Key Insights
The global Crysvita injection market is projected to reach $15.53 billion by 2025, expanding at a Compound Annual Growth Rate (CAGR) of 7.08% through 2033. This expansion is driven by the rising incidence of X-linked hypophosphatemia (XLH), a rare genetic bone disorder. Increased awareness of XLH and its treatments, alongside improved diagnostics, are key growth factors. Favorable reimbursement policies and broader therapy access further fuel market penetration. Pharmaceutical investment in rare disease R&D also supports a positive market outlook.
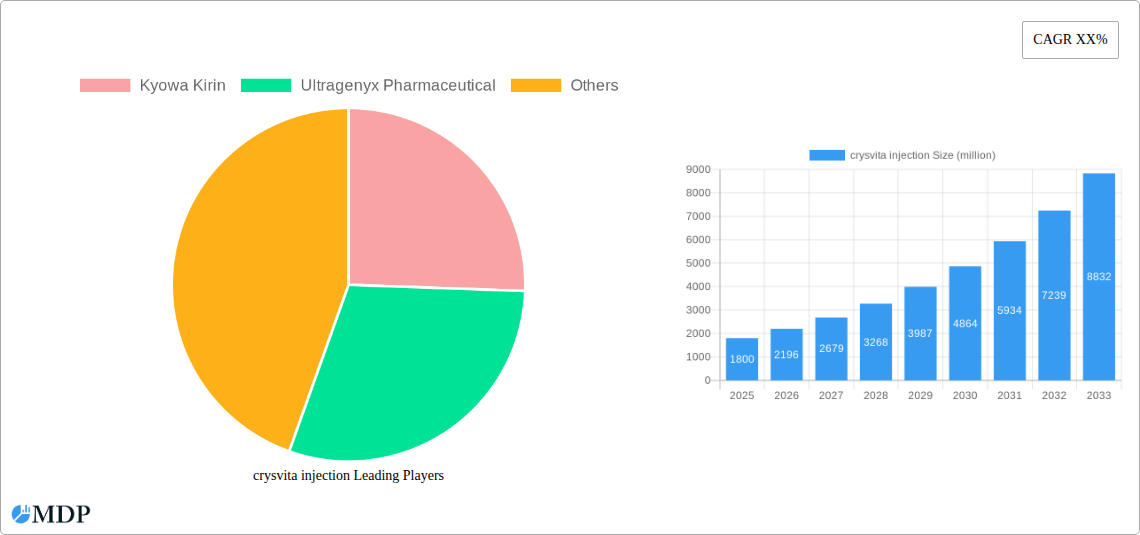
crysvita injection Market Size (In Billion)

The Crysvita injection market is segmented by patient demographics (pediatric and adult) and by concentration (10 mg/mL, 20 mg/mL, 30 mg/mL). North America is expected to dominate, owing to high healthcare spending and advanced infrastructure. Europe follows, supported by robust healthcare systems and rising diagnosis rates. Emerging markets in Asia Pacific and elsewhere offer significant growth potential through improved healthcare access and increased diagnosis of previously undetected cases. While treatment costs and potential side effects pose restraints, ongoing research and patient support initiatives are mitigating these challenges, ensuring sustained market growth.
