Key Insights
The global Intravenous (IV) fluid bags market is poised for robust expansion, projected to reach an estimated \$2596 million in 2025 and grow at a Compound Annual Growth Rate (CAGR) of 6.2% through 2033. This sustained growth is fueled by an escalating global demand for healthcare services, driven by an aging population, the increasing prevalence of chronic diseases, and a greater emphasis on preventative care and hospital-acquired infection control. Advancements in material science have led to the development of safer and more efficient IV bag solutions, with a notable shift towards flexible and semi-rigid plastic IV bags due to their superior durability, lightweight properties, and reduced risk of breakage compared to traditional glass bottles. The expanding healthcare infrastructure, particularly in emerging economies, and the rising adoption of advanced drug delivery systems further contribute to the market's upward trajectory.
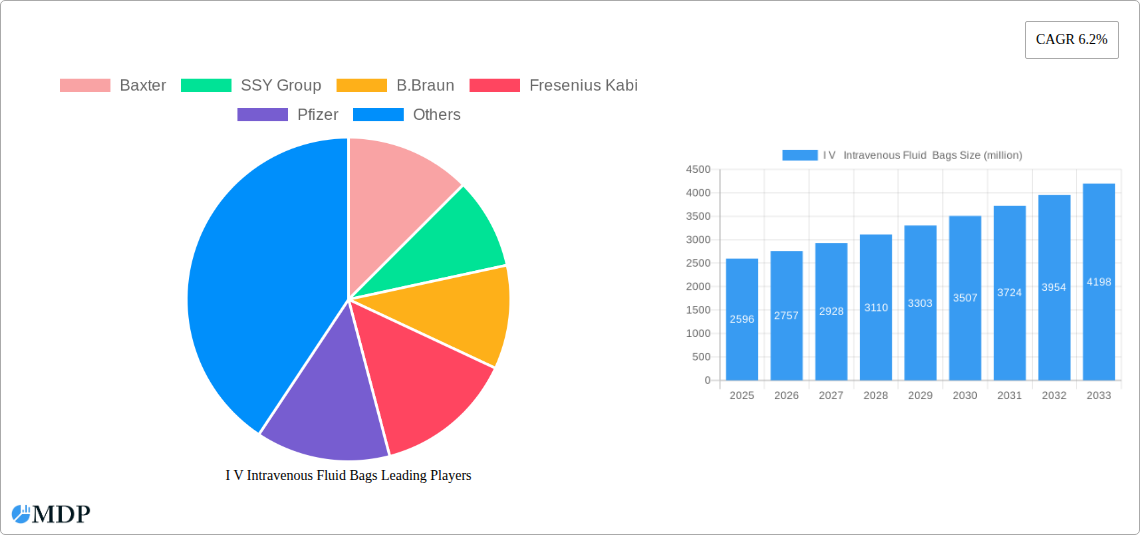
I V Intravenous Fluid Bags Market Size (In Billion)

Key market drivers include the continuous innovation in IV bag designs, such as enhanced barrier properties to maintain drug stability and the integration of safety features to prevent needlestick injuries. The growing preference for single-use, sterile IV bags minimizes the risk of cross-contamination, a critical factor in hospital settings, thereby boosting the adoption of advanced polymeric materials. While the market benefits from these positive trends, certain restraints, such as stringent regulatory approvals for new materials and devices, and the initial capital investment required for advanced manufacturing facilities, could present challenges. Nevertheless, the strong emphasis on patient safety, the increasing volume of intravenous drug administrations, and the expanding applications of IV fluids in various therapeutic areas, including oncology, critical care, and nutritional support, ensure a promising future for the IV fluid bags market.
