Key Insights
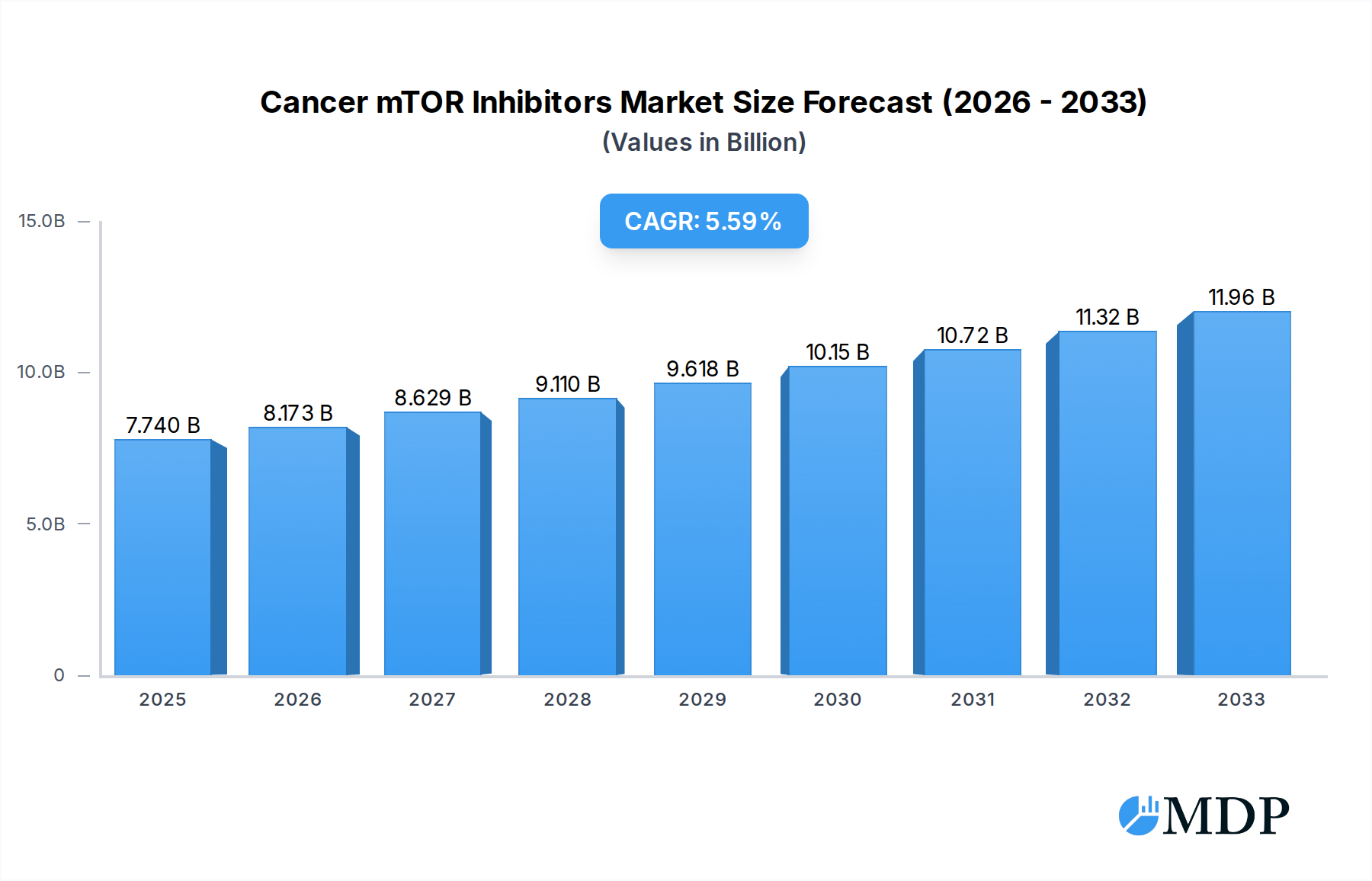
The global Cancer mTOR Inhibitors market is poised for significant expansion, with an estimated market size of $7.74 billion in 2025, driven by a robust compound annual growth rate (CAGR) of 5.5%. This growth is underpinned by several key factors, including advancements in understanding the mechanistic target of rapamycin (mTOR) pathway's role in various cancers and the subsequent development of novel inhibitor therapies. The increasing incidence of prevalent cancers such as breast cancer and hematological malignancies, coupled with a growing focus on precision medicine and personalized treatment approaches, are major catalysts. Furthermore, the approval and expanding use of approved mTOR inhibitors like Afinitor and Torisel for specific indications, including rare pediatric brain tumors and neuroendocrine tumors, are contributing to market penetration. The ongoing research and development pipeline, featuring innovative molecules and combination therapies, is also expected to fuel sustained market growth throughout the forecast period.