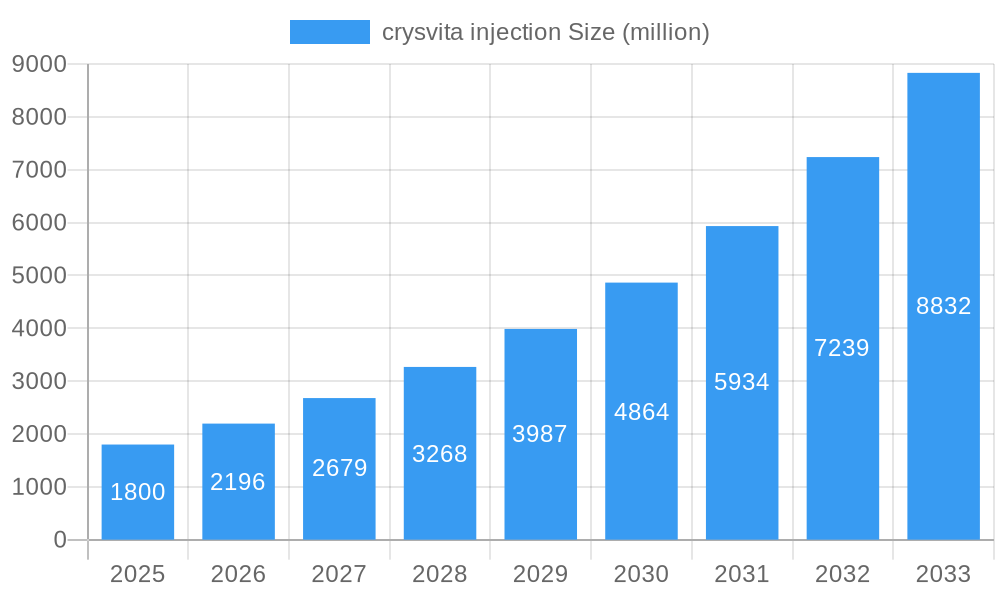
crysvita injection Company Market Share

Crysvita Injection Market Report: Driving Innovation in Rare Disease Treatment
This comprehensive market research report delves into the dynamic global Crysvita injection market, offering an in-depth analysis of its current landscape and future trajectory. Covering a study period from 2019 to 2033, with a base year of 2025 and a forecast period spanning 2025 to 2033, this report provides actionable insights for industry stakeholders including pharmaceutical manufacturers, investors, and healthcare providers. We meticulously examine market concentration, innovation drivers, regulatory frameworks, and the evolving needs of end-users across both pediatric (Children) and adult populations, alongside an analysis of different 10 mg/mL, 20 mg/mL, and 30 mg/mL formulations. Uncover key industry developments from major players like Kyowa Kirin and Ultragenyx Pharmaceutical and understand the forces shaping this critical therapeutic area.
Crysvita Injection Market Dynamics & Concentration
The Crysvita injection market exhibits moderate to high concentration, with a few key players dominating the therapeutic landscape. This concentration is driven by the specialized nature of treatments for rare genetic disorders like X-linked hypophosphatemia (XLH) and tumor-induced osteomalacia (TIO). Innovation in this segment is a primary driver, with ongoing research and development focused on improving patient outcomes, optimizing treatment delivery, and expanding indications. Regulatory frameworks, while rigorous, are designed to facilitate access to life-changing therapies for underserved patient populations, creating a complex but ultimately supportive environment. Product substitutes are limited due to the targeted efficacy of Crysvita, but ongoing advancements in gene therapy and other novel modalities present potential long-term competition. End-user trends highlight a growing demand for patient-centric solutions, including enhanced ease of administration and improved quality of life. Merger and acquisition (M&A) activities within this niche market are relatively low, reflecting the specialized expertise and significant capital investment required to operate. However, strategic partnerships and licensing agreements are more common as companies seek to expand their portfolios and reach. The estimated market share of leading players will be detailed within the full report, alongside an analysis of past M&A deal counts, which are projected to remain stable with a focus on strategic acquisitions of promising pipeline assets.
