Key Insights
The global Tumor Immune Cell Therapy market is experiencing robust expansion, projected to reach an estimated market size of approximately \$35,000 million in 2025. This growth is fueled by a projected Compound Annual Growth Rate (CAGR) of around 18% during the forecast period of 2025-2033. This significant expansion is primarily driven by the increasing prevalence of both hematological and solid tumors worldwide, alongside a growing understanding of the human immune system's potential in combating cancer. Key advancements in therapies such as Immune Checkpoint Inhibitors (ICIs), Chimeric Antigen Receptor T-cell Therapy (CAR-T), and Tumor-Infiltrating Lymphocytes (TILs) are revolutionizing cancer treatment paradigms. The effectiveness of these therapies in achieving durable remissions and improving patient outcomes, even in previously intractable cancers, is a major catalyst for market growth. Furthermore, substantial investments in research and development by leading biopharmaceutical companies are continually introducing novel therapeutic approaches and expanding the application of existing ones.
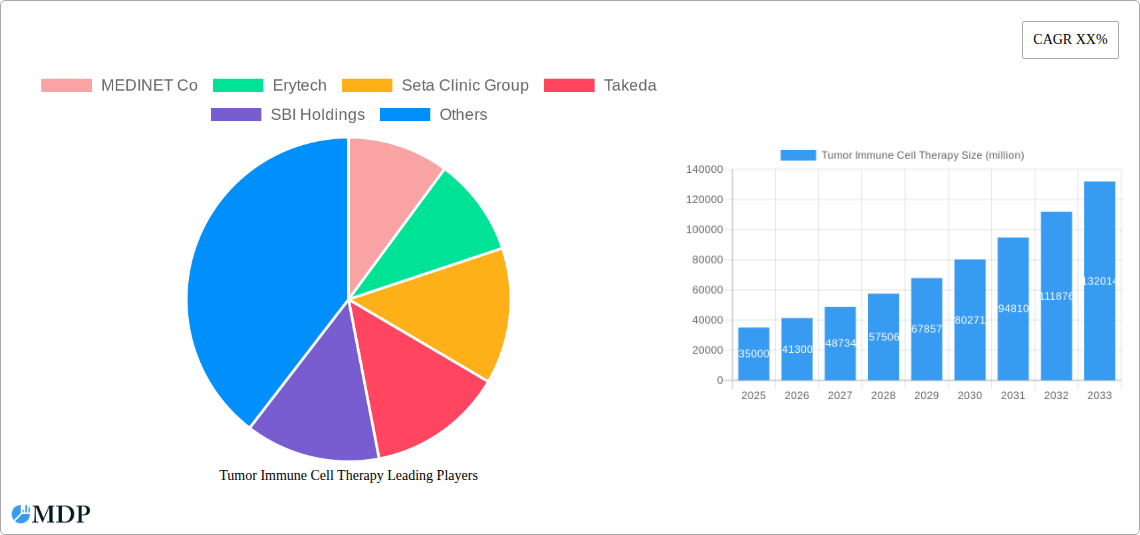
Tumor Immune Cell Therapy Market Size (In Billion)

The market landscape is characterized by a dynamic interplay of established pharmaceutical giants and innovative biotech firms, including companies like Novartis, Gilead Sciences, and Legend Biotech, alongside emerging players such as Fate Therapeutics and Vericel Corporation. These companies are actively pursuing the development of next-generation cell therapies, exploring new targets, enhancing efficacy, and addressing manufacturing complexities. Regional dominance is observed in North America, particularly the United States, driven by advanced healthcare infrastructure, high cancer incidence, and significant R&D expenditure. Europe and Asia Pacific are also witnessing substantial growth, with China and India emerging as key markets due to increasing healthcare access and government initiatives supporting advanced therapies. However, challenges such as high treatment costs, the need for specialized manufacturing facilities, and potential side effects associated with these potent therapies present ongoing hurdles. Nonetheless, the transformative potential of immune cell therapies in oncology continues to drive innovation and market expansion, promising a significant impact on patient lives.
