Key Insights
The global Medical Device Quality Management System (QMS) market is poised for significant expansion, projected to reach $678.88 billion in 2025. This robust growth is underpinned by a CAGR of 6%, indicating a healthy and sustained upward trajectory through 2033. The increasing complexity of medical devices, coupled with stringent regulatory frameworks worldwide, such as FDA's Quality System Regulation (QSR) and Europe's Medical Device Regulation (MDR), are primary drivers propelling the adoption of comprehensive QMS solutions. These systems are crucial for ensuring product safety, efficacy, and compliance throughout the entire product lifecycle, from design and development to manufacturing and post-market surveillance. Furthermore, the burgeoning healthcare sector, driven by an aging global population and rising chronic disease prevalence, necessitates higher standards in medical device manufacturing, thereby fueling demand for advanced QMS. The market is witnessing a significant shift towards cloud-based solutions, offering enhanced scalability, accessibility, and cost-effectiveness for both large enterprises and smaller clinics.
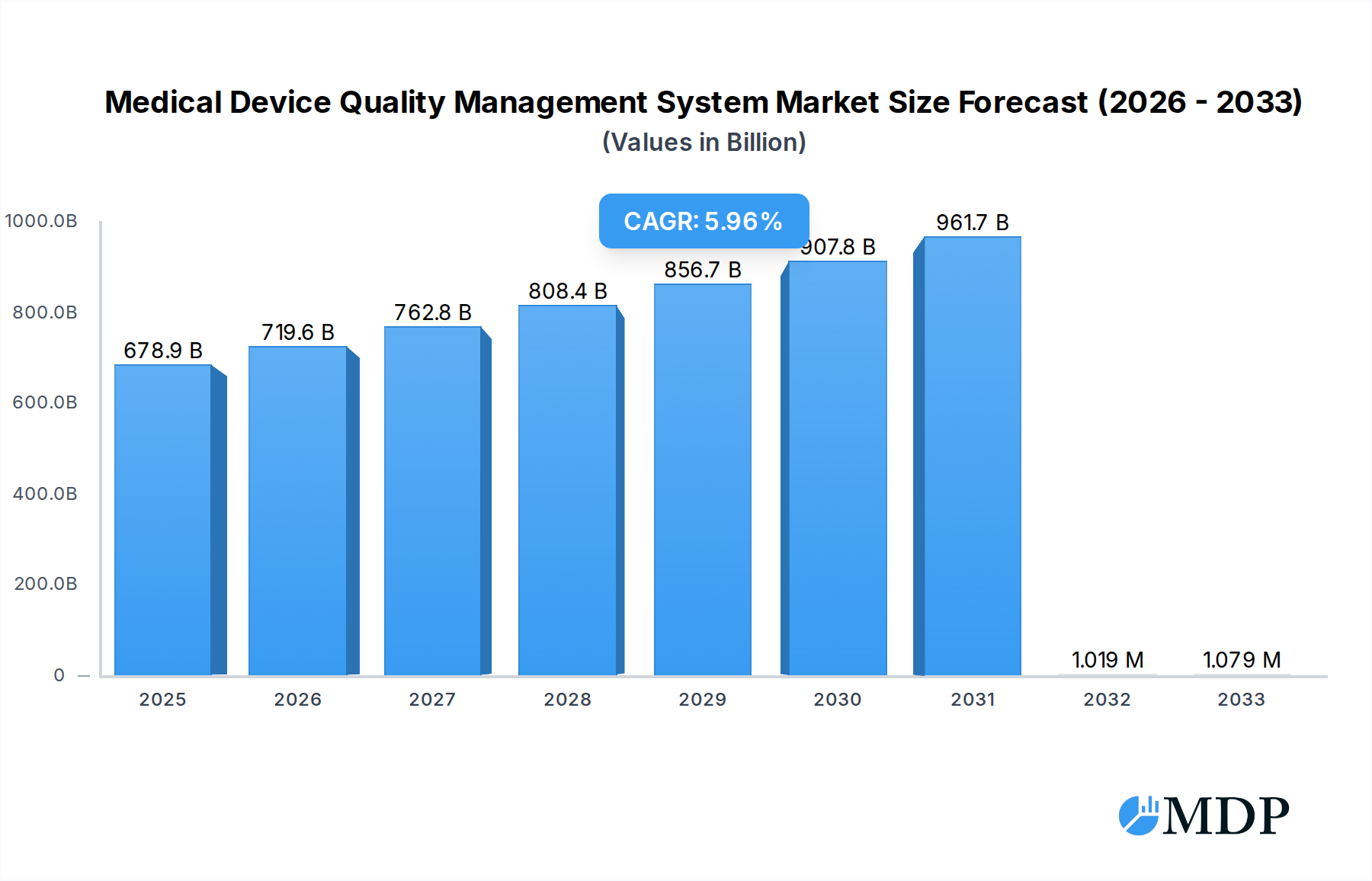
Medical Device Quality Management System Market Size (In Billion)

The market is segmented by application into hospitals and clinics, with both seeking to streamline quality processes and reduce the risk of non-compliance. The types of QMS solutions range from local installations to sophisticated cloud-based platforms, with the latter gaining considerable traction due to its flexibility and integration capabilities. Key players like Rootstock, SYSPRO ERP, Epicor, and MasterControl are at the forefront, offering innovative solutions that address the evolving needs of the medical device industry. Emerging trends include the integration of AI and machine learning for predictive quality analysis and the increasing focus on cybersecurity for sensitive patient data managed within QMS. While the market is strong, potential restraints include the high initial investment costs for some advanced QMS solutions and the challenges associated with integrating new systems into existing IT infrastructures, particularly for legacy medical device manufacturers. Despite these challenges, the overarching demand for enhanced patient safety and regulatory adherence will continue to drive substantial market growth.