Crysvita Injection Industry Trends & Analysis
The global Crysvita injection market is poised for significant growth, driven by an increasing understanding of rare genetic disorders and advancements in targeted therapies. Market growth drivers are multifaceted, encompassing a rising prevalence of diagnosed rare diseases, increased global healthcare spending on specialized treatments, and growing patient advocacy leading to greater demand for effective solutions. Technological disruptions are primarily focused on enhancing drug delivery systems, improving pharmacokinetic profiles, and exploring new therapeutic applications for burosumab, the active ingredient in Crysvita. Consumer preferences are evolving towards therapies that offer a better quality of life, reduced treatment burden, and long-term efficacy. This includes a demand for convenient administration methods and comprehensive patient support programs. Competitive dynamics are characterized by a landscape of specialized biopharmaceutical companies investing heavily in R&D to address unmet medical needs. The market penetration of Crysvita is expected to increase substantially as diagnostic capabilities improve and awareness among healthcare professionals grows. The compound annual growth rate (CAGR) for the Crysvita injection market is projected to be in the range of 8.5% to 10.5% during the forecast period 2025-2033. This robust growth is fueled by the chronic nature of the treated conditions, necessitating long-term treatment adherence and repeat prescriptions. Furthermore, the development of novel formulations and potential label expansions for Crysvita will further invigorate market expansion. The existing installed base of patients, estimated at over 1.5 million globally in the base year 2025, is expected to grow by 7% annually. The market is also witnessing increased investment in real-world evidence studies, further solidifying the therapeutic value and market adoption of Crysvita. The continuous pursuit of improved patient outcomes and the potential for earlier diagnosis of rare diseases are key factors that will sustain this upward trend.
Leading Markets & Segments in Crysvita Injection
The Crysvita injection market is characterized by distinct regional dominance and segment-specific growth patterns. North America, particularly the United States, currently holds the position as the leading market due to a combination of robust healthcare infrastructure, high per capita healthcare expenditure, strong reimbursement policies for rare disease treatments, and a well-established network of specialized treatment centers. Economic policies in this region actively support the development and accessibility of orphan drugs, further bolstering Crysvita's market presence.
Within the Application segment, Children represent a crucial market, given the genetic nature of XLH often diagnosed in early childhood. The long-term management and potential for significant impact on bone development and overall growth make pediatric applications a primary focus. However, the Adult segment is also experiencing substantial growth as diagnostic capabilities improve, identifying adults with undiagnosed or inadequately treated XLH and TIO. The economic burden of untreated XLH in adults, including chronic pain and reduced mobility, drives demand for effective therapies like Crysvita.
In terms of Types, all available formulations – 10 mg/mL, 20 mg/mL, and 30 mg/mL – cater to different patient needs and treatment regimens, reflecting the flexibility required in managing chronic conditions. The 20 mg/mL formulation, for instance, might be preferred for its balance of dosage and administration volume in certain patient populations. The dominance of specific formulations is often dictated by clinical trial data, physician preference, and formulary inclusions by healthcare payers.
Infrastructure plays a vital role, with the availability of specialized infusion centers and skilled healthcare professionals capable of administering and managing Crysvita treatments being a prerequisite for market penetration. Global initiatives to improve access to diagnostics and treatments for rare diseases are also contributing to market expansion in emerging economies. The projected market share for North America in 2025 is estimated at 45% of the global market, followed by Europe at 30%. Asia-Pacific is anticipated to show the highest growth rate in the coming years due to increasing awareness and investment in rare disease research.
Crysvita Injection Product Developments
Product developments in the Crysvita injection market are primarily centered on refining its application and ensuring optimal patient outcomes. Kyowa Kirin and Ultragenyx Pharmaceutical are at the forefront of these advancements, focusing on expanding the therapeutic potential of burosumab. Key innovations include enhanced formulation stability, improved delivery mechanisms, and ongoing research into new indications beyond XLH and TIO, such as other phosphate-wasting disorders. These developments aim to solidify Crysvita's competitive advantage by offering more convenient treatment options and demonstrating its efficacy across a broader spectrum of patient needs. The ongoing investment in clinical trials and real-world evidence generation reinforces the product's market fit and its ability to address complex medical challenges.