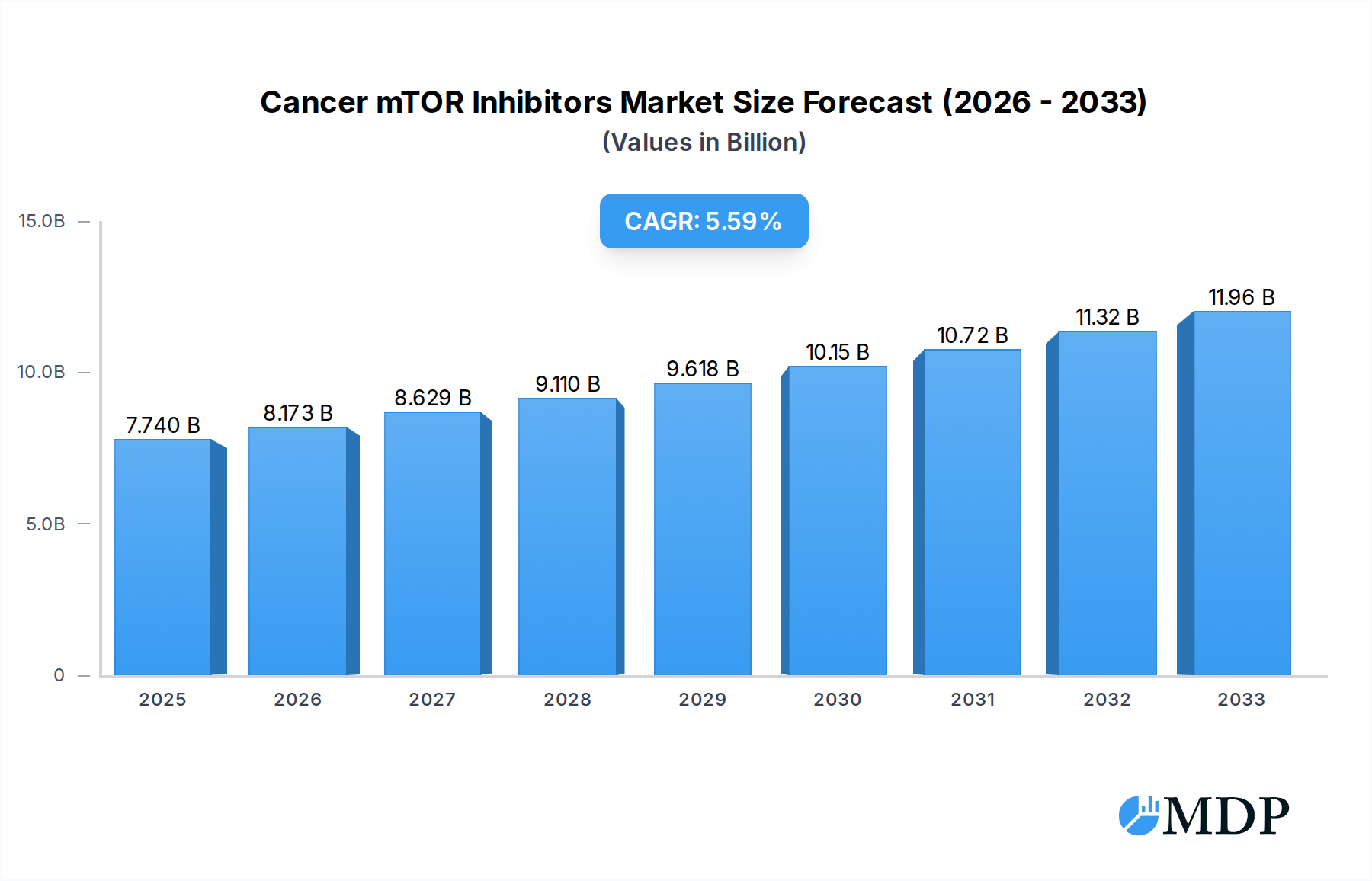
Cancer mTOR Inhibitors Market Size (In Billion)

The market is characterized by a dynamic competitive landscape with a significant number of established pharmaceutical giants and emerging biotechnology firms actively engaged in research, development, and commercialization. Key therapeutic applications include breast cancer, hematological malignancies, neuroendocrine tumors, hepatocellular carcinoma, and glioblastoma, reflecting the broad applicability of mTOR inhibition. The market is segmented by product types, with Afinitor/Votubia and Torisel (Temsirolimus) being prominent examples, alongside newer formulations and investigational compounds. While the market exhibits strong growth potential, it is not without its challenges. Restrains such as the high cost of these advanced therapies, potential for drug resistance, and stringent regulatory approval processes for new drug entities present hurdles. However, the ongoing unmet medical needs in oncology and the continuous pursuit of more effective cancer treatments by both healthcare providers and patients are expected to largely outweigh these limitations, ensuring a promising future for the Cancer mTOR Inhibitors market.
Cancer mTOR Inhibitors Company Market Share

Unlocking the Future of Oncology: A Comprehensive Report on Cancer mTOR Inhibitors (2019-2033)
This in-depth report, covering the period of 2019 through 2033, provides an unparalleled analysis of the global Cancer mTOR Inhibitors market. With a base year of 2025 and an estimated year also of 2025, this research delves into the historical trajectory (2019-2024) and projects a robust growth forecast for the period of 2025-2033. Gain critical insights into the dynamic landscape of mTOR inhibitor therapies, their applications in treating a