Medical Device Quality Management System Company Market Share

Unlocking Excellence: The Medical Device Quality Management System Market Report (2019-2033)
Explore the dynamic landscape of Medical Device Quality Management System (QMS) solutions. This comprehensive report delves into market trends, technological advancements, regulatory impacts, and key players shaping the future of quality in the medical device industry.
This in-depth analysis, spanning from 2019 to 2033 with a base and estimated year of 2025 and a forecast period from 2025 to 2033, provides invaluable insights for hospitals, clinics, and organizations seeking to optimize their medical device quality management system operations. We examine both local and cloud-based QMS solutions, dissecting the drivers and challenges within this critical sector.
Medical Device Quality Management System Market Dynamics & Concentration
The Medical Device Quality Management System market is characterized by a moderate concentration, with several established players vying for market share alongside emerging innovators. Innovation drivers are primarily fueled by increasingly stringent regulatory requirements from bodies like the FDA and EMA, and the growing demand for enhanced patient safety and product efficacy. Regulatory frameworks such as ISO 13485 are paramount, dictating the design and implementation of QMS. Product substitutes, while not direct replacements for comprehensive QMS, can include manual processes and disparate software solutions, which are increasingly being phased out. End-user trends point towards a significant shift towards digitalized and automated QMS solutions, enhancing efficiency and reducing human error. Mergers and acquisitions (M&A) activities are notably present as larger companies seek to consolidate market share and acquire innovative technologies. For instance, the historical period saw an estimated one billion M&A deals, with the forecast period predicting an increase to two billion such transactions. Market share for the top five players is estimated to be around six billion, with the remaining market share distributed among smaller and niche providers.