Tumor Immune Cell Therapy Company Market Share

Unlock the Future of Cancer Treatment: Tumor Immune Cell Therapy Market Report (2019-2033)
Gain unparalleled insights into the rapidly evolving Tumor Immune Cell Therapy market, a critical frontier in oncology. This comprehensive report, covering the study period of 2019–2033 with a base year of 2025, provides an in-depth analysis of market dynamics, technological advancements, and strategic opportunities. Discover how leading companies like MEDINET Co, Erytech, Seta Clinic Group, Takeda, SBI Holdings, Oxford BioTherapeutics, Fate Therapeutics, Gilead Sciences, Novartis, Osiris, Vericel Corporation, Fujifilm Cellular Dynamics, JCR Pharmaceuticals, Beike Biotechnology, Golden Meditech, Guanhao Biotech, Beroni Group, and Legend Biotech are shaping the landscape of cancer immunotherapy. With a projected market size reaching hundreds of millions, this report is essential for investors, researchers, and stakeholders seeking to capitalize on this transformative sector. We meticulously analyze the market penetration and growth of key therapeutic types including Immune Checkpoint Inhibitors, Chimeric Antigen Receptor T-cell Therapy (CAR T-cell Therapy), Tumor-Infiltrating Lymphocytes Therapy, Cancer Vaccines, Bispecific Antibodies, and Cytokine Therapy, across both Hematological Tumors and Solid Tumors. This report is your definitive guide to navigating the complexities and harnessing the immense potential of cancer treatment innovation.
Tumor Immune Cell Therapy Market Dynamics & Concentration
The Tumor Immune Cell Therapy market is characterized by moderate to high concentration, with key players actively investing in research and development, driving innovation and market share. Market share distribution is influenced by the success of CAR T-cell Therapy and the expanding applications of Immune Checkpoint Inhibitors. Significant innovation drivers include advancements in genetic engineering, better understanding of tumor microenvironments, and personalized medicine approaches. Regulatory frameworks, while evolving, present both opportunities and challenges, with agencies like the FDA and EMA playing a crucial role in product approvals and market access. Product substitutes, such as traditional chemotherapy and radiation therapy, continue to be present but are increasingly being complemented or replaced by immunotherapy. End-user trends show a growing demand for targeted and less toxic treatments, with patients and clinicians favoring therapies with demonstrated efficacy and improved quality of life. M&A activities are a prominent feature, with numerous M&A deal counts reflecting strategic consolidation and pipeline expansion. For example, a recent acquisition in the CAR T-cell Therapy space by a major pharmaceutical company aimed to strengthen its solid tumor portfolio. Collaboration and licensing agreements are also prevalent, fostering a dynamic competitive environment.
Tumor Immune Cell Therapy Industry Trends & Analysis
The Tumor Immune Cell Therapy industry is experiencing robust growth, driven by a confluence of escalating cancer incidence, increasing healthcare expenditure, and groundbreaking scientific discoveries. The market is projected to witness a substantial CAGR of over 15% during the forecast period. A significant trend is the rising market penetration of CAR T-cell Therapy for hematological tumors, with ongoing research expanding its efficacy into solid tumors. Technological disruptions are at the core of this market's dynamism, with continuous innovation in areas such as gene editing tools (e.g., CRISPR-Cas9) for enhanced T-cell engineering, development of novel cancer vaccines tailored to specific tumor antigens, and the refinement of bispecific antibodies designed to bridge immune cells and tumor cells. Consumer preferences are shifting towards therapies offering durable responses and a favorable safety profile, leading to increased demand for advanced immunotherapies. Competitive dynamics are intensifying, with both established pharmaceutical giants and emerging biotechnology firms vying for market leadership through significant R&D investments and strategic partnerships. The development of next-generation cytokine therapy and tumor-infiltrating lymphocytes (TILs) therapy also presents significant growth avenues. The increasing prevalence of cancer globally, coupled with government initiatives supporting biomedical research, further propels market expansion.
Leading Markets & Segments in Tumor Immune Cell Therapy
The North America region currently dominates the Tumor Immune Cell Therapy market, with the United States leading in terms of research, development, and adoption. This dominance is fueled by a strong healthcare infrastructure, significant R&D investments from both public and private sectors, and a high prevalence of cancer. Within the application segments, Hematological Tumors have been the primary focus, particularly for Chimeric Antigen Receptor T-cell Therapy (CAR T-cell Therapy), which has shown remarkable success in treating B-cell malignancies like leukemia and lymphoma. However, the market for Solid Tumors is rapidly expanding, presenting a vast untapped potential for various immunotherapies.