spectrum of cancers including Breast Cancer, Hematological Malignancy, Neuroendocrine Tumors, Hepatocellular Carcinoma, and Glioblastoma, and the pivotal role of key drugs like Afinitor/Votubia, Afinitor Disperz – mTOR inhibitor for Rare Pediatric Brain Tumor, Torisel (Temsirolimus), and Evertor. This report is an indispensable resource for pharmaceutical companies, biotech innovators, healthcare providers, investors, and researchers seeking to understand and capitalize on the burgeoning Cancer mTOR Inhibitors market, projected to reach trillions in value.
Cancer mTOR Inhibitors Market Dynamics & Concentration
The Cancer mTOR Inhibitors market is characterized by a moderate level of concentration, with a few key players holding significant market share. Innovation drivers are primarily fueled by the ongoing advancements in understanding the mTOR signaling pathway's role in various cancer types and the development of next-generation inhibitors with improved efficacy and reduced side effects. Regulatory frameworks, while stringent, are evolving to accommodate novel therapies, creating opportunities for well-positioned companies. Product substitutes, though present in the broader oncology landscape, are less direct for targeted mTOR inhibition. End-user trends are increasingly shifting towards personalized medicine and combination therapies, demanding innovative drug development. Mergers and acquisitions (M&A) are a notable feature, with an estimated XX M&A deals projected within the forecast period, driven by the desire to acquire promising pipelines and expand market reach. The market share landscape is dynamic, with established players defending their positions and emerging biotechs vying for dominance, contributing to a competitive yet collaborative ecosystem focused on patient outcomes.
