Key Insights
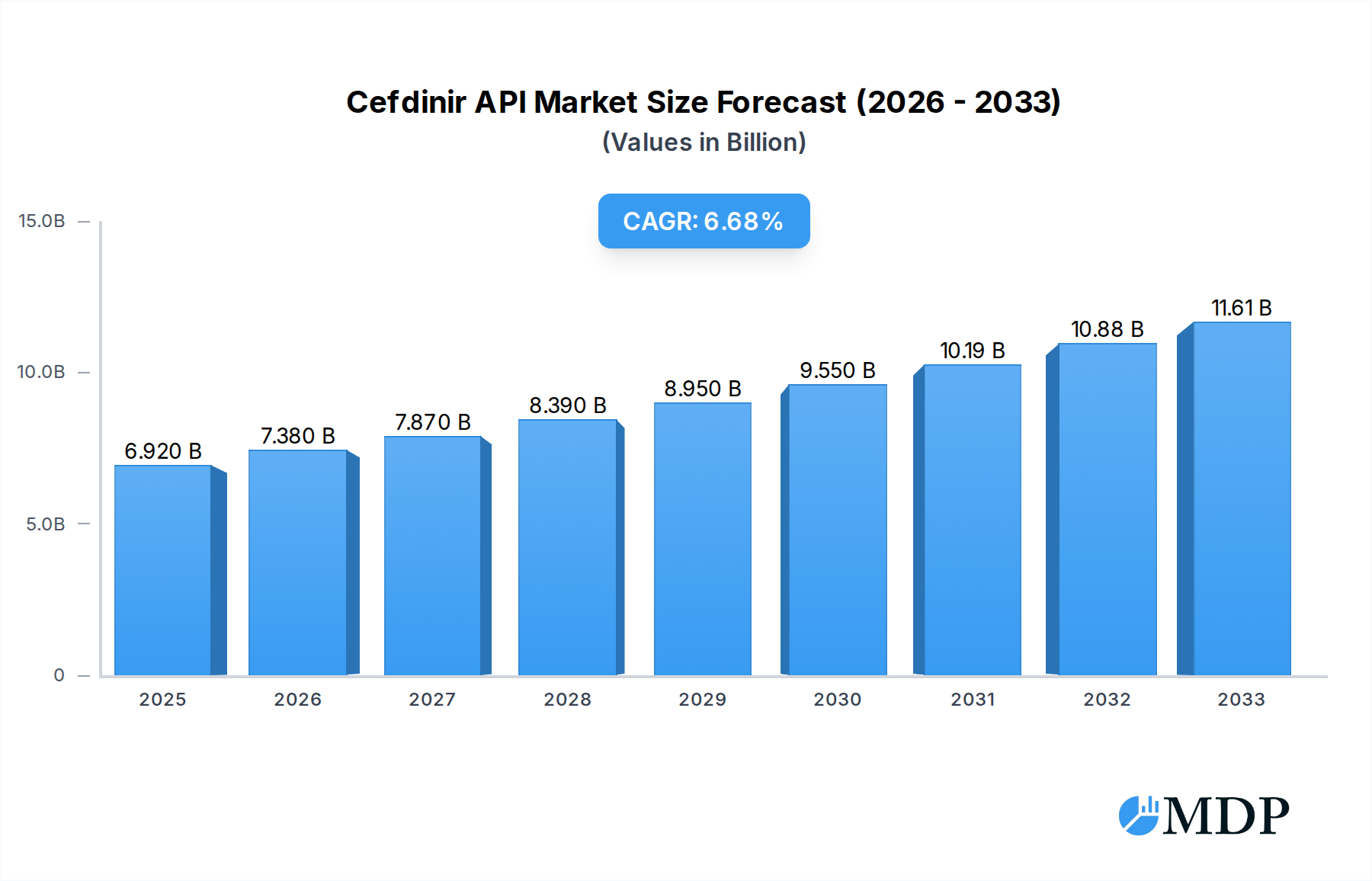
The Cefdinir API market is poised for significant expansion, projected to reach a USD 6.92 billion valuation in 2025. This robust growth is underpinned by a compelling CAGR of 6.69%, indicating sustained demand and investment within the sector through to 2033. The increasing prevalence of bacterial infections globally, coupled with the well-established efficacy and safety profile of Cefdinir as a broad-spectrum antibiotic, serves as a primary driver for this market's ascent. Furthermore, advancements in pharmaceutical manufacturing technologies and a growing emphasis on stringent quality control for active pharmaceutical ingredients (APIs) are contributing to the market's upward trajectory. The demand for Cefdinir API is particularly strong in oral dosage forms, such as tablets and capsules, catering to a wide range of outpatient and inpatient treatments. The market is also witnessing a growing preference for high-purity Cefdinir API, with both ≥98% and ≥99% purity grades experiencing consistent demand.