Key drivers for this regional dominance include:
- Economic Policies: Favorable government funding for cancer research and biopharmaceutical innovation.
- Infrastructure: Advanced clinical trial networks and robust manufacturing capabilities for cell and gene therapies.
- Regulatory Environment: A well-established and efficient regulatory pathway for novel therapies, albeit rigorous.
- Healthcare Expenditure: High per capita healthcare spending enabling greater access to advanced treatments.
Among the therapeutic types, Immune Checkpoint Inhibitors have achieved widespread adoption across multiple cancer types, demonstrating significant survival benefits. CAR T-cell Therapy continues to be a major growth segment, with ongoing efforts to improve its efficacy and safety profile, and to expand its application to solid tumors. Tumor-Infiltrating Lymphocytes Therapy (TILs Therapy) is emerging as a promising approach for solid tumors, particularly melanoma. Cancer Vaccines, both therapeutic and prophylactic, are witnessing renewed interest and development. Bispecific Antibodies offer a versatile platform for engaging the immune system against cancer, with a growing pipeline. Cytokine Therapy, while a more established modality, is being revisited with novel delivery systems and combination strategies. The increasing investment in R&D for these diverse therapeutic types underscores the dynamic nature of the market.
Tumor Immune Cell Therapy Product Developments
Product development in Tumor Immune Cell Therapy is characterized by a relentless pursuit of enhanced efficacy and expanded applicability. Innovations focus on engineering T-cells with improved persistence and tumor-targeting capabilities for CAR T-cell Therapy, and developing novel cancer vaccines that elicit robust and durable anti-tumor immune responses. Companies are also advancing bispecific antibodies to simultaneously target multiple tumor antigens or engage different immune cell populations. The development of combination therapies, integrating immunotherapies with conventional treatments or other immunomodulatory agents, is a key trend aimed at overcoming resistance mechanisms and improving patient outcomes, particularly in solid tumors.
Key Drivers of Tumor Immune Cell Therapy Growth
Several key drivers are propelling the growth of the Tumor Immune Cell Therapy market. Technologically, advancements in genetic engineering, cell culture techniques, and bioinformatics are enabling the development of more potent and targeted therapies. Economically, rising global healthcare expenditure and increasing patient affordability for advanced treatments are significant contributors. Regulatory bodies are also streamlining approval pathways for innovative immunotherapies, albeit with stringent safety and efficacy requirements. Furthermore, the growing understanding of tumor immunology and the tumor microenvironment is unlocking new therapeutic targets and strategies. The increasing prevalence of chronic diseases and the demand for personalized medicine further bolster growth.
Challenges in the Tumor Immune Cell Therapy Market
Despite its immense promise, the Tumor Immune Cell Therapy market faces several significant challenges. High manufacturing costs and complex production processes for cell-based therapies remain a substantial barrier, impacting affordability and accessibility. Regulatory hurdles, including the need for extensive clinical trials to demonstrate safety and efficacy, particularly for novel CAR T-cell Therapy applications in solid tumors, can lead to lengthy approval timelines. Supply chain complexities and the need for specialized infrastructure for cell manipulation and cryopreservation are also critical considerations. Intense competition from established and emerging players, along with the potential for off-target toxicities and immune-related adverse events, necessitate continuous innovation and rigorous patient monitoring.
Emerging Opportunities in Tumor Immune Cell Therapy
Emerging opportunities in the Tumor Immune Cell Therapy market are primarily driven by scientific breakthroughs and strategic market expansion. The exploration of new CAR T-cell Therapy targets for solid tumors, the development of "off-the-shelf" allogeneic cell therapies to overcome the limitations of autologous treatments, and advancements in cancer vaccine technologies represent significant growth catalysts. Strategic partnerships between academic institutions, biotechnology firms, and large pharmaceutical companies are accelerating the translation of research into clinical applications. Furthermore, the expansion of these therapies into emerging markets and their potential use in earlier lines of cancer treatment offer substantial long-term growth prospects.