Cancer mTOR Inhibitors Industry Trends & Analysis
The global Cancer mTOR Inhibitors market is experiencing significant expansion, driven by a projected Compound Annual Growth Rate (CAGR) of XX% over the forecast period. This robust growth is propelled by an increasing incidence of cancer globally, coupled with a growing understanding of the mechanistic role of the mammalian target of rapamycin (mTOR) pathway in tumor proliferation, angiogenesis, and metabolism. The market penetration of mTOR inhibitors is steadily rising as clinical evidence accumulates, demonstrating their efficacy as monotherapies and in combination with other oncological agents. Technological disruptions, such as advancements in drug delivery systems and the development of highly selective mTOR inhibitors, are further enhancing treatment efficacy and patient compliance. Consumer preferences are evolving towards targeted therapies that offer improved quality of life alongside disease control. Competitive dynamics are intensifying, with pharmaceutical giants and innovative biotechnology firms investing heavily in research and development to bring novel mTOR inhibitors to market for various cancer indications. The estimated market size is anticipated to reach trillions by 2033, reflecting the immense therapeutic potential and commercial viability of these agents in addressing unmet medical needs in oncology.
Leading Markets & Segments in Cancer mTOR Inhibitors
The dominance of specific regions and segments within the Cancer mTOR Inhibitors market is a critical area of analysis. North America is a leading market, driven by a high prevalence of cancer diagnoses, advanced healthcare infrastructure, and significant investment in pharmaceutical R&D. The United States, in particular, spearheads market growth due to its robust regulatory environment and the early adoption of innovative cancer therapies.
Application Dominance:
- Breast Cancer: This segment represents a significant market share due to the high incidence of breast cancer globally and the established efficacy of mTOR inhibitors in certain subtypes, such as hormone receptor-positive, HER2-negative advanced breast cancer.
- Hematological Malignancy: The application in treating various blood cancers, including lymphomas and leukemias, also contributes substantially to market value, with ongoing research exploring novel therapeutic strategies.
- Neuroendocrine Tumors: mTOR inhibitors have demonstrated notable efficacy in this niche but growing segment, making it a key area of focus for market expansion.
- Hepatocellular Carcinoma: The increasing burden of liver cancer globally positions this application as a significant growth driver for mTOR inhibitor therapies.
- Glioblastoma: While challenging to treat, advancements in understanding tumor heterogeneity and the development of targeted agents are slowly increasing the role of mTOR inhibitors in glioblastoma treatment.
Types Dominance:
- Afinitor/Votubia (Everolimus): These branded formulations, particularly Everolimus, have historically dominated the market, benefiting from early approvals and established clinical data across multiple cancer types. Their continued use in approved indications and ongoing clinical trials ensures sustained market presence.
- Afinitor Disperz – mTOR inhibitor for Rare Pediatric Brain Tumor: This specific formulation addresses a critical unmet need in pediatric oncology, securing a specialized but vital market segment.
- Torisel (Temsirolimus): While having a strong presence in renal cell carcinoma, Temsirolimus also finds application in other indications, contributing to its market significance.
- Evertor: As a newer entrant or investigational compound, Evertor represents the ongoing innovation and potential for future market disruption.
Key drivers for segment dominance include robust clinical trial outcomes, favorable reimbursement policies, the prevalence of specific cancer subtypes within a region, and the strategic market penetration efforts of leading pharmaceutical companies.
