burden of influenza-related illnesses, estimated to impact billions of individuals annually. The demand for accurate diagnostics is also influenced by seasonal influenza outbreaks and the potential for novel influenza strains, necessitating robust and responsive diagnostic capabilities. The integration of artificial intelligence (AI) and machine learning (ML) into diagnostic workflows is also emerging as a trend, promising enhanced data analysis and predictive capabilities.
Leading Markets & Segments in General Influenza Diagnostics
The General Influenza Diagnostics market exhibits distinct regional and segmental dominance. North America currently leads the market, driven by a well-established healthcare system, high adoption rates of advanced diagnostic technologies, and significant government investment in public health initiatives. The United States, in particular, represents a substantial market due to its large population and robust research and development landscape, with an estimated market share in the billions.
Dominant Application Segment:
- Hospitals: Hospitals remain the largest application segment, owing to the critical need for rapid and accurate influenza diagnosis in inpatient settings for effective patient management and outbreak control. The higher volume of patient admissions and the severity of influenza cases in hospital environments necessitate sophisticated diagnostic tools.
- Clinical Laboratories: Clinical laboratories represent another significant segment, benefiting from the increasing outsourcing of diagnostic testing by healthcare providers and the demand for high-throughput testing capabilities. These labs are crucial for comprehensive disease surveillance and research.
- Other End-Users: This segment, encompassing retail clinics, community health centers, and occupational health facilities, is experiencing steady growth as the demand for accessible point-of-care testing rises.
Dominant Type Segment:
- RIDT (Rapid Influenza Diagnostic Tests): RIDTs hold a dominant position due to their speed, ease of use, and cost-effectiveness, making them ideal for point-of-care settings and initial screening. Their widespread availability and ability to deliver results within minutes are key drivers of their market leadership.
- Viral Culture: While considered a gold standard for sensitivity, viral culture methods are less prevalent in routine diagnostics due to their longer turnaround times and requirement for specialized laboratory infrastructure. They are primarily used for research and confirmation of complex cases.
- DFA (Direct Fluorescent Antibody): DFA offers a balance between speed and sensitivity, making it a valuable tool, particularly in hospital settings and during peak influenza season. Its ability to detect viral antigens directly from respiratory specimens contributes to its utility.
- Serological Assays: Serological assays are primarily used for retrospective diagnosis and epidemiological studies, identifying past exposure to influenza viruses. Their role in acute diagnosis is limited but crucial for understanding population immunity and viral evolution.
Economic policies encouraging widespread testing and public health infrastructure development are key drivers of this dominance. The increasing prevalence of influenza and the need for timely interventions further solidify the market position of these leading segments.
General Influenza Diagnostics Product Developments
Product developments in General Influenza Diagnostics are focused on enhancing speed, accuracy, and multiplexing capabilities. Innovations are leading to point-of-care tests that can simultaneously detect multiple respiratory pathogens, including various influenza strains and other common viruses like RSV and COVID-19. Companies are investing in technologies that improve sensitivity, reduce false negatives, and offer quantitative results, crucial for patient management. The development of user-friendly interfaces and cloud-connectivity for data management are also key trends, facilitating efficient workflow in hospitals and clinical laboratories. These advancements aim to provide clinicians with actionable insights rapidly, improving patient outcomes and reducing the spread of infection.
Key Drivers of General Influenza Diagnostics Growth
The General Influenza Diagnostics market is propelled by several key drivers. Technological advancements in molecular diagnostics, such as PCR-based assays and next-generation sequencing, are enabling faster and more accurate detection. Increasing global incidence of influenza and heightened awareness of pandemic preparedness necessitate robust diagnostic capabilities. Government initiatives and funding for public health surveillance and disease control programs also play a crucial role. Furthermore, the growing demand for point-of-care diagnostics driven by convenience and the need for rapid treatment decisions is a significant growth catalyst. The aging global population and the increased susceptibility of this demographic to severe influenza complications further contribute to market expansion.