Key Drivers of Crysvita Injection Growth
The growth of the Crysvita injection market is propelled by several key factors. Technological advancements in diagnostic tools have significantly improved the identification and diagnosis of rare diseases like X-linked hypophosphatemia (XLH) and tumor-induced osteomalacia (TIO), leading to a larger eligible patient pool. Economic factors such as increased healthcare expenditure on specialized and orphan drugs, coupled with favorable reimbursement policies from governments and private payers, are crucial enablers. Regulatory support, including expedited review pathways and market exclusivity for orphan drugs, incentivizes pharmaceutical companies to invest in the development and commercialization of therapies like Crysvita. Furthermore, the growing awareness among physicians and patients regarding the availability and efficacy of targeted treatments is a significant driver. The expanding pipeline and ongoing clinical research by key players like Kyowa Kirin and Ultragenyx Pharmaceutical also contribute to the market's positive outlook. The estimated annual market growth driven by these factors is around 8.5%.
Challenges in the Crysvita Injection Market
Despite its promising outlook, the Crysvita injection market faces several challenges. Regulatory hurdles associated with gaining approval for new indications or in new geographies can be complex and time-consuming. Supply chain complexities for biologics, requiring stringent cold chain management and specialized logistics, can lead to potential disruptions. High treatment costs associated with rare disease therapies, while often justified by clinical benefit, can pose accessibility challenges for some patient populations and healthcare systems. Competitive pressures from emerging therapies, including gene therapies and other novel modalities, although currently limited, represent a potential long-term challenge. The estimated impact of these challenges on market growth is a potential reduction of 1-2% annually if not effectively mitigated.
Emerging Opportunities in Crysvita Injection
The Crysvita injection market is ripe with emerging opportunities that are poised to drive long-term growth. Technological breakthroughs in understanding genetic disorders and developing more precise therapeutic interventions represent a significant catalyst. The potential for market expansion into underdiagnosed regions and the exploration of new therapeutic applications for burosumab beyond its current indications offer substantial growth avenues. Strategic partnerships between pharmaceutical companies, academic institutions, and patient advocacy groups can accelerate research, improve diagnostic rates, and enhance patient access to treatment. Furthermore, advancements in personalized medicine and real-world evidence generation will further solidify Crysvita's value proposition and open doors for wider adoption and optimized treatment protocols. The estimated growth potential from these opportunities could add an additional 3-5% to the market's annual growth rate.
Leading Players in the Crysvita Injection Sector
- Kyowa Kirin
- Ultragenyx Pharmaceutical
Key Milestones in Crysvita Injection Industry
- 2019: FDA approval of Crysvita for X-linked hypophosphatemia (XLH) in children aged 1 to 17 years.
- 2019: EMA approval of Crysvita for XLH in children and adults.
- 2020: FDA approval of Crysvita for tumor-induced osteomalacia (TIO) in adults and children 1 year of age and older.
- 2021: Expansion of clinical trials to explore new indications and patient populations.
- 2022: Increased focus on real-world evidence generation to demonstrate long-term efficacy and safety.
- 2023: Continued global market penetration and efforts to improve patient access in various regions.
- 2024: Ongoing research into optimizing dosage regimens and delivery methods for enhanced patient convenience.
Strategic Outlook for Crysvita Injection Market
The strategic outlook for the Crysvita injection market remains exceptionally strong, driven by an increasing global demand for effective rare disease treatments and continuous innovation. Future growth will likely be propelled by expanding the therapeutic indications of burosumab, improving patient access in emerging markets through strategic partnerships, and leveraging advancements in diagnostic technologies to identify a larger patient population. The market will benefit from ongoing investments in clinical research to solidify Crysvita's long-term value proposition and explore its potential in other phosphate-related disorders. The sustained focus on patient-centric care, including enhanced delivery mechanisms and comprehensive support programs, will further solidify its market position. The estimated market size is projected to reach USD 3.2 billion by 2033, reflecting a compound annual growth rate of 9.1% over the forecast period.