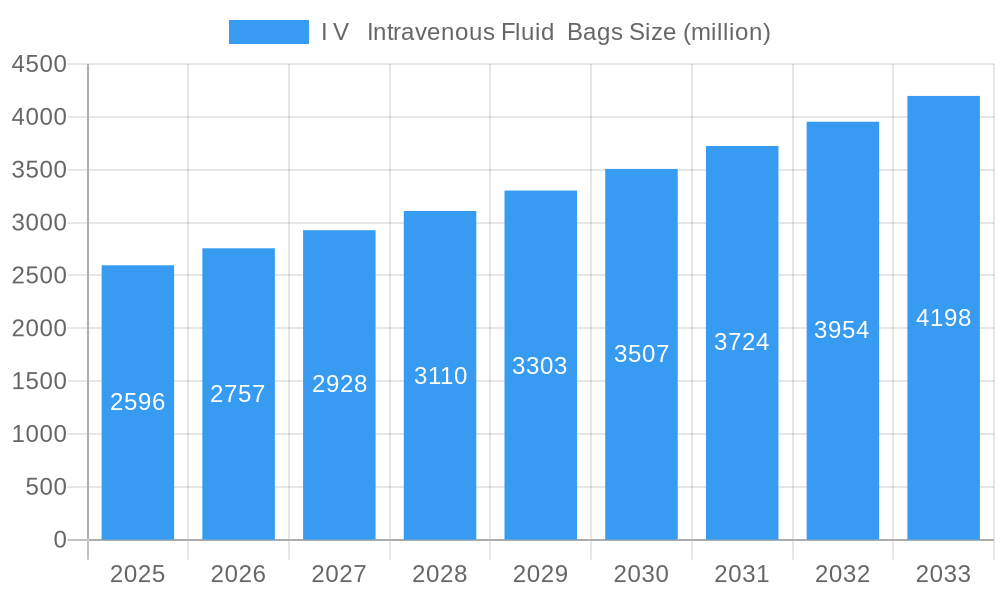
I V Intravenous Fluid Bags Company Market Share

The I V Intravenous Fluid Bags Market is poised for substantial growth and evolution. This comprehensive report delves deep into the intricate dynamics, trends, and strategic outlook of the global IV Intravenous Fluid Bags industry. Examining a study period from 2019 to 2033, with a base and estimated year of 2025 and a forecast period of 2025-2033, this analysis provides actionable insights for industry stakeholders. We cover the historical period from 2019-2024, offering a robust understanding of past performance and current market penetration, estimated at approximately 150 million units in the base year. The market for IV Intravenous Fluid Bags is projected to witness a Compound Annual Growth Rate (CAGR) of approximately 6.5% throughout the forecast period, reaching an estimated market size of over 250 million units by 2033. This report is essential for understanding market concentration, innovation drivers, regulatory frameworks, product substitutes, end-user trends, and M&A activities, alongside detailed segment analysis and leading player profiling.
I V Intravenous Fluid Bags Market Dynamics & Concentration
The I V Intravenous Fluid Bags market exhibits a moderately concentrated landscape, with a few key global players holding significant market share, estimated to be around 60% combined. Innovation is primarily driven by the demand for enhanced patient safety, reduced contamination risks, and improved drug compatibility. Advancements in materials science are leading to the development of novel flexible plastic IV bags with superior barrier properties and reduced leaching. Regulatory frameworks, particularly those from the FDA and EMA, play a crucial role in shaping market entry and product development, with stringent approval processes requiring extensive clinical validation. Product substitutes, while limited in the core IV fluid delivery segment, include pre-mixed drug solutions and alternative delivery methods for specific therapies, though traditional IV bags remain dominant due to cost-effectiveness and broad applicability. End-user trends are shifting towards home healthcare settings, increasing the demand for user-friendly and sterile IV bag solutions. Mergers and acquisitions (M&A) activity is moderate, with an estimated 5-7 major deals annually, often aimed at expanding product portfolios or geographical reach. For instance, a recent acquisition in 2023 involved a specialized manufacturer of semi-rigid IV bags, signifying a strategic move to diversify offerings, with deal values ranging from 10 million to 50 million. The market share of leading players is dynamic, with top companies like Baxter and B. Braun holding approximately 15% and 12% respectively.