Cancer mTOR Inhibitors Product Developments
The Cancer mTOR Inhibitors market is witnessing a wave of innovative product developments aimed at enhancing therapeutic outcomes. These advancements primarily focus on developing next-generation inhibitors with improved specificity, potency, and reduced off-target toxicities. Innovations include the design of dual inhibitors targeting mTOR and other critical oncogenic pathways, as well as novel drug formulations for better bioavailability and patient compliance. The applications are expanding beyond established indications, with ongoing research exploring the efficacy of mTOR inhibitors in a broader spectrum of solid tumors and hematological malignancies. Competitive advantages are being carved out through superior clinical trial data demonstrating significant improvements in progression-free survival and overall survival, alongside favorable safety profiles. The integration of these inhibitors into combination therapy regimens is also a key area of development, aiming to overcome treatment resistance and achieve synergistic anti-cancer effects, further solidifying their market position.
Key Drivers of Cancer mTOR Inhibitors Growth
The growth of the Cancer mTOR Inhibitors market is primarily propelled by several key factors. Technologically, advancements in understanding the intricate role of the mTOR pathway in cancer pathogenesis are leading to the development of more targeted and effective inhibitors. Economically, increasing healthcare expenditure globally and a growing demand for advanced cancer treatments are creating a fertile ground for market expansion. Regulatory frameworks are becoming more streamlined for oncology drugs, facilitating faster approvals for promising mTOR inhibitor therapies. Furthermore, the rising incidence of various cancer types, such as breast cancer and hematological malignancies, directly translates into a larger patient pool requiring innovative treatment options. The development of personalized medicine approaches, where mTOR inhibitors can be precisely matched to specific tumor genetic profiles, is also a significant growth accelerator.
Challenges in the Cancer mTOR Inhibitors Market
Despite promising growth, the Cancer mTOR Inhibitors market faces several significant challenges. Regulatory hurdles, though improving, can still lead to prolonged approval timelines for new drugs and indications, impacting market entry. The emergence of drug resistance, a common phenomenon in cancer therapy, poses a substantial challenge, necessitating the development of combination strategies or novel inhibitors. High development costs associated with novel drug discovery and extensive clinical trials can limit the number of companies actively pursuing new mTOR inhibitors. Competition from other targeted therapies and immunotherapies also presents a restraint, requiring mTOR inhibitors to demonstrate clear advantages. Furthermore, issues related to supply chain complexities and manufacturing scalability for advanced therapeutics can impact market accessibility and cost-effectiveness, potentially affecting market penetration.
Emerging Opportunities in Cancer mTOR Inhibitors
The Cancer mTOR Inhibitors market is ripe with emerging opportunities, driven by a confluence of scientific advancements and strategic initiatives. Technological breakthroughs in understanding tumor heterogeneity and resistance mechanisms are opening avenues for developing more refined mTOR inhibitor therapies and predictive biomarkers for patient selection. Strategic partnerships between pharmaceutical giants and academic research institutions are accelerating the discovery and development of novel compounds and combination regimens. Market expansion into underserved geographical regions with a growing cancer burden presents a significant opportunity for increased patient access and commercial growth. The exploration of mTOR inhibitors in combination with emerging modalities like CAR T-cell therapy and oncolytic viruses holds immense potential for synergistic anti-cancer effects. Furthermore, the development of oral formulations and improved drug delivery systems can enhance patient convenience and adherence, driving further market adoption.
Leading Players in the Cancer mTOR Inhibitors Sector
- Abraxis BioScience
- Adimab
- Celgene Corporation
- Celator Pharmaceuticals
- Eli Lilly
- Exelixis
- GlaxoSmithKline
- HEC Pharm
- Intellikine
- Novartis
- Oneness Biotech
- PIQUR Therapeutics
- Semafore Pharmaceuticals
- Takeda
- Wyeth
Key Milestones in Cancer mTOR Inhibitors Industry
- 2019: Significant progress in clinical trials for novel mTOR inhibitors in combination therapies for advanced solid tumors.
- 2020: Approval of a new mTOR inhibitor formulation for a rare pediatric brain tumor, addressing a critical unmet need.