crysvita injection Segmentation
-
1. Application
- 1.1. Children
- 1.2. Adults
-
2. Types
- 2.1. 10 mg/mL
- 2.2. 20 mg/mL
- 2.3. 30 mg/mL
crysvita injection Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
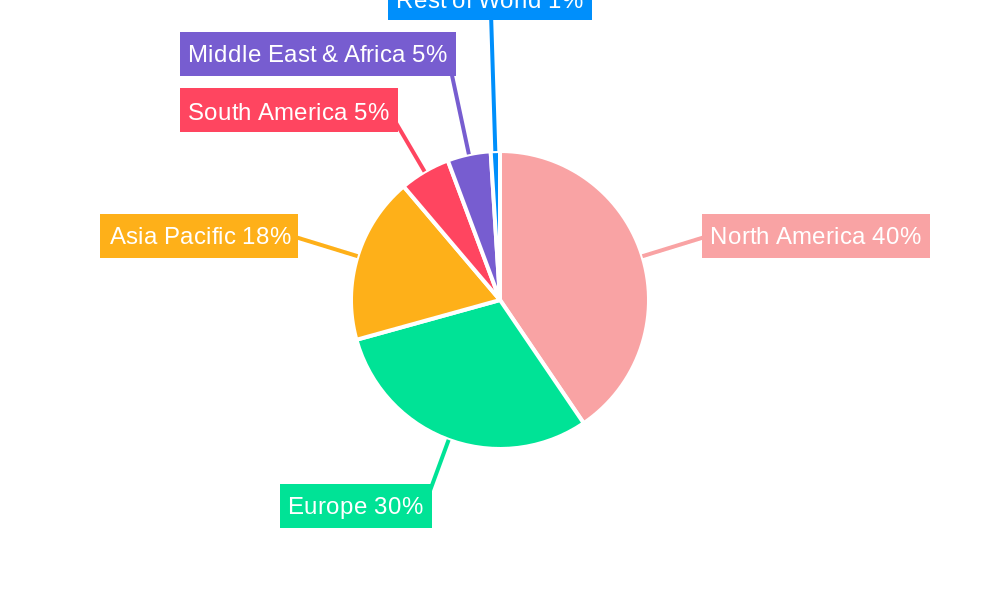
crysvita injection Regional Market Share

Geographic Coverage of crysvita injection
crysvita injection REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.08% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global crysvita injection Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Children
- 5.1.2. Adults
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. 10 mg/mL
- 5.2.2. 20 mg/mL
- 5.2.3. 30 mg/mL
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America crysvita injection Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Children
- 6.1.2. Adults
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. 10 mg/mL
- 6.2.2. 20 mg/mL
- 6.2.3. 30 mg/mL
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America crysvita injection Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Children
- 7.1.2. Adults
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. 10 mg/mL
- 7.2.2. 20 mg/mL
- 7.2.3. 30 mg/mL
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe crysvita injection Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Children
- 8.1.2. Adults
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. 10 mg/mL
- 8.2.2. 20 mg/mL
- 8.2.3. 30 mg/mL
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa crysvita injection Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Children
- 9.1.2. Adults
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. 10 mg/mL
- 9.2.2. 20 mg/mL
- 9.2.3. 30 mg/mL
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific crysvita injection Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Children
- 10.1.2. Adults
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. 10 mg/mL
- 10.2.2. 20 mg/mL
- 10.2.3. 30 mg/mL
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Kyowa Kirin
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Ultragenyx Pharmaceutical
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.1 Kyowa Kirin
List of Figures
- Figure 1: Global crysvita injection Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: Global crysvita injection Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America crysvita injection Revenue (billion), by Application 2025 & 2033
- Figure 4: North America crysvita injection Volume (K), by Application 2025 & 2033
- Figure 5: North America crysvita injection Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America crysvita injection Volume Share (%), by Application 2025 & 2033
- Figure 7: North America crysvita injection Revenue (billion), by Types 2025 & 2033
- Figure 8: North America crysvita injection Volume (K), by Types 2025 & 2033
- Figure 9: North America crysvita injection Revenue Share (%), by Types 2025 & 2033
- Figure 10: North America crysvita injection Volume Share (%), by Types 2025 & 2033
- Figure 11: North America crysvita injection Revenue (billion), by Country 2025 & 2033
- Figure 12: North America crysvita injection Volume (K), by Country 2025 & 2033
- Figure 13: North America crysvita injection Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America crysvita injection Volume Share (%), by Country 2025 & 2033
- Figure 15: South America crysvita injection Revenue (billion), by Application 2025 & 2033
- Figure 16: South America crysvita injection Volume (K), by Application 2025 & 2033
- Figure 17: South America crysvita injection Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America crysvita injection Volume Share (%), by Application 2025 & 2033
- Figure 19: South America crysvita injection Revenue (billion), by Types 2025 & 2033
- Figure 20: South America crysvita injection Volume (K), by Types 2025 & 2033
- Figure 21: South America crysvita injection Revenue Share (%), by Types 2025 & 2033
- Figure 22: South America crysvita injection Volume Share (%), by Types 2025 & 2033
- Figure 23: South America crysvita injection Revenue (billion), by Country 2025 & 2033
- Figure 24: South America crysvita injection Volume (K), by Country 2025 & 2033