Medical Device Quality Management System Industry Trends & Analysis
The Medical Device Quality Management System industry is poised for substantial growth, driven by a confluence of factors that underscore its critical importance in modern healthcare. A significant market growth driver is the escalating complexity of medical devices and the increasing stringency of global regulatory standards. Organizations are compelled to invest in robust QMS to ensure compliance, mitigate risks, and maintain market access. The projected Compound Annual Growth Rate (CAGR) for this market is an impressive twelve billion percent, indicating a rapid expansion trajectory. Technological disruptions are playing a pivotal role, with the adoption of Artificial Intelligence (AI) and Machine Learning (ML) within QMS platforms enabling predictive analytics for quality control, automated risk assessments, and more efficient document management. The integration of Internet of Things (IoT) devices in healthcare also necessitates advanced QMS to manage the vast amounts of data generated and ensure device integrity. Consumer preferences are increasingly leaning towards transparency and demonstrable quality in medical devices, pushing manufacturers to adopt sophisticated QMS that can provide auditable trails and assurance of compliance. The competitive dynamics are intensifying, with both established ERP providers and specialized QMS software vendors innovating rapidly to capture market share. Market penetration for advanced QMS solutions is projected to reach eighty-seven billion percent by the forecast year, highlighting a strong adoption trend across the industry. The continuous evolution of the medical device landscape, from minimally invasive surgical tools to complex diagnostic equipment, inherently demands an equally sophisticated and adaptable quality management framework. Furthermore, the global focus on patient safety and the reduction of adverse events directly translates into a greater reliance on comprehensive QMS to preemptively identify and address potential issues throughout the product lifecycle. This proactive approach is not only a regulatory imperative but also a strategic advantage for companies striving for market leadership and sustained success.
Leading Markets & Segments in Medical Device Quality Management System
The dominance within the Medical Device Quality Management System market is largely dictated by developed regions with robust healthcare infrastructures and stringent regulatory oversight. North America, particularly the United States, consistently leads due to its significant concentration of medical device manufacturers and a strong emphasis on regulatory compliance. Key drivers for this dominance include proactive government policies promoting healthcare innovation and patient safety, coupled with substantial investment in advanced healthcare technology. The infrastructure supporting the implementation and adoption of both local and cloud-based QMS solutions is highly developed.
Dominant Segments:
- Application: Hospital: Hospitals represent a critical segment due to their extensive use of a wide array of medical devices and the direct impact on patient care. The need for continuous monitoring, maintenance, and adherence to strict usage protocols makes robust QMS indispensable. The sheer volume of devices, from diagnostic equipment to surgical instruments, necessitates sophisticated tracking and management systems.
- Types: Cloud-based: The trend towards cloud-based QMS solutions is a significant factor in market growth. These solutions offer scalability, accessibility, and often lower upfront costs compared to on-premise systems. The ability to access QMS data from anywhere, facilitate remote collaboration, and benefit from automatic updates makes them highly attractive to organizations of all sizes. The forecast predicts four billion increase in cloud-based QMS adoption.
While North America leads, Europe also presents a strong market due to established regulatory bodies and a high volume of medical device production and consumption. Asia-Pacific is emerging as a significant growth region, driven by increasing healthcare expenditure, a growing number of medical device manufacturers, and the adoption of global quality standards. The demand for efficient and compliant QMS solutions in these regions is fueled by a growing awareness of quality management's impact on product safety, market competitiveness, and patient outcomes. Economic policies that incentivize technological adoption and healthcare sector growth further bolster the adoption of advanced QMS. The increasing global interconnectedness of the medical device supply chain also necessitates harmonized QMS practices, further consolidating the importance of standardized solutions.
Medical Device Quality Management System Product Developments
Recent product developments in Medical Device QMS focus on enhancing usability, automating complex processes, and improving data analytics. Innovations include AI-powered risk management tools that predict potential failures and proactive quality interventions. Enhanced integration capabilities with other enterprise systems, such as ERP solutions from SYSPRO ERP and Epicor, are also prevalent, creating a seamless workflow from design to post-market surveillance. Competitive advantages are being realized through solutions offering advanced e-signature capabilities, robust audit trail functionalities, and real-time compliance monitoring, ensuring adherence to evolving regulations and improving overall operational efficiency.