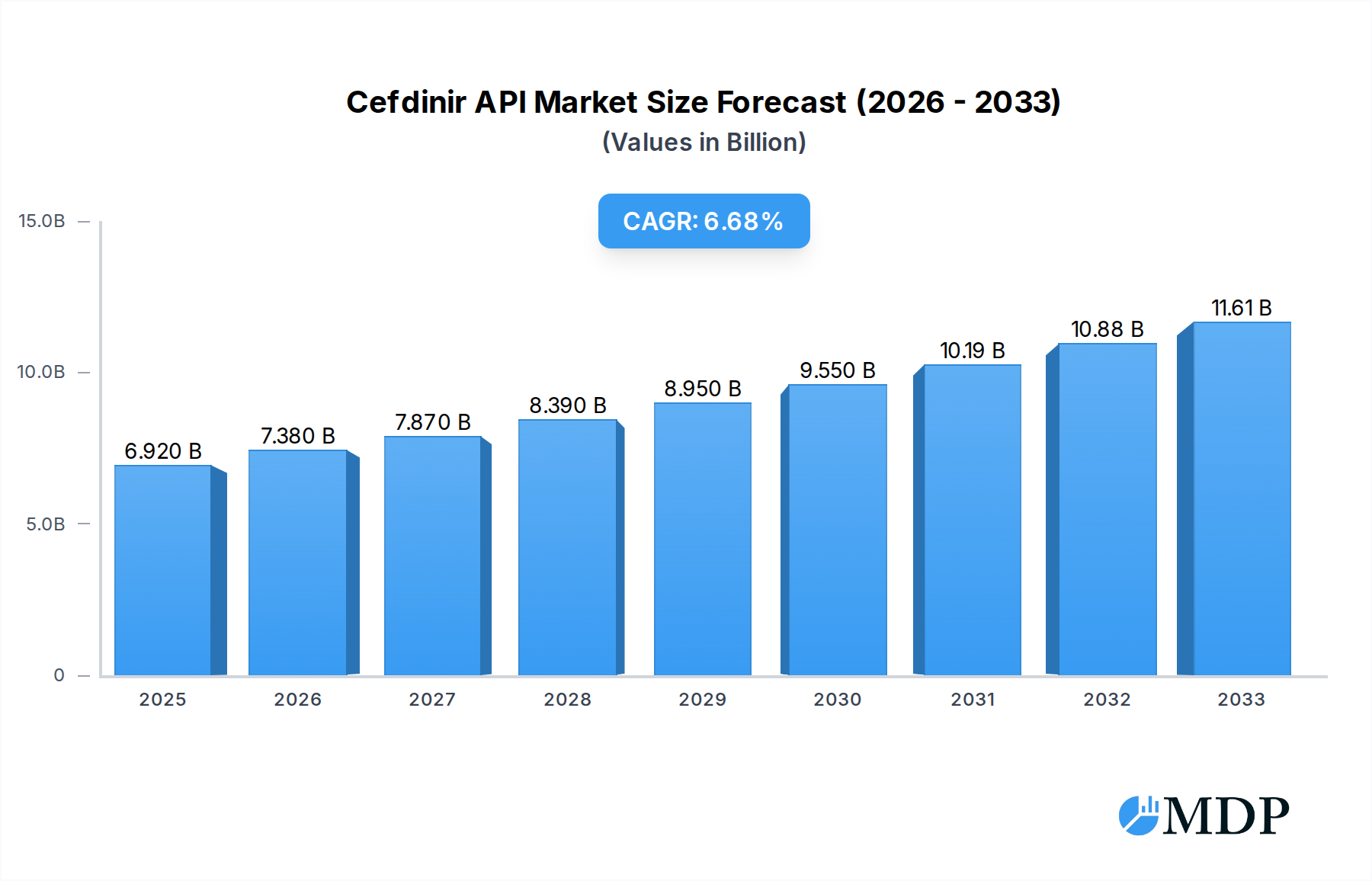
Cefdinir API Market Size (In Billion)

The market's expansion is further influenced by evolving healthcare landscapes and increased access to essential medicines in developing economies. Key players like Apeloa Pharmaceutical, Zhejiang Yongning Pharmaceutical, and Hejia Pharmaceutical are actively investing in R&D and production capacity to meet this escalating demand. Strategic collaborations and expansions within the Asia Pacific region, especially China and India, are critical to the supply chain dynamics. While the market benefits from strong drivers, potential restraints such as the emergence of antibiotic resistance and the development of novel therapeutic alternatives could pose challenges. However, the established therapeutic value of Cefdinir and ongoing efforts to optimize its use and production are expected to mitigate these risks, ensuring a dynamic and growing market for Cefdinir API over the forecast period.
Cefdinir API Company Market Share

Cefdinir API Market Report: Comprehensive Analysis and Forecast (2019–2033)
This in-depth market research report provides a panoramic view of the global Cefdinir API (Active Pharmaceutical Ingredient) market. Spanning the study period from 2019 to 2033, with a base year of 2025, this report offers critical insights into market dynamics, industry trends, leading segments, product developments, growth drivers, challenges, and emerging opportunities. Leveraging high-traffic keywords such as "Cefdinir API," "antibiotic API," "pharmaceutical ingredients," "generics market," and "API manufacturing," this report aims to maximize search visibility and