I V Intravenous Fluid Bags Industry Trends & Analysis
The I V Intravenous Fluid Bags industry is experiencing robust growth, fueled by several interconnected trends and an increasing global demand for healthcare services. A primary growth driver is the expanding geriatric population worldwide, which necessitates more frequent and prolonged medical interventions, including intravenous fluid administration. Coupled with this is the rising prevalence of chronic diseases such as diabetes, cardiovascular disorders, and cancer, all of which often require regular IV therapy. The increasing number of surgical procedures, both elective and emergency, further contributes to the demand for IV fluid bags. Technological advancements in IV bag design and materials are also playing a pivotal role. The shift from traditional glass bottles to more convenient and safer flexible plastic IV bags, such as PVC and non-PVC alternatives, is a significant trend. These advancements aim to reduce breakage, improve handling, minimize waste, and enhance sterility. Furthermore, the development of specialized IV bags designed for specific drug formulations, including light-sensitive or oxygen-sensitive medications, is opening up new market niches. The increasing adoption of smart IV bags, which can integrate sensors for monitoring flow rates or fluid levels, represents a nascent but promising area of innovation. The expansion of healthcare infrastructure, particularly in emerging economies, is creating substantial market opportunities. Governments and private organizations are investing heavily in building and upgrading hospitals and clinics, directly impacting the demand for essential medical supplies like IV fluid bags. The shift towards ambulatory care and home infusion therapy, driven by cost-effectiveness and patient convenience, is another key trend, necessitating portable and easy-to-use IV bag systems. Competitive dynamics are characterized by a mix of large, established global manufacturers and smaller, specialized regional players. Companies are competing on product quality, innovation, regulatory compliance, supply chain efficiency, and pricing. The market penetration of advanced IV bag technologies is steadily increasing, although cost remains a consideration for wider adoption, especially in price-sensitive markets. The overall market penetration of IV fluid bags is estimated at 85% across developed healthcare systems, with significant room for growth in developing regions, projected to reach over 90% by 2033. The global market size for I V Intravenous Fluid Bags is estimated to be around 190 million units in the base year 2025, with a projected market value exceeding 12 billion.
Leading Markets & Segments in I V Intravenous Fluid Bags
North America currently stands as the dominant region in the global I V Intravenous Fluid Bags market, driven by its highly developed healthcare infrastructure, significant per capita healthcare expenditure, and a large aging population. The United States, in particular, leads this regional dominance due to extensive hospital networks, advanced medical technologies, and a strong emphasis on patient safety and quality of care. Within the Application segment, Hospitals represent the largest and most influential market. This dominance is attributable to the high volume of patients requiring intravenous therapies, from routine hydration and electrolyte replacement to complex drug administration in critical care units. Hospitals are equipped with the necessary infrastructure, trained personnel, and procurement channels to consistently utilize a vast quantity of IV fluid bags. The segment's growth is further propelled by government healthcare initiatives and insurance coverage that support a wide range of inpatient treatments. The second-largest application segment is Clinics, which are experiencing growth due to the increasing trend of outpatient procedures and specialized treatments being administered in less intensive settings.
In terms of Type, Flex Plastic IV Bags command the most significant market share. This is a direct result of their inherent advantages over traditional glass bottles: superior safety profile (reduced risk of breakage), lightweight nature, ease of handling and storage, and cost-effectiveness in manufacturing and transportation. Flexible plastic IV bags are highly adaptable to various IV fluid formulations and are compatible with automated infusion systems, making them the preferred choice for most healthcare settings. Semi-rigid IV Bags are also gaining traction, particularly for specific applications requiring enhanced rigidity or protection for sensitive contents. Their market share, while smaller than flex plastic, is growing due to advancements in material science and their suitability for certain pharmaceutical products. Glass Bottles, though historically dominant, now represent a smaller and declining segment. Their use is largely confined to specific niche applications or older established protocols where replacement is not economically feasible. The key drivers for the dominance of Hospitals and Flex Plastic IV Bags include:
- Economic Policies: Robust healthcare spending and favorable reimbursement policies in North America and other developed regions directly stimulate demand for medical supplies like IV bags.
- Infrastructure: The widespread presence of well-equipped hospitals and clinics, particularly in developed nations, ensures a consistent demand.
- Technological Adoption: The rapid integration of advanced infusion technologies and the preference for user-friendly, safe, and efficient medical devices favor flexible plastic IV bags.
- Regulatory Compliance: Stringent quality and safety regulations often mandate the use of materials and designs that are most effectively realized in flexible plastic formats.
- Patient Demographics: The aging population and rising chronic disease burden necessitate extensive use of IV therapies, with hospitals being the primary point of care.
The projected growth in emerging economies, coupled with increasing healthcare investments, is expected to further solidify the dominance of these segments while also presenting opportunities for market expansion in other regions and types.