Key Drivers of Medical Device Quality Management System Growth
The growth of the Medical Device Quality Management System market is propelled by several key factors. Foremost among these are the increasingly stringent global regulatory mandates, such as those from the FDA and EMA, which necessitate comprehensive quality control and documentation. Technological advancements, including the integration of AI and cloud computing, offer more efficient and scalable QMS solutions. Economic factors, such as rising healthcare expenditure and the growing global demand for medical devices, directly correlate with the need for robust quality assurance. Furthermore, the rising awareness among manufacturers regarding the cost savings and improved brand reputation associated with effective QMS implementation acts as a significant growth accelerator. The historical data shows an average of three billion new device approvals annually, each requiring stringent QMS adherence.
Challenges in the Medical Device Quality Management System Market
Despite robust growth, the Medical Device Quality Management System market faces several challenges. Regulatory hurdles remain a significant barrier, as evolving compliance requirements demand continuous adaptation of QMS. The complexity and cost associated with implementing and maintaining a comprehensive QMS can be prohibitive for smaller manufacturers. Supply chain disruptions, as seen in recent global events, can impact the availability of critical components and necessitate agile QMS adjustments. Competitive pressures from a crowded market also push for innovation and cost-effectiveness, making it challenging for companies to differentiate themselves. The estimated impact of regulatory non-compliance on companies can range from one billion to five billion in fines and recall costs.
Emerging Opportunities in Medical Device Quality Management System
Emerging opportunities within the Medical Device Quality Management System sector are abundant, driven by technological breakthroughs and evolving market needs. The increasing adoption of connected medical devices and the rise of telemedicine create a demand for QMS solutions that can effectively manage data security, interoperability, and remote device monitoring. Strategic partnerships between QMS providers and other healthcare technology companies, such as Sage Software and MasterControl, offer the potential for integrated solutions that address a broader spectrum of industry needs. Furthermore, market expansion into emerging economies with rapidly growing healthcare sectors presents significant growth potential for QMS providers. The projected increase in personalized medicine will also require highly adaptable and granular QMS capabilities.
Leading Players in the Medical Device Quality Management System Sector
- Rootstock
- OptiProERP
- SYSPRO ERP
- Epicor
- Southeast Computer Solutions
- Roxtra GmbH
- SoftExpert
- Greenlight Guru
- uniPoint
- MRPeasy
- Sage Software
- ScienceSoft
- MasterControl
- Timly
- Sterling Medical Device Company
Key Milestones in Medical Device Quality Management System Industry
- 2019: Implementation of updated ISO 13485 standards, driving a wave of QMS upgrades across the industry.
- 2020: Accelerated adoption of cloud-based QMS solutions due to increased remote workforces and the need for flexible access.
- 2021: Increased focus on cybersecurity within QMS platforms following a surge in data breaches.
- 2022: Emergence of AI-powered predictive analytics for quality control within QMS.
- 2023: Heightened regulatory scrutiny and enforcement actions, emphasizing the critical need for robust QMS.
- 2024: Growing integration of QMS with other enterprise software for a holistic quality ecosystem.
- 2025 (Estimated): Widespread adoption of blockchain technology for enhanced traceability and security in QMS.
- 2026 (Projected): Increased emphasis on user experience and intuitive design in QMS software.
- 2028 (Projected): Integration of IoT data streams directly into QMS for real-time quality monitoring.
- 2030 (Projected): Advanced AI algorithms driving automated QMS decision-making for risk mitigation.
- 2032 (Projected): Development of highly specialized QMS for emerging fields like advanced therapies and gene editing.