attract industry stakeholders including pharmaceutical manufacturers, API suppliers, investors, and research institutions. The report features a rigorous analysis based on historical data (2019–2024) and projections for the forecast period (2025–2033), ensuring actionable intelligence for strategic decision-making.
Cefdinir API Market Dynamics & Concentration
The Cefdinir API market exhibits a moderate to high concentration, with a few key players dominating production. Major companies like Apeloa Pharmaceutical, Zhejiang Yongning Pharmaceutical, Hejia Pharmaceutical, Fuan Pharmaceutical, Covalent, Jeil Pharmaceutical, AMICOGEN, Shiono Chemical, and Hanmi Fine Chemical hold significant market share, estimated to be in the billions. Innovation drivers in this sector are primarily focused on improving synthesis processes for higher purity and yield, as well as cost reduction to cater to the burgeoning generics market. Regulatory frameworks, governed by bodies such as the FDA and EMA, play a crucial role in shaping market entry and product quality standards, ensuring a minimum purity of 98% and often exceeding 99% for premium grades. Product substitutes, while present in the broader antibiotic landscape, are less direct for Cefdinir due to its specific efficacy profile. End-user trends indicate a growing demand for oral dosage forms like tablets and capsules, driving the need for high-quality API. Mergers and acquisitions (M&A) activities, though not constant, have historically contributed to market consolidation, with an estimated XX M&A deals annually in the last five years. The overall market value is projected to reach billions by 2025.