Challenges in the General Influenza Diagnostics Market
Despite robust growth, the General Influenza Diagnostics market faces several challenges. Stringent regulatory hurdles for new product approvals can lead to extended market entry timelines and significant development costs. Reimbursement policies for diagnostic tests can also impact market adoption, especially in resource-constrained regions. High initial investment costs for advanced molecular diagnostic platforms can be a barrier for smaller laboratories. Supply chain disruptions for critical reagents and components can affect manufacturing and availability. Competition from established players and emerging innovators exerts pressure on pricing and market share. The potential for antimicrobial resistance and emergence of novel influenza strains also necessitates continuous innovation and adaptation, posing an ongoing challenge for diagnostic manufacturers.
Emerging Opportunities in General Influenza Diagnostics
Emerging opportunities in the General Influenza Diagnostics market are centered around technological breakthroughs and strategic market expansion. The development of highly sensitive and specific multiplex assays capable of detecting a broad range of respiratory pathogens in a single test presents a significant opportunity for improved clinical management and reduced diagnostic complexity. The increasing adoption of at-home and self-testing solutions driven by consumer demand for convenience and accessibility opens new market avenues. Strategic partnerships between diagnostic companies, pharmaceutical firms, and public health organizations can accelerate the development and deployment of innovative diagnostic tools, especially in response to emerging infectious threats. Furthermore, expansion into emerging economies with growing healthcare infrastructure and increasing demand for diagnostic services offers substantial long-term growth potential. The integration of digital health technologies and AI-powered analytics for real-time disease surveillance and predictive diagnostics represents another promising area for future development.
Leading Players in the General Influenza Diagnostics Sector
- F. Hoffmann-La Roche
- Quidel Corporation
- Thermo Fisher Scientific
- Abbott Laboratories
- Becton, Dickinson, and Company
- DiaSorin
- bioMérieux
Key Milestones in General Influenza Diagnostics Industry
- 2019 October: Launch of a novel multiplex PCR assay for simultaneous detection of influenza A, B, and other respiratory viruses.
- 2020 March: Significant surge in demand and development of rapid diagnostic tests for emerging viral threats, including influenza.
- 2021 February: Major acquisition of a molecular diagnostics company by a leading player to expand its influenza testing portfolio.
- 2022 April: FDA approval for a new point-of-care RIDT with enhanced sensitivity and specificity.
- 2023 January: Introduction of a cloud-connected diagnostic platform enabling remote monitoring and data analysis for influenza surveillance.
- 2023 September: Major research publication detailing advancements in universal influenza vaccine development, indirectly impacting diagnostic needs.
- 2024 May: Strategic collaboration announced for the development of advanced AI algorithms for influenza outbreak prediction.
Strategic Outlook for General Influenza Diagnostics Market
The strategic outlook for the General Influenza Diagnostics market is highly positive, driven by sustained demand for accurate and rapid diagnostic solutions. Growth accelerators include the continuous evolution of molecular diagnostic technologies, leading to more sensitive and multiplexed testing capabilities. The increasing emphasis on pandemic preparedness and response will further fuel investment in influenza diagnostics. Strategic opportunities lie in expanding product portfolios to include a wider range of respiratory pathogens, developing user-friendly point-of-care and at-home testing options, and forging collaborations to enhance global accessibility. The integration of digital health solutions and AI for improved data analysis and predictive diagnostics will also be crucial for future market leadership. Companies that focus on innovation, regulatory compliance, and strategic market penetration are well-positioned to capitalize on the robust growth trajectory of this vital sector.