I V Intravenous Fluid Bags Product Developments
Recent product developments in the I V Intravenous Fluid Bags market focus on enhancing patient safety, improving drug stability, and streamlining administration processes. Innovations include the introduction of advanced non-PVC plastic formulations that reduce the risk of plasticizer leaching, addressing concerns over potential patient exposure. Furthermore, specialized IV bags with integrated drug-delivery systems, such as pre-filled syringes designed for direct connection, are emerging, offering greater convenience and reducing medication errors. Companies are also investing in developing IV bags with improved barrier properties to protect sensitive medications from light and oxygen degradation, thereby extending shelf life and maintaining therapeutic efficacy. The competitive advantage of these developments lies in their ability to meet evolving clinical needs and stringent regulatory requirements, offering safer, more efficient, and reliable solutions for intravenous therapy.
Key Drivers of I V Intravenous Fluid Bags Growth
The growth of the I V Intravenous Fluid Bags market is propelled by several key factors. Foremost is the increasing global prevalence of chronic diseases, such as cardiovascular disorders, diabetes, and cancer, which often require long-term intravenous therapies. The expanding elderly population, a demographic that typically necessitates more frequent medical interventions, also significantly boosts demand. Technological advancements in IV bag materials and design, leading to enhanced safety, reduced contamination risks, and improved drug compatibility, are driving adoption rates. Furthermore, the growing healthcare expenditure worldwide, particularly in emerging economies, coupled with the expansion of healthcare infrastructure, is creating substantial market opportunities. Finally, the increasing shift towards outpatient and home healthcare settings is fueling demand for convenient and easy-to-use IV bag solutions.
Challenges in the I V Intravenous Fluid Bags Market
Despite robust growth, the I V Intravenous Fluid Bags market faces several challenges. Stringent regulatory hurdles and lengthy approval processes for new materials and product designs can slow down innovation and market entry, with compliance costs estimated to be in the millions for each new product line. Price pressures and competition from generic manufacturers, especially in high-volume segments, can impact profit margins. Supply chain disruptions, as seen in recent global events, can affect the availability and cost of raw materials, such as specialized plastics and chemicals, potentially leading to an estimated 10-15% increase in manufacturing costs during such periods. Concerns over environmental impact and the disposal of medical waste are also prompting a demand for more sustainable packaging solutions.
Emerging Opportunities in I V Intravenous Fluid Bags
Emerging opportunities in the I V Intravenous Fluid Bags market are largely driven by technological innovation and strategic market expansion. The development and adoption of smart IV bags equipped with sensors for real-time monitoring of fluid levels, flow rates, and drug infusion parameters present a significant growth avenue. These technologies enhance patient safety and provide valuable data for healthcare providers. Furthermore, the increasing demand for specialized IV bags for targeted drug delivery and personalized medicine is opening up niche markets. Strategic partnerships between IV bag manufacturers and pharmaceutical companies to develop co-packaged or integrated drug-device solutions are also creating synergistic growth opportunities. Expansion into emerging economies with developing healthcare systems offers substantial untapped market potential, requiring tailored product offerings and distribution strategies.
Leading Players in the I V Intravenous Fluid Bags Sector
- Baxter
- SSY Group
- B.Braun
- Fresenius Kabi
- Pfizer
- Otsuka
- Cisen Pharmaceutical
- Renolit
- Technoflex
- Huaren Pharmaceutical
- CR Double-Crane
- ICU Medical
- Pharmaceutical Solutions Industry Ltd
- Vioser
- Sippex
- Well Pharma
- Zhejiang CHIMIN
Key Milestones in I V Intravenous Fluid Bags Industry
- 2019: Introduction of advanced non-PVC flexible IV bags by major manufacturers, addressing concerns about plasticizer leaching and enhancing patient safety.
- 2020: Heightened demand for IV fluid bags due to the global COVID-19 pandemic, leading to increased production and supply chain focus.
- 2021: Regulatory approvals for novel multi-layer plastic films in IV bags, offering improved barrier properties and extended shelf life for sensitive medications.
- 2022: Increased M&A activity as larger companies sought to consolidate market share and expand their product portfolios, with several deals exceeding 50 million.
- 2023: Growing interest and initial product launches for "smart" IV bags with integrated sensor technology for enhanced infusion monitoring.
- 2024: Significant investments in expanding manufacturing capacities by key players to meet projected long-term demand.
Strategic Outlook for I V Intravenous Fluid Bags Market
The strategic outlook for the I V Intravenous Fluid Bags market is characterized by continued innovation, market consolidation, and expansion into high-growth regions. Key growth accelerators include the ongoing development of advanced materials for enhanced drug compatibility and safety, alongside the integration of digital technologies for smarter infusion management. Companies will focus on optimizing supply chains to ensure resilience and cost-effectiveness, particularly in light of global economic fluctuations. Strategic partnerships and collaborations will be crucial for co-developing innovative drug-device combinations and penetrating new therapeutic areas. Furthermore, market expansion in emerging economies, driven by increasing healthcare access and investment, will remain a primary focus, necessitating localized strategies and product adaptations.