Leading Players in the Tumor Immune Cell Therapy Sector
MEDINET Co Erytech Seta Clinic Group Takeda SBI Holdings Oxford BioTherapeutics Fate Therapeutics Gilead Sciences Novartis Osiris Vericel Corporation Fujifilm Cellular Dynamics JCR Pharmaceuticals Beike Biotechnology Golden Meditech Guanhao Biotech Beroni Group Legend Biotech
Key Milestones in Tumor Immune Cell Therapy Industry
- 2019: Approval of an additional CAR T-cell therapy for adult diffuse large B-cell lymphoma.
- 2020: Significant advancements in bispecific antibody development for various cancers.
- 2021: Increased focus on overcoming CAR T-cell Therapy resistance in solid tumors.
- 2022: Major clinical trial results published for novel cancer vaccine candidates.
- 2023: Expansion of Immune Checkpoint Inhibitors to new cancer indications and combination therapies.
- 2024: Emerging data showcasing promising results for Tumor-Infiltrating Lymphocytes Therapy in difficult-to-treat cancers.
Strategic Outlook for Tumor Immune Cell Therapy Market
The strategic outlook for the Tumor Immune Cell Therapy market remains exceptionally positive, driven by continuous innovation and an expanding therapeutic addressable market. Future growth will be fueled by the development of next-generation cell therapies, including allogeneic and gene-edited approaches, and the successful translation of promising cancer vaccine and bispecific antibody candidates into clinical practice. Increased investment in research for solid tumor applications and the exploration of novel immune-modulating agents are critical. Strategic collaborations, market penetration in underserved regions, and a focus on optimizing manufacturing and affordability will be key to realizing the full potential of this transformative field in cancer treatment.
Tumor Immune Cell Therapy Segmentation
-
1. Application
- 1.1. Hematological Tumors
- 1.2. Solid Tumors
-
2. Types
- 2.1. Immune Checkpoint Inhibitors
- 2.2. Chimeric Antigen Receptor T-cell Therapy
- 2.3. Tumor-Infiltrating Lymphocytes Therapy
- 2.4. Cancer Vaccines
- 2.5. Bispecific Antibodies
- 2.6. Cytokine Therapy
Tumor Immune Cell Therapy Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
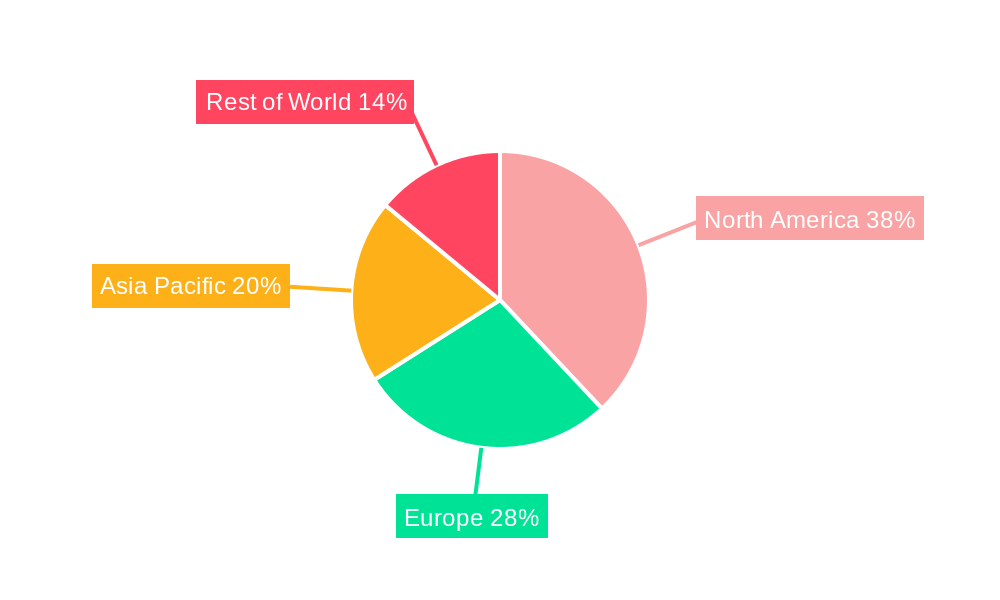
Tumor Immune Cell Therapy Regional Market Share

Geographic Coverage of Tumor Immune Cell Therapy
Tumor Immune Cell Therapy REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 35.39% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. MDP Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hematological Tumors
- 5.1.2. Solid Tumors
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Immune Checkpoint Inhibitors
- 5.2.2. Chimeric Antigen Receptor T-cell Therapy
- 5.2.3. Tumor-Infiltrating Lymphocytes Therapy
- 5.2.4. Cancer Vaccines
- 5.2.5. Bispecific Antibodies
- 5.2.6. Cytokine Therapy
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Tumor Immune Cell Therapy Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hematological Tumors
- 6.1.2. Solid Tumors
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Immune Checkpoint Inhibitors
- 6.2.2. Chimeric Antigen Receptor T-cell Therapy
- 6.2.3. Tumor-Infiltrating Lymphocytes Therapy
- 6.2.4. Cancer Vaccines
- 6.2.5. Bispecific Antibodies
- 6.2.6. Cytokine Therapy
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Tumor Immune Cell Therapy Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hematological Tumors
- 7.1.2. Solid Tumors
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Immune Checkpoint Inhibitors
- 7.2.2. Chimeric Antigen Receptor T-cell Therapy
- 7.2.3. Tumor-Infiltrating Lymphocytes Therapy
- 7.2.4. Cancer Vaccines
- 7.2.5. Bispecific Antibodies
- 7.2.6. Cytokine Therapy
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Tumor Immune Cell Therapy Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hematological Tumors
- 8.1.2. Solid Tumors
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Immune Checkpoint Inhibitors
- 8.2.2. Chimeric Antigen Receptor T-cell Therapy
- 8.2.3. Tumor-Infiltrating Lymphocytes Therapy
- 8.2.4. Cancer Vaccines
- 8.2.5. Bispecific Antibodies
- 8.2.6. Cytokine Therapy
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Tumor Immune Cell Therapy Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hematological Tumors
- 9.1.2. Solid Tumors
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Immune Checkpoint Inhibitors
- 9.2.2. Chimeric Antigen Receptor T-cell Therapy
- 9.2.3. Tumor-Infiltrating Lymphocytes Therapy