General Influenza Diagnostics Segmentation
-
1. Application
- 1.1. Hospitals
- 1.2. Clinical Laboratories
- 1.3. Other End-User
-
2. Type
- 2.1. RIDT
- 2.2. Viral Culture
- 2.3. DFA
- 2.4. Serological Assays
General Influenza Diagnostics Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
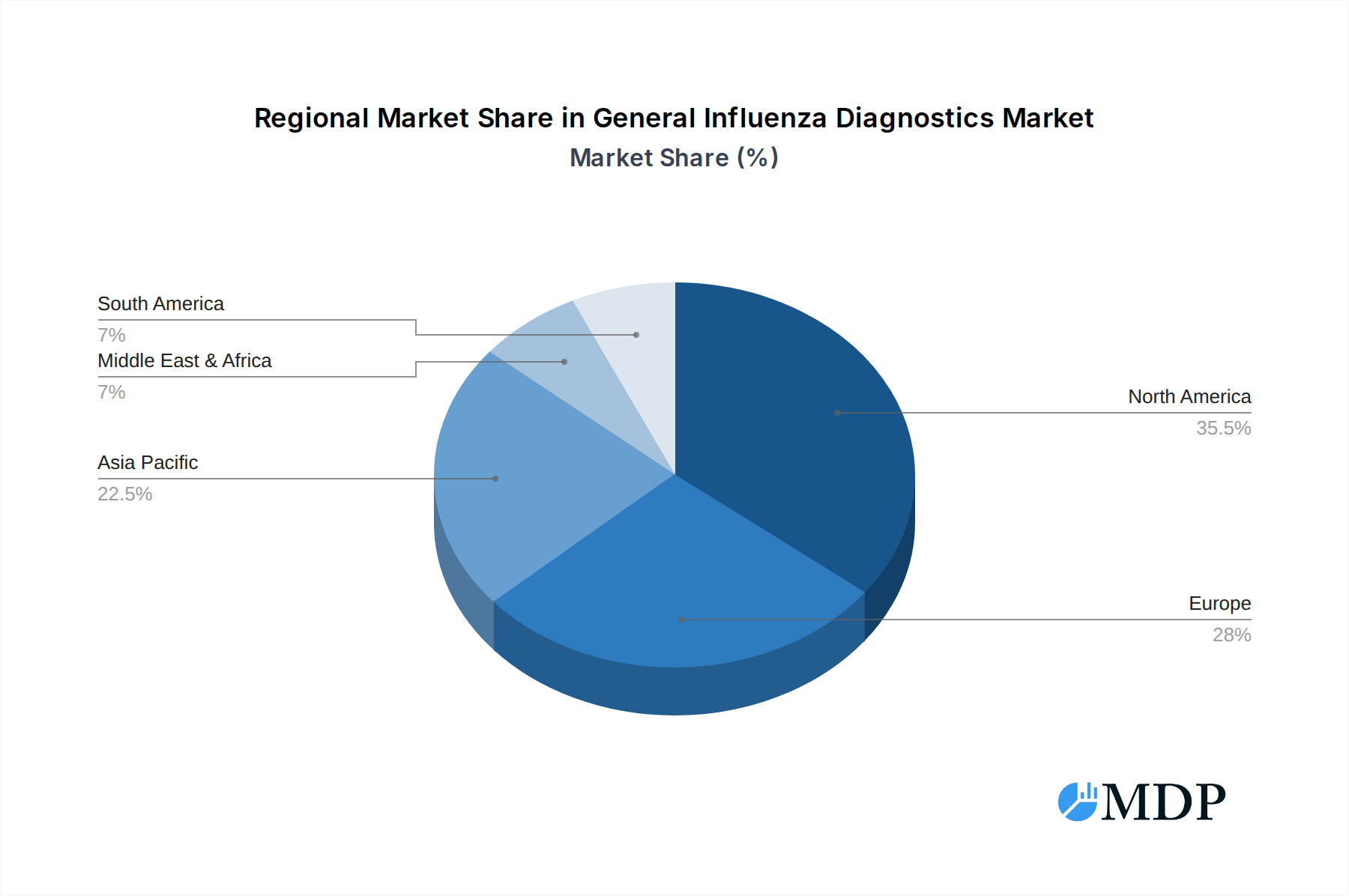
General Influenza Diagnostics Regional Market Share

Geographic Coverage of General Influenza Diagnostics
General Influenza Diagnostics REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 11.37% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global General Influenza Diagnostics Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals
- 5.1.2. Clinical Laboratories
- 5.1.3. Other End-User
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. RIDT
- 5.2.2. Viral Culture
- 5.2.3. DFA
- 5.2.4. Serological Assays
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America General Influenza Diagnostics Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospitals
- 6.1.2. Clinical Laboratories
- 6.1.3. Other End-User
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. RIDT
- 6.2.2. Viral Culture
- 6.2.3. DFA
- 6.2.4. Serological Assays
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America General Influenza Diagnostics Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospitals
- 7.1.2. Clinical Laboratories
- 7.1.3. Other End-User
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. RIDT
- 7.2.2. Viral Culture
- 7.2.3. DFA
- 7.2.4. Serological Assays
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe General Influenza Diagnostics Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospitals
- 8.1.2. Clinical Laboratories
- 8.1.3. Other End-User
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. RIDT
- 8.2.2. Viral Culture
- 8.2.3. DFA
- 8.2.4. Serological Assays
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa General Influenza Diagnostics Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospitals
- 9.1.2. Clinical Laboratories
- 9.1.3. Other End-User
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. RIDT
- 9.2.2. Viral Culture
- 9.2.3. DFA
- 9.2.4. Serological Assays
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific General Influenza Diagnostics Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospitals
- 10.1.2. Clinical Laboratories
- 10.1.3. Other End-User
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. RIDT
- 10.2.2. Viral Culture
- 10.2.3. DFA
- 10.2.4. Serological Assays
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 F. Hoffmann-La Roche
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Quidel Corporation
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Thermo Fisher Scientific
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Abbott Laboratories
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Becton Dickinson
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 and Company
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 DiaSorin
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 bioMérieux
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.1 F. Hoffmann-La Roche
List of Figures
- Figure 1: Global General Influenza Diagnostics Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America General Influenza Diagnostics Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America General Influenza Diagnostics Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America General Influenza Diagnostics Revenue (undefined), by Type 2025 & 2033
- Figure 5: North America General Influenza Diagnostics Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America General Influenza Diagnostics Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America General Influenza Diagnostics Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America General Influenza Diagnostics Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America General Influenza Diagnostics Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America General Influenza Diagnostics Revenue (undefined), by Type 2025 & 2033