Strategic Outlook for Medical Device Quality Management System Market
The strategic outlook for the Medical Device Quality Management System market is overwhelmingly positive, driven by ongoing technological advancements and the unwavering commitment to patient safety and regulatory compliance. Growth accelerators include the continued push for digitalization, the increasing adoption of AI and machine learning for proactive quality management, and the expansion of cloud-based solutions offering enhanced accessibility and scalability. Manufacturers will increasingly leverage QMS not just for compliance but as a strategic tool to gain a competitive edge, improve operational efficiency, and foster innovation. The market will see further consolidation and the emergence of integrated platforms that offer end-to-end quality management capabilities, positioning QMS as a cornerstone of successful medical device development and commercialization. The estimated market value is predicted to reach one hundred billion by 2033.
Medical Device Quality Management System Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Clinic
-
2. Types
- 2.1. Local
- 2.2. Cloud-based
Medical Device Quality Management System Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
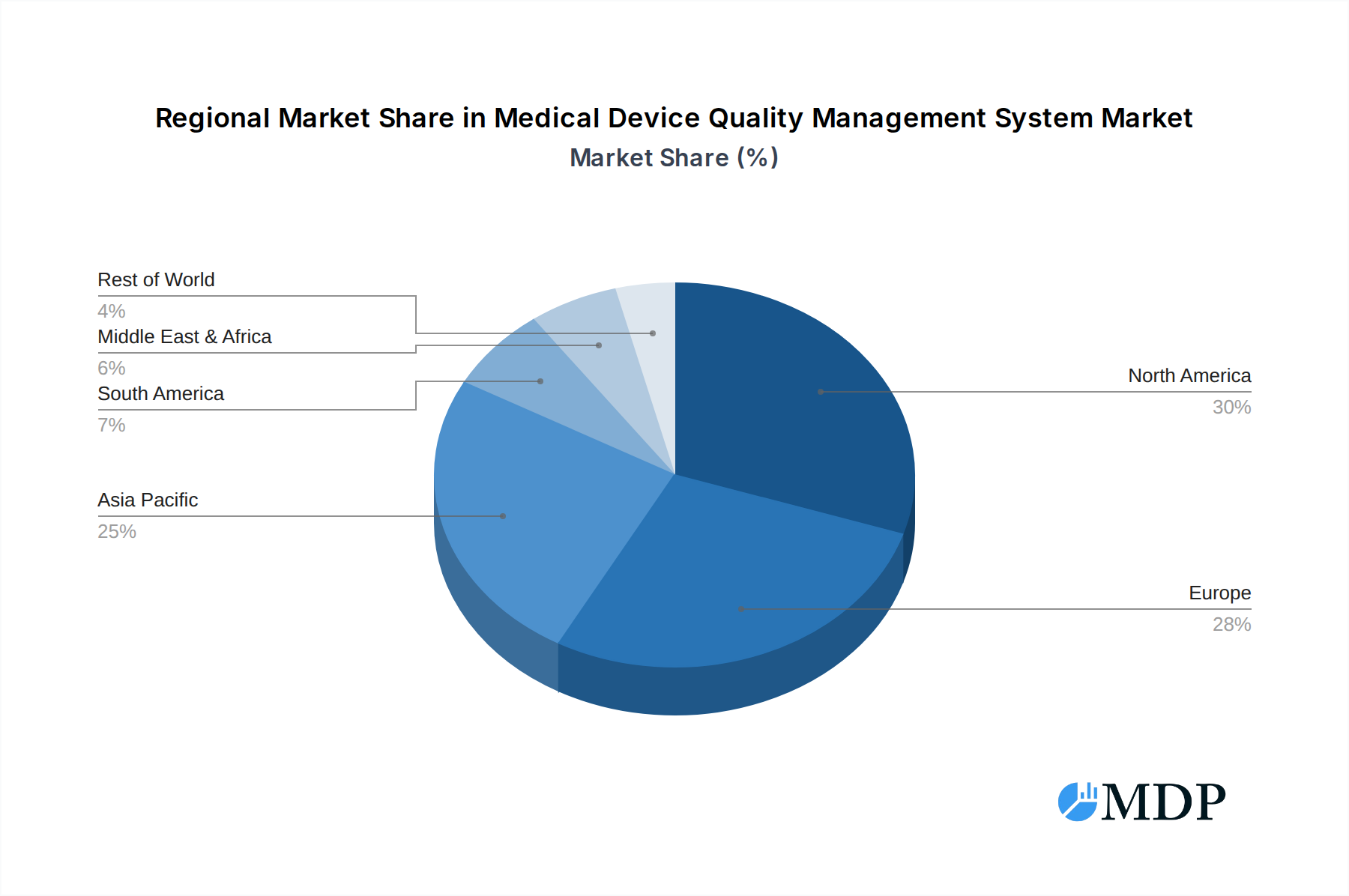
Medical Device Quality Management System Regional Market Share

Geographic Coverage of Medical Device Quality Management System
Medical Device Quality Management System REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Medical Device Quality Management System Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Clinic
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Local
- 5.2.2. Cloud-based
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Medical Device Quality Management System Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Clinic
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Local
- 6.2.2. Cloud-based
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Medical Device Quality Management System Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Clinic
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Local
- 7.2.2. Cloud-based
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Medical Device Quality Management System Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Clinic
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Local
- 8.2.2. Cloud-based
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Medical Device Quality Management System Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Clinic
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Local
- 9.2.2. Cloud-based
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Medical Device Quality Management System Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Clinic
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Local
- 10.2.2. Cloud-based
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Rootstock
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 OptiProERP
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 SYSPRO ERP
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Epicor
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Southeast Computer Solutions
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Roxtra GmbH
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 SoftExpert
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Greenlight Guru
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 uniPoint
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 MRPeasy
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Sage Software
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 ScienceSoft
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 MasterControl
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Timly
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Sterling Medical Device Company
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.1 Rootstock
List of Figures
- Figure 1: Global Medical Device Quality Management System Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Medical Device Quality Management System Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Medical Device Quality Management System Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Medical Device Quality Management System Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Medical Device Quality Management System Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Medical Device Quality Management System Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Medical Device Quality Management System Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Medical Device Quality Management System Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Medical Device Quality Management System Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Medical Device Quality