- 9.2.4. Cancer Vaccines
- 9.2.5. Bispecific Antibodies
- 9.2.6. Cytokine Therapy
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Tumor Immune Cell Therapy Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hematological Tumors
- 10.1.2. Solid Tumors
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Immune Checkpoint Inhibitors
- 10.2.2. Chimeric Antigen Receptor T-cell Therapy
- 10.2.3. Tumor-Infiltrating Lymphocytes Therapy
- 10.2.4. Cancer Vaccines
- 10.2.5. Bispecific Antibodies
- 10.2.6. Cytokine Therapy
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Tumor Immune Cell Therapy Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Hematological Tumors
- 11.1.2. Solid Tumors
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Immune Checkpoint Inhibitors
- 11.2.2. Chimeric Antigen Receptor T-cell Therapy
- 11.2.3. Tumor-Infiltrating Lymphocytes Therapy
- 11.2.4. Cancer Vaccines
- 11.2.5. Bispecific Antibodies
- 11.2.6. Cytokine Therapy
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 MEDINET Co
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Erytech
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Seta Clinic Group
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Takeda
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 SBI Holdings
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Oxford BioTherapeutics
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Fate Therapeutics
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Gilead Sciences
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Novartis
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Osiris
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Vericel Corporation
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Fujifilm Cellular Dynamics
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 JCR Pharmaceuticals
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.14 Beike Biotechnology
- 12.1.14.1. Company Overview
- 12.1.14.2. Products
- 12.1.14.3. Company Financials
- 12.1.14.4. SWOT Analysis
- 12.1.15 Golden Meditech
- 12.1.15.1. Company Overview
- 12.1.15.2. Products
- 12.1.15.3. Company Financials
- 12.1.15.4. SWOT Analysis
- 12.1.16 Guanhao Biotech
- 12.1.16.1. Company Overview
- 12.1.16.2. Products
- 12.1.16.3. Company Financials
- 12.1.16.4. SWOT Analysis
- 12.1.17 Beroni Group
- 12.1.17.1. Company Overview
- 12.1.17.2. Products
- 12.1.17.3. Company Financials
- 12.1.17.4. SWOT Analysis
- 12.1.18 Legend Biotech
- 12.1.18.1. Company Overview
- 12.1.18.2. Products
- 12.1.18.3. Company Financials
- 12.1.18.4. SWOT Analysis
- 12.1.1 MEDINET Co
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Tumor Immune Cell Therapy Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America Tumor Immune Cell Therapy Revenue (billion), by Application 2025 & 2033
- Figure 3: North America Tumor Immune Cell Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Tumor Immune Cell Therapy Revenue (billion), by Types 2025 & 2033
- Figure 5: North America Tumor Immune Cell Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Tumor Immune Cell Therapy Revenue (billion), by Country 2025 & 2033
- Figure 7: North America Tumor Immune Cell Therapy Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Tumor Immune Cell Therapy Revenue (billion), by Application 2025 & 2033
- Figure 9: South America Tumor Immune Cell Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Tumor Immune Cell Therapy Revenue (billion), by Types 2025 & 2033
- Figure 11: South America Tumor Immune Cell Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Tumor Immune Cell Therapy Revenue (billion), by Country 2025 & 2033
- Figure 13: South America Tumor Immune Cell Therapy Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Tumor Immune Cell Therapy Revenue (billion), by Application 2025 & 2033
- Figure 15: Europe Tumor Immune Cell Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Tumor Immune Cell Therapy Revenue (billion), by Types 2025 & 2033
- Figure 17: Europe Tumor Immune Cell Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Tumor Immune Cell Therapy Revenue (billion), by Country 2025 & 2033
- Figure 19: Europe Tumor Immune Cell Therapy Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Tumor Immune Cell Therapy Revenue (billion), by Application 2025 & 2033
- Figure 21: Middle East & Africa Tumor Immune Cell Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Tumor Immune Cell Therapy Revenue (billion), by Types 2025 & 2033