- Figure 11: South America General Influenza Diagnostics Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America General Influenza Diagnostics Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America General Influenza Diagnostics Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe General Influenza Diagnostics Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe General Influenza Diagnostics Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe General Influenza Diagnostics Revenue (undefined), by Type 2025 & 2033
- Figure 17: Europe General Influenza Diagnostics Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe General Influenza Diagnostics Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe General Influenza Diagnostics Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa General Influenza Diagnostics Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa General Influenza Diagnostics Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa General Influenza Diagnostics Revenue (undefined), by Type 2025 & 2033
- Figure 23: Middle East & Africa General Influenza Diagnostics Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa General Influenza Diagnostics Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa General Influenza Diagnostics Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific General Influenza Diagnostics Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific General Influenza Diagnostics Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific General Influenza Diagnostics Revenue (undefined), by Type 2025 & 2033
- Figure 29: Asia Pacific General Influenza Diagnostics Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific General Influenza Diagnostics Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific General Influenza Diagnostics Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global General Influenza Diagnostics Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global General Influenza Diagnostics Revenue undefined Forecast, by Type 2020 & 2033
- Table 3: Global General Influenza Diagnostics Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global General Influenza Diagnostics Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global General Influenza Diagnostics Revenue undefined Forecast, by Type 2020 & 2033
- Table 6: Global General Influenza Diagnostics Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global General Influenza Diagnostics Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global General Influenza Diagnostics Revenue undefined Forecast, by Type 2020 & 2033
- Table 12: Global General Influenza Diagnostics Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global General Influenza Diagnostics Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global General Influenza Diagnostics Revenue undefined Forecast, by Type 2020 & 2033
- Table 18: Global General Influenza Diagnostics Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global General Influenza Diagnostics Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global General Influenza Diagnostics Revenue undefined Forecast, by Type 2020 & 2033
- Table 30: Global General Influenza Diagnostics Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global General Influenza Diagnostics Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global General Influenza Diagnostics Revenue undefined Forecast, by Type 2020 & 2033
- Table 39: Global General Influenza Diagnostics Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific General Influenza Diagnostics Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the General Influenza Diagnostics?
The projected CAGR is approximately 11.37%.
2. Which companies are prominent players in the General Influenza Diagnostics?
Key companies in the market include F. Hoffmann-La Roche, Quidel Corporation, Thermo Fisher Scientific, Abbott Laboratories, Becton, Dickinson, and Company, DiaSorin, bioMérieux.
3. What are the main segments of the General Influenza Diagnostics?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "General Influenza Diagnostics," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the General Influenza Diagnostics report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the General Influenza Diagnostics?
To stay informed about further developments, trends, and reports in the General Influenza Diagnostics, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