- 2021: Strategic acquisition of a biotech company with a promising pipeline of next-generation mTOR inhibitors by a major pharmaceutical firm.
- 2022: Publication of groundbreaking research elucidating new resistance mechanisms to mTOR inhibitors, paving the way for improved therapeutic strategies.
- 2023: Expansion of an existing mTOR inhibitor's indication to include a novel cancer type, boosting its market reach.
- 2024: Initiation of large-scale Phase III clinical trials for a dual-targeting mTOR inhibitor, signaling potential for significant market disruption.
- 2025 (Estimated): Further regulatory approvals for mTOR inhibitors in combination regimens are anticipated, driving market growth.
Strategic Outlook for Cancer mTOR Inhibitors Market
The strategic outlook for the Cancer mTOR Inhibitors market is exceptionally positive, driven by continuous innovation and expanding therapeutic applications. The future market potential lies in the development of highly personalized treatment strategies, leveraging advanced diagnostics to identify patients most likely to benefit from mTOR inhibition. Growth accelerators include the exploration of novel combination therapies, particularly with emerging immunotherapies, to overcome treatment resistance and enhance efficacy. Strategic opportunities will also arise from expanding into new geographical markets and addressing the growing burden of rare cancers with targeted mTOR inhibitor solutions. The continued investment in R&D by leading players, coupled with evolving regulatory landscapes, will foster a dynamic and expanding market, ultimately improving outcomes for a wider range of cancer patients.
Cancer mTOR Inhibitors Segmentation
-
1. Application
- 1.1. Breast Cancer
- 1.2. Hematological Malignancy
- 1.3. Neuroendocrine Tumors
- 1.4. Hepatocellular Carcinoma
- 1.5. Glioblastoma
-
2. Types
- 2.1. Afinitor/Votubia
- 2.2. Afinitor Disperz and ndash; mTOR inhibitor for Rare Pediatric Brain Tumor
- 2.3. Torisel (Temsirolimus)
- 2.4. Evertor andndash
Cancer mTOR Inhibitors Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
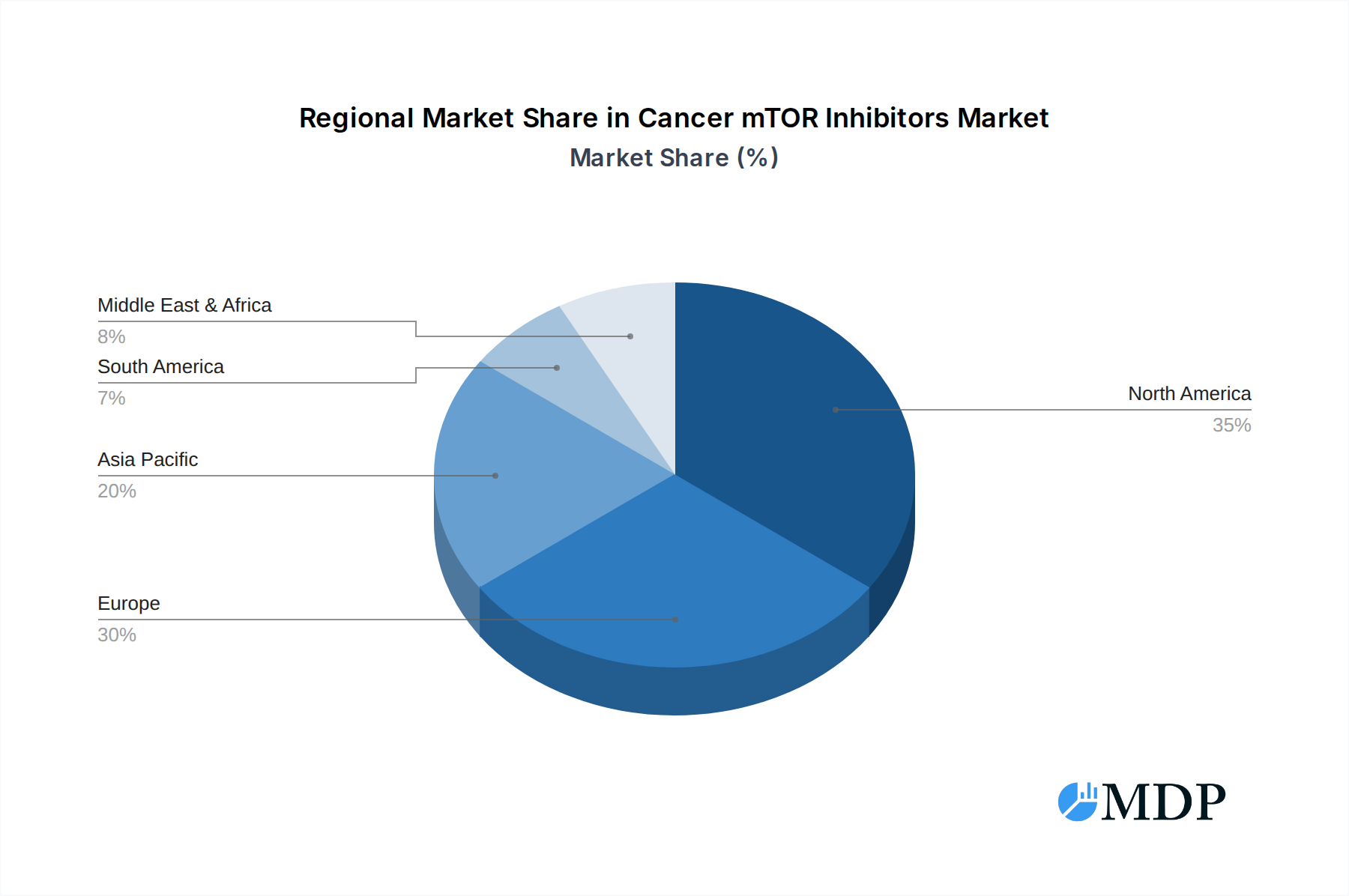
Cancer mTOR Inhibitors Regional Market Share

Geographic Coverage of Cancer mTOR Inhibitors
Cancer mTOR Inhibitors REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.5% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Cancer mTOR Inhibitors Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Breast Cancer
- 5.1.2. Hematological Malignancy
- 5.1.3. Neuroendocrine Tumors
- 5.1.4. Hepatocellular Carcinoma
- 5.1.5. Glioblastoma
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Afinitor/Votubia
- 5.2.2. Afinitor Disperz and ndash; mTOR inhibitor for Rare Pediatric Brain Tumor
- 5.2.3. Torisel (Temsirolimus)
- 5.2.4. Evertor andndash
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Cancer mTOR Inhibitors Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Breast Cancer
- 6.1.2. Hematological Malignancy
- 6.1.3. Neuroendocrine Tumors
- 6.1.4. Hepatocellular Carcinoma
- 6.1.5. Glioblastoma
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Afinitor/Votubia
- 6.2.2. Afinitor Disperz and ndash; mTOR inhibitor for Rare Pediatric Brain Tumor