I V Intravenous Fluid Bags Segmentation
-
1. Application
- 1.1. Hospitals
- 1.2. Clinics
-
2. Type
- 2.1. Flex Plastic IV Bags
- 2.2. Semi-rigid IV Bags
- 2.3. Glass Bottles
I V Intravenous Fluid Bags Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
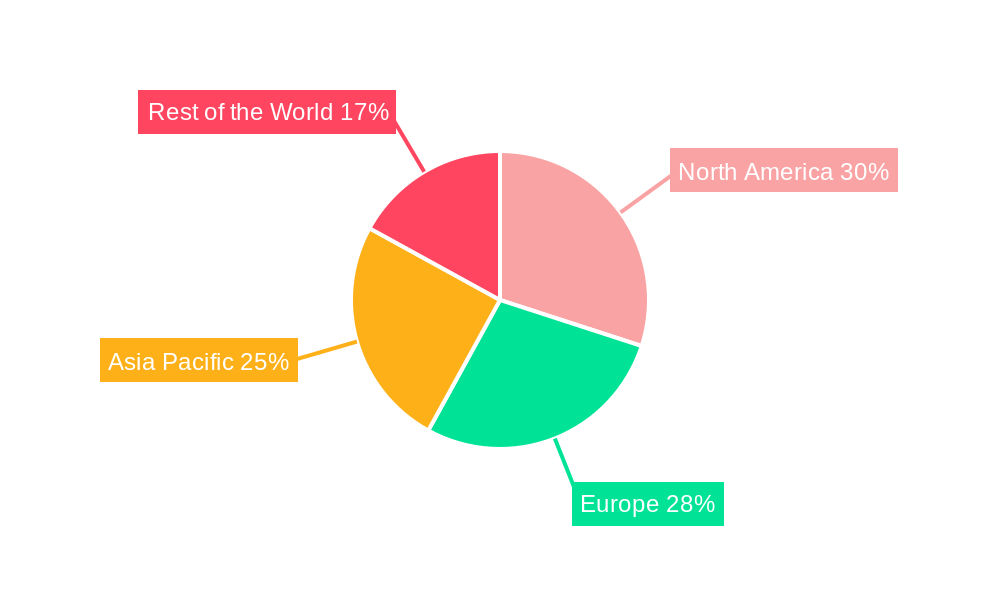
I V Intravenous Fluid Bags Regional Market Share

Geographic Coverage of I V Intravenous Fluid Bags
I V Intravenous Fluid Bags REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.2% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global I V Intravenous Fluid Bags Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals
- 5.1.2. Clinics
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Flex Plastic IV Bags
- 5.2.2. Semi-rigid IV Bags
- 5.2.3. Glass Bottles
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America I V Intravenous Fluid Bags Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospitals
- 6.1.2. Clinics
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Flex Plastic IV Bags
- 6.2.2. Semi-rigid IV Bags
- 6.2.3. Glass Bottles
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America I V Intravenous Fluid Bags Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospitals
- 7.1.2. Clinics
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Flex Plastic IV Bags
- 7.2.2. Semi-rigid IV Bags
- 7.2.3. Glass Bottles
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe I V Intravenous Fluid Bags Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospitals
- 8.1.2. Clinics
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Flex Plastic IV Bags
- 8.2.2. Semi-rigid IV Bags
- 8.2.3. Glass Bottles
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa I V Intravenous Fluid Bags Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospitals
- 9.1.2. Clinics
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Flex Plastic IV Bags
- 9.2.2. Semi-rigid IV Bags
- 9.2.3. Glass Bottles
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific I V Intravenous Fluid Bags Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospitals
- 10.1.2. Clinics
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Flex Plastic IV Bags
- 10.2.2. Semi-rigid IV Bags
- 10.2.3. Glass Bottles
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Baxter
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 SSY Group
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 B.Braun
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Fresenius Kabi
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Pfizer
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Otsuka
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Cisen Pharmaceutical
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Renolit
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Technoflex
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Huaren Pharmaceutical
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 CR Double-Crane
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 ICU Medical
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Pharmaceutical Solutions Industry Ltd
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Vioser
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Sippex
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Well Pharma
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 Zhejiang CHIMIN
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.1 Baxter
List of Figures
- Figure 1: Global I V Intravenous Fluid Bags Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America I V Intravenous Fluid Bags Revenue (million), by Application 2025 & 2033
- Figure 3: North America I V Intravenous Fluid Bags Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America I V Intravenous Fluid Bags Revenue (million), by Type 2025 & 2033
- Figure 5: North America I V Intravenous Fluid Bags Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America I V Intravenous Fluid Bags Revenue (million), by Country 2025 & 2033
- Figure 7: North America I V Intravenous Fluid Bags Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America I V Intravenous Fluid Bags Revenue (million), by Application 2025 & 2033
- Figure 9: South America I V Intravenous Fluid Bags Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America I V Intravenous Fluid Bags Revenue (million), by Type 2025 & 2033
- Figure 11: South America I V Intravenous Fluid Bags Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America I V Intravenous Fluid Bags Revenue (million), by Country 2025 & 2033