- Figure 23: Middle East & Africa Tumor Immune Cell Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Tumor Immune Cell Therapy Revenue (billion), by Country 2025 & 2033
- Figure 25: Middle East & Africa Tumor Immune Cell Therapy Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Tumor Immune Cell Therapy Revenue (billion), by Application 2025 & 2033
- Figure 27: Asia Pacific Tumor Immune Cell Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Tumor Immune Cell Therapy Revenue (billion), by Types 2025 & 2033
- Figure 29: Asia Pacific Tumor Immune Cell Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Tumor Immune Cell Therapy Revenue (billion), by Country 2025 & 2033
- Figure 31: Asia Pacific Tumor Immune Cell Therapy Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Application 2020 & 2033
- Table 2: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Types 2020 & 2033
- Table 3: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Application 2020 & 2033
- Table 5: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Types 2020 & 2033
- Table 6: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Country 2020 & 2033
- Table 7: United States Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Canada Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 9: Mexico Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Application 2020 & 2033
- Table 11: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Types 2020 & 2033
- Table 12: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Brazil Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: Argentina Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Application 2020 & 2033
- Table 17: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Types 2020 & 2033
- Table 18: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 20: Germany Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 21: France Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 22: Italy Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Spain Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: Russia Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 25: Benelux Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Nordics Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Application 2020 & 2033
- Table 29: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Types 2020 & 2033
- Table 30: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Country 2020 & 2033
- Table 31: Turkey Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: Israel Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 33: GCC Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: North Africa Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 35: South Africa Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 37: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Application 2020 & 2033
- Table 38: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Types 2020 & 2033
- Table 39: Global Tumor Immune Cell Therapy Revenue billion Forecast, by Country 2020 & 2033
- Table 40: China Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 41: India Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 42: Japan Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 43: South Korea Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 45: Oceania Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Tumor Immune Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Tumor Immune Cell Therapy?
The projected CAGR is approximately 35.39%.
2. Which companies are prominent players in the Tumor Immune Cell Therapy?
Key companies in the market include MEDINET Co, Erytech, Seta Clinic Group, Takeda, SBI Holdings, Oxford BioTherapeutics, Fate Therapeutics, Gilead Sciences, Novartis, Osiris, Vericel Corporation, Fujifilm Cellular Dynamics, JCR Pharmaceuticals, Beike Biotechnology, Golden Meditech, Guanhao Biotech, Beroni Group, Legend Biotech.
3. What are the main segments of the Tumor Immune Cell Therapy?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 7.45 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3950.00, USD 5925.00, and USD 7900.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Tumor Immune Cell Therapy," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Tumor Immune Cell Therapy report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Tumor Immune Cell Therapy?
To stay informed about further developments, trends, and reports in the Tumor Immune Cell Therapy, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