- 6.2.3. Torisel (Temsirolimus)
- 6.2.4. Evertor andndash
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Cancer mTOR Inhibitors Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Breast Cancer
- 7.1.2. Hematological Malignancy
- 7.1.3. Neuroendocrine Tumors
- 7.1.4. Hepatocellular Carcinoma
- 7.1.5. Glioblastoma
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Afinitor/Votubia
- 7.2.2. Afinitor Disperz and ndash; mTOR inhibitor for Rare Pediatric Brain Tumor
- 7.2.3. Torisel (Temsirolimus)
- 7.2.4. Evertor andndash
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Cancer mTOR Inhibitors Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Breast Cancer
- 8.1.2. Hematological Malignancy
- 8.1.3. Neuroendocrine Tumors
- 8.1.4. Hepatocellular Carcinoma
- 8.1.5. Glioblastoma
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Afinitor/Votubia
- 8.2.2. Afinitor Disperz and ndash; mTOR inhibitor for Rare Pediatric Brain Tumor
- 8.2.3. Torisel (Temsirolimus)
- 8.2.4. Evertor andndash
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Cancer mTOR Inhibitors Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Breast Cancer
- 9.1.2. Hematological Malignancy
- 9.1.3. Neuroendocrine Tumors
- 9.1.4. Hepatocellular Carcinoma
- 9.1.5. Glioblastoma
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Afinitor/Votubia
- 9.2.2. Afinitor Disperz and ndash; mTOR inhibitor for Rare Pediatric Brain Tumor
- 9.2.3. Torisel (Temsirolimus)
- 9.2.4. Evertor andndash
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Cancer mTOR Inhibitors Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Breast Cancer
- 10.1.2. Hematological Malignancy
- 10.1.3. Neuroendocrine Tumors
- 10.1.4. Hepatocellular Carcinoma
- 10.1.5. Glioblastoma
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Afinitor/Votubia
- 10.2.2. Afinitor Disperz and ndash; mTOR inhibitor for Rare Pediatric Brain Tumor
- 10.2.3. Torisel (Temsirolimus)
- 10.2.4. Evertor andndash
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Abraxis BioScience
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Adimab
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Celgene Corporation
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Celator Pharmaceuticals
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Eli Lilly
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Exelixis
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 GlaxoSmithKline
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 HEC Pharm
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Intellikine
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Novartis
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Oneness Biotech
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 PIQUR Therapeutics
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Semafore Pharmaceuticals
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Takeda
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Wyeth
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.1 Abraxis BioScience
List of Figures