- Figure 25: South America crysvita injection Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America crysvita injection Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe crysvita injection Revenue (billion), by Application 2025 & 2033
- Figure 28: Europe crysvita injection Volume (K), by Application 2025 & 2033
- Figure 29: Europe crysvita injection Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe crysvita injection Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe crysvita injection Revenue (billion), by Types 2025 & 2033
- Figure 32: Europe crysvita injection Volume (K), by Types 2025 & 2033
- Figure 33: Europe crysvita injection Revenue Share (%), by Types 2025 & 2033
- Figure 34: Europe crysvita injection Volume Share (%), by Types 2025 & 2033
- Figure 35: Europe crysvita injection Revenue (billion), by Country 2025 & 2033
- Figure 36: Europe crysvita injection Volume (K), by Country 2025 & 2033
- Figure 37: Europe crysvita injection Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe crysvita injection Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa crysvita injection Revenue (billion), by Application 2025 & 2033
- Figure 40: Middle East & Africa crysvita injection Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa crysvita injection Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa crysvita injection Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa crysvita injection Revenue (billion), by Types 2025 & 2033
- Figure 44: Middle East & Africa crysvita injection Volume (K), by Types 2025 & 2033
- Figure 45: Middle East & Africa crysvita injection Revenue Share (%), by Types 2025 & 2033
- Figure 46: Middle East & Africa crysvita injection Volume Share (%), by Types 2025 & 2033
- Figure 47: Middle East & Africa crysvita injection Revenue (billion), by Country 2025 & 2033
- Figure 48: Middle East & Africa crysvita injection Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa crysvita injection Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa crysvita injection Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific crysvita injection Revenue (billion), by Application 2025 & 2033
- Figure 52: Asia Pacific crysvita injection Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific crysvita injection Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific crysvita injection Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific crysvita injection Revenue (billion), by Types 2025 & 2033
- Figure 56: Asia Pacific crysvita injection Volume (K), by Types 2025 & 2033
- Figure 57: Asia Pacific crysvita injection Revenue Share (%), by Types 2025 & 2033
- Figure 58: Asia Pacific crysvita injection Volume Share (%), by Types 2025 & 2033
- Figure 59: Asia Pacific crysvita injection Revenue (billion), by Country 2025 & 2033
- Figure 60: Asia Pacific crysvita injection Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific crysvita injection Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific crysvita injection Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global crysvita injection Revenue billion Forecast, by Application 2020 & 2033
- Table 2: Global crysvita injection Volume K Forecast, by Application 2020 & 2033
- Table 3: Global crysvita injection Revenue billion Forecast, by Types 2020 & 2033
- Table 4: Global crysvita injection Volume K Forecast, by Types 2020 & 2033
- Table 5: Global crysvita injection Revenue billion Forecast, by Region 2020 & 2033
- Table 6: Global crysvita injection Volume K Forecast, by Region 2020 & 2033
- Table 7: Global crysvita injection Revenue billion Forecast, by Application 2020 & 2033
- Table 8: Global crysvita injection Volume K Forecast, by Application 2020 & 2033
- Table 9: Global crysvita injection Revenue billion Forecast, by Types 2020 & 2033
- Table 10: Global crysvita injection Volume K Forecast, by Types 2020 & 2033
- Table 11: Global crysvita injection Revenue billion Forecast, by Country 2020 & 2033
- Table 12: Global crysvita injection Volume K Forecast, by Country 2020 & 2033
- Table 13: United States crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: United States crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Canada crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 18: Mexico crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global crysvita injection Revenue billion Forecast, by Application 2020 & 2033