Management System Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Medical Device Quality Management System Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Medical Device Quality Management System Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Medical Device Quality Management System Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Medical Device Quality Management System Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Medical Device Quality Management System Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Medical Device Quality Management System Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Medical Device Quality Management System Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Medical Device Quality Management System Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Medical Device Quality Management System Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Medical Device Quality Management System Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Medical Device Quality Management System Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Medical Device Quality Management System Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Medical Device Quality Management System Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Medical Device Quality Management System Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Medical Device Quality Management System Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Medical Device Quality Management System Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Medical Device Quality Management System Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Medical Device Quality Management System Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Medical Device Quality Management System Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Medical Device Quality Management System Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Medical Device Quality Management System Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Medical Device Quality Management System Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Medical Device Quality Management System Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Medical Device Quality Management System Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Medical Device Quality Management System Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Medical Device Quality Management System Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Medical Device Quality Management System Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Medical Device Quality Management System Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Medical Device Quality Management System Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Medical Device Quality Management System Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Medical Device Quality Management System Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Medical Device Quality Management System Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Medical Device Quality Management System Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Medical Device Quality Management System Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Medical Device Quality Management System Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Medical Device Quality Management System Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Medical Device Quality Management System Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Medical Device Quality Management System Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Medical Device Quality Management System Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Medical Device Quality Management System Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Medical Device Quality Management System?
The projected CAGR is approximately 6%.
2. Which companies are prominent players in the Medical Device Quality Management System?
Key companies in the market include Rootstock, OptiProERP, SYSPRO ERP, Epicor, Southeast Computer Solutions, Roxtra GmbH, SoftExpert, Greenlight Guru, uniPoint, MRPeasy, Sage Software, ScienceSoft, MasterControl, Timly, Sterling Medical Device Company.
3. What are the main segments of the Medical Device Quality Management System?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3350.00, USD 5025.00, and USD 6700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Medical Device Quality Management System," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Medical Device Quality Management System report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Medical Device Quality Management System?
To stay informed about further developments, trends, and reports in the Medical Device Quality Management System, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