- Figure 13: South America I V Intravenous Fluid Bags Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe I V Intravenous Fluid Bags Revenue (million), by Application 2025 & 2033
- Figure 15: Europe I V Intravenous Fluid Bags Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe I V Intravenous Fluid Bags Revenue (million), by Type 2025 & 2033
- Figure 17: Europe I V Intravenous Fluid Bags Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe I V Intravenous Fluid Bags Revenue (million), by Country 2025 & 2033
- Figure 19: Europe I V Intravenous Fluid Bags Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa I V Intravenous Fluid Bags Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa I V Intravenous Fluid Bags Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa I V Intravenous Fluid Bags Revenue (million), by Type 2025 & 2033
- Figure 23: Middle East & Africa I V Intravenous Fluid Bags Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa I V Intravenous Fluid Bags Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa I V Intravenous Fluid Bags Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific I V Intravenous Fluid Bags Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific I V Intravenous Fluid Bags Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific I V Intravenous Fluid Bags Revenue (million), by Type 2025 & 2033
- Figure 29: Asia Pacific I V Intravenous Fluid Bags Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific I V Intravenous Fluid Bags Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific I V Intravenous Fluid Bags Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global I V Intravenous Fluid Bags Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global I V Intravenous Fluid Bags Revenue million Forecast, by Type 2020 & 2033
- Table 3: Global I V Intravenous Fluid Bags Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global I V Intravenous Fluid Bags Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global I V Intravenous Fluid Bags Revenue million Forecast, by Type 2020 & 2033
- Table 6: Global I V Intravenous Fluid Bags Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global I V Intravenous Fluid Bags Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global I V Intravenous Fluid Bags Revenue million Forecast, by Type 2020 & 2033
- Table 12: Global I V Intravenous Fluid Bags Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global I V Intravenous Fluid Bags Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global I V Intravenous Fluid Bags Revenue million Forecast, by Type 2020 & 2033
- Table 18: Global I V Intravenous Fluid Bags Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global I V Intravenous Fluid Bags Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global I V Intravenous Fluid Bags Revenue million Forecast, by Type 2020 & 2033
- Table 30: Global I V Intravenous Fluid Bags Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global I V Intravenous Fluid Bags Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global I V Intravenous Fluid Bags Revenue million Forecast, by Type 2020 & 2033
- Table 39: Global I V Intravenous Fluid Bags Revenue million Forecast, by Country 2020 & 2033
- Table 40: China I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific I V Intravenous Fluid Bags Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the I V Intravenous Fluid Bags?
The projected CAGR is approximately 6.2%.
2. Which companies are prominent players in the I V Intravenous Fluid Bags?
Key companies in the market include Baxter, SSY Group, B.Braun, Fresenius Kabi, Pfizer, Otsuka, Cisen Pharmaceutical, Renolit, Technoflex, Huaren Pharmaceutical, CR Double-Crane, ICU Medical, Pharmaceutical Solutions Industry Ltd, Vioser, Sippex, Well Pharma, Zhejiang CHIMIN.
3. What are the main segments of the I V Intravenous Fluid Bags?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 2596 million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "I V Intravenous Fluid Bags," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the I V Intravenous Fluid Bags report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the I V Intravenous Fluid Bags?
To stay informed about further developments, trends, and reports in the I V Intravenous Fluid Bags, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