- Table 20: Global crysvita injection Volume K Forecast, by Application 2020 & 2033
- Table 21: Global crysvita injection Revenue billion Forecast, by Types 2020 & 2033
- Table 22: Global crysvita injection Volume K Forecast, by Types 2020 & 2033
- Table 23: Global crysvita injection Revenue billion Forecast, by Country 2020 & 2033
- Table 24: Global crysvita injection Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Brazil crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Argentina crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global crysvita injection Revenue billion Forecast, by Application 2020 & 2033
- Table 32: Global crysvita injection Volume K Forecast, by Application 2020 & 2033
- Table 33: Global crysvita injection Revenue billion Forecast, by Types 2020 & 2033
- Table 34: Global crysvita injection Volume K Forecast, by Types 2020 & 2033
- Table 35: Global crysvita injection Revenue billion Forecast, by Country 2020 & 2033
- Table 36: Global crysvita injection Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 40: Germany crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 42: France crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: Italy crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Spain crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 48: Russia crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 50: Benelux crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 52: Nordics crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global crysvita injection Revenue billion Forecast, by Application 2020 & 2033
- Table 56: Global crysvita injection Volume K Forecast, by Application 2020 & 2033
- Table 57: Global crysvita injection Revenue billion Forecast, by Types 2020 & 2033
- Table 58: Global crysvita injection Volume K Forecast, by Types 2020 & 2033
- Table 59: Global crysvita injection Revenue billion Forecast, by Country 2020 & 2033
- Table 60: Global crysvita injection Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 62: Turkey crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 64: Israel crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 66: GCC crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 68: North Africa crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 70: South Africa crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global crysvita injection Revenue billion Forecast, by Application 2020 & 2033
- Table 74: Global crysvita injection Volume K Forecast, by Application 2020 & 2033
- Table 75: Global crysvita injection Revenue billion Forecast, by Types 2020 & 2033
- Table 76: Global crysvita injection Volume K Forecast, by Types 2020 & 2033
- Table 77: Global crysvita injection Revenue billion Forecast, by Country 2020 & 2033
- Table 78: Global crysvita injection Volume K Forecast, by Country 2020 & 2033
- Table 79: China crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 80: China crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 82: India crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 84: Japan crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 86: South Korea crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 88: ASEAN crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 90: Oceania crysvita injection Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific crysvita injection Revenue (billion) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific crysvita injection Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the crysvita injection?
The projected CAGR is approximately 7.08%.
2. Which companies are prominent players in the crysvita injection?
Key companies in the market include Kyowa Kirin, Ultragenyx Pharmaceutical.
3. What are the main segments of the crysvita injection?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 15.53 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4350.00, USD 6525.00, and USD 8700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "crysvita injection," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the crysvita injection report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the crysvita injection?
To stay informed about further developments, trends, and reports in the crysvita injection, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