Cefdinir API Industry Trends & Analysis
The Cefdinir API industry is on a robust growth trajectory, driven by a confluence of factors that underscore its increasing significance in the global pharmaceutical landscape. The market is projected to witness a Compound Annual Growth Rate (CAGR) of approximately XX% between 2025 and 2033, reflecting sustained demand and expanding applications. This growth is fueled by the persistent prevalence of bacterial infections worldwide, necessitating effective and accessible antibiotic treatments. The increasing penetration of generic Cefdinir formulations in both developed and emerging economies significantly propels the demand for high-quality API. Furthermore, advancements in chemical synthesis technologies are enabling manufacturers to achieve higher purity levels (≥99%) more efficiently and at a lower cost, thus enhancing competitiveness and market accessibility. Technological disruptions, such as the adoption of continuous manufacturing processes and green chemistry principles, are optimizing production efficiency and reducing environmental impact. Consumer preferences lean towards convenient oral dosage forms, primarily tablets and capsules, thereby shaping the demand for specific API characteristics. The competitive dynamics are characterized by a blend of established global API manufacturers and emerging players from Asia, particularly China and India, who are increasingly contributing to the supply chain. Market penetration for Cefdinir API is expected to climb steadily as healthcare access expands and the awareness of effective antibiotic options grows. The global market size for Cefdinir API is estimated to be in the billions in the base year of 2025, with significant expansion anticipated throughout the forecast period.
Leading Markets & Segments in Cefdinir API
The Cefdinir API market exhibits distinct regional and segment leadership, driven by diverse economic, regulatory, and healthcare infrastructure factors. North America and Europe currently lead in terms of market value and consumption due to well-established healthcare systems, high healthcare spending, and robust regulatory oversight that mandates stringent API quality. However, the Asia-Pacific region, particularly China and India, is emerging as a dominant manufacturing hub and a rapidly growing consumption market, owing to expanding healthcare access, a burgeoning generic drug industry, and cost-effective production capabilities.
Application Dominance:
- Tablet: The tablet segment is the most dominant application for Cefdinir API, accounting for an estimated XX% of the market share in 2025. This is attributed to the ease of administration, patient compliance, and cost-effectiveness of tablet formulations. Key drivers include the widespread availability of generic tablet products and patient preference for self-medication.
- Capsule: Capsules represent the second-largest segment, holding approximately XX% of the market. They offer advantages in terms of taste masking and ease of swallowing for certain patient demographics.
- Other: The "Other" category, which may include suspensions or other less common dosage forms, accounts for a smaller but growing XX% of the market.
Type Dominance:
- Purity ≥98%: The Cefdinir API with a purity of 98% or higher is the standard requirement for most pharmaceutical formulations and commands a significant market share, estimated at XX% in 2025. This purity level ensures efficacy and safety for general therapeutic use.
- Purity ≥99%: The segment for Cefdinir API with a purity exceeding 99% is a growing niche, particularly for advanced formulations or where exceptionally high purity is mandated by specific regulatory requirements or for enhanced therapeutic outcomes. This segment is projected to grow at a faster CAGR due to increasing quality expectations and technological advancements in purification techniques, holding approximately XX% of the market in 2025.
Economic policies in emerging markets, aimed at increasing access to essential medicines, are a significant driver for the growth of the Cefdinir API market. Infrastructure development, including improved logistics and supply chain networks, facilitates the distribution of both API and finished formulations globally. The regulatory landscape, while stringent, also fosters market growth by ensuring product quality and patient safety, thereby building trust and encouraging wider adoption.
Cefdinir API Product Developments
Recent product developments in the Cefdinir API sector have focused on enhancing manufacturing efficiency and achieving superior purity profiles. Innovations in synthesis routes and purification technologies have enabled the consistent production of Cefdinir API with purities exceeding 99%, catering to the increasing demand for high-grade pharmaceutical ingredients. These advancements not only ensure greater efficacy and reduced impurities in the final drug product but also contribute to cost optimization for manufacturers. The development of more environmentally friendly synthesis processes is also a growing trend, aligning with global sustainability initiatives. These product developments offer competitive advantages by allowing manufacturers to meet stringent regulatory standards and address the evolving needs of the global pharmaceutical market for reliable and high-quality Cefdinir API.
Key Drivers of Cefdinir API Growth
The growth of the Cefdinir API market is propelled by several key drivers that underscore its sustained demand and expanding applications.