- Figure 1: Global Cancer mTOR Inhibitors Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Cancer mTOR Inhibitors Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Cancer mTOR Inhibitors Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Cancer mTOR Inhibitors Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Cancer mTOR Inhibitors Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Cancer mTOR Inhibitors Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Cancer mTOR Inhibitors Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Cancer mTOR Inhibitors Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Cancer mTOR Inhibitors Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Cancer mTOR Inhibitors Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Cancer mTOR Inhibitors Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Cancer mTOR Inhibitors Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Cancer mTOR Inhibitors Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Cancer mTOR Inhibitors Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Cancer mTOR Inhibitors Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Cancer mTOR Inhibitors Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Cancer mTOR Inhibitors Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Cancer mTOR Inhibitors Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Cancer mTOR Inhibitors Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Cancer mTOR Inhibitors Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Cancer mTOR Inhibitors Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Cancer mTOR Inhibitors Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Cancer mTOR Inhibitors Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Cancer mTOR Inhibitors Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Cancer mTOR Inhibitors Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Cancer mTOR Inhibitors Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Cancer mTOR Inhibitors Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Cancer mTOR Inhibitors Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Cancer mTOR Inhibitors Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Cancer mTOR Inhibitors Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Cancer mTOR Inhibitors Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Cancer mTOR Inhibitors Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Cancer mTOR Inhibitors Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Cancer mTOR Inhibitors?
The projected CAGR is approximately 5.5%.
2. Which companies are prominent players in the Cancer mTOR Inhibitors?
Key companies in the market include Abraxis BioScience, Adimab, Celgene Corporation, Celator Pharmaceuticals, Eli Lilly, Exelixis, GlaxoSmithKline, HEC Pharm, Intellikine, Novartis, Oneness Biotech, PIQUR Therapeutics, Semafore Pharmaceuticals, Takeda, Wyeth.
3. What are the main segments of the Cancer mTOR Inhibitors?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Cancer mTOR Inhibitors," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Cancer mTOR Inhibitors report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Cancer mTOR Inhibitors?
To stay informed about further developments, trends, and reports in the Cancer mTOR Inhibitors, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