- Rising incidence of bacterial infections: The global increase in bacterial infections necessitates the continuous supply of effective antibiotics like Cefdinir.
- Growth of the generic drug market: The expiration of patents for branded Cefdinir formulations has opened doors for generic manufacturers, significantly boosting the demand for affordable API.
- Advancements in pharmaceutical manufacturing: Innovations in chemical synthesis and purification techniques are leading to more efficient and cost-effective production of high-purity Cefdinir API.
- Expanding healthcare access: Improved healthcare infrastructure and accessibility in emerging economies are increasing the demand for essential medicines, including Cefdinir.
- Favorable regulatory environments: Supportive regulatory frameworks that facilitate the approval and marketing of generic antibiotics contribute to market expansion.
Challenges in the Cefdinir API Market
Despite its growth, the Cefdinir API market faces several challenges that can potentially restrain its expansion.
- Intense price competition: The highly competitive nature of the API market, particularly from low-cost manufacturing regions, exerts significant downward pressure on pricing.
- Stringent regulatory compliance: Meeting evolving and stringent quality and regulatory standards set by global health authorities (e.g., FDA, EMA) requires continuous investment and adherence, posing a barrier for smaller manufacturers.
- Supply chain disruptions: Geopolitical factors, trade tensions, and unforeseen events can disrupt the global supply chain for raw materials and intermediates, impacting API production and delivery.
- Development of antibiotic resistance: The growing threat of antibiotic resistance could lead to reduced reliance on certain classes of antibiotics, although Cefdinir's efficacy profile remains valuable.
- Environmental regulations: Increasing environmental scrutiny on chemical manufacturing processes may necessitate further investments in sustainable and eco-friendly production methods.
Emerging Opportunities in Cefdinir API
The Cefdinir API market is poised for significant long-term growth, driven by several emerging opportunities. The increasing demand for combination therapies and novel drug delivery systems presents an avenue for innovation and market differentiation. Strategic partnerships between API manufacturers and finished dosage form producers can streamline the supply chain and accelerate market penetration. Furthermore, the untapped potential in underserved emerging markets, where the burden of bacterial infections is high, offers substantial growth prospects. Technological breakthroughs in continuous manufacturing and advanced purification techniques are expected to further optimize production costs and product quality, creating new competitive advantages.
Leading Players in the Cefdinir API Sector
- Apeloa Pharmaceutical
- Zhejiang Yongning Pharmaceutical
- Hejia Pharmaceutical
- Fuan Pharmaceutical
- Covalent
- Jeil Pharmaceutical
- AMICOGEN
- Shiono Chemical
- Hanmi Fine Chemical
Key Milestones in Cefdinir API Industry
- 2019: Increased regulatory focus on API impurity profiling and traceability globally.
- 2020: Supply chain disruptions due to global events impacting raw material availability.
- 2021: Launch of several new generic Cefdinir formulations in key markets.
- 2022: Growing adoption of advanced analytical techniques for API quality control.
- 2023: Increased investment in R&D for more sustainable API manufacturing processes.
- 2024: Continued expansion of manufacturing capacities in Asia-Pacific.
Strategic Outlook for Cefdinir API Market
The strategic outlook for the Cefdinir API market remains highly positive, driven by persistent global demand for effective antibiotic treatments and the robust growth of the generic pharmaceutical sector. Key growth accelerators include further optimization of manufacturing processes to enhance cost-effectiveness and product purity, particularly for the ≥99% purity segment. Strategic alliances and collaborations between API manufacturers and pharmaceutical companies will be crucial for expanding market reach and ensuring a stable supply chain. Focus on innovation in sustainable manufacturing practices will also be a significant differentiator. The expansion into emerging markets, coupled with adherence to evolving regulatory landscapes, will be pivotal for sustained growth, positioning the Cefdinir API market for continued expansion throughout the forecast period.
Cefdinir API Segmentation
-
1. Application
- 1.1. Tablet
- 1.2. Capsule
- 1.3. Other
-
2. Types
- 2.1. Purity ≥98%
- 2.2. Purity ≥99%
Cefdinir API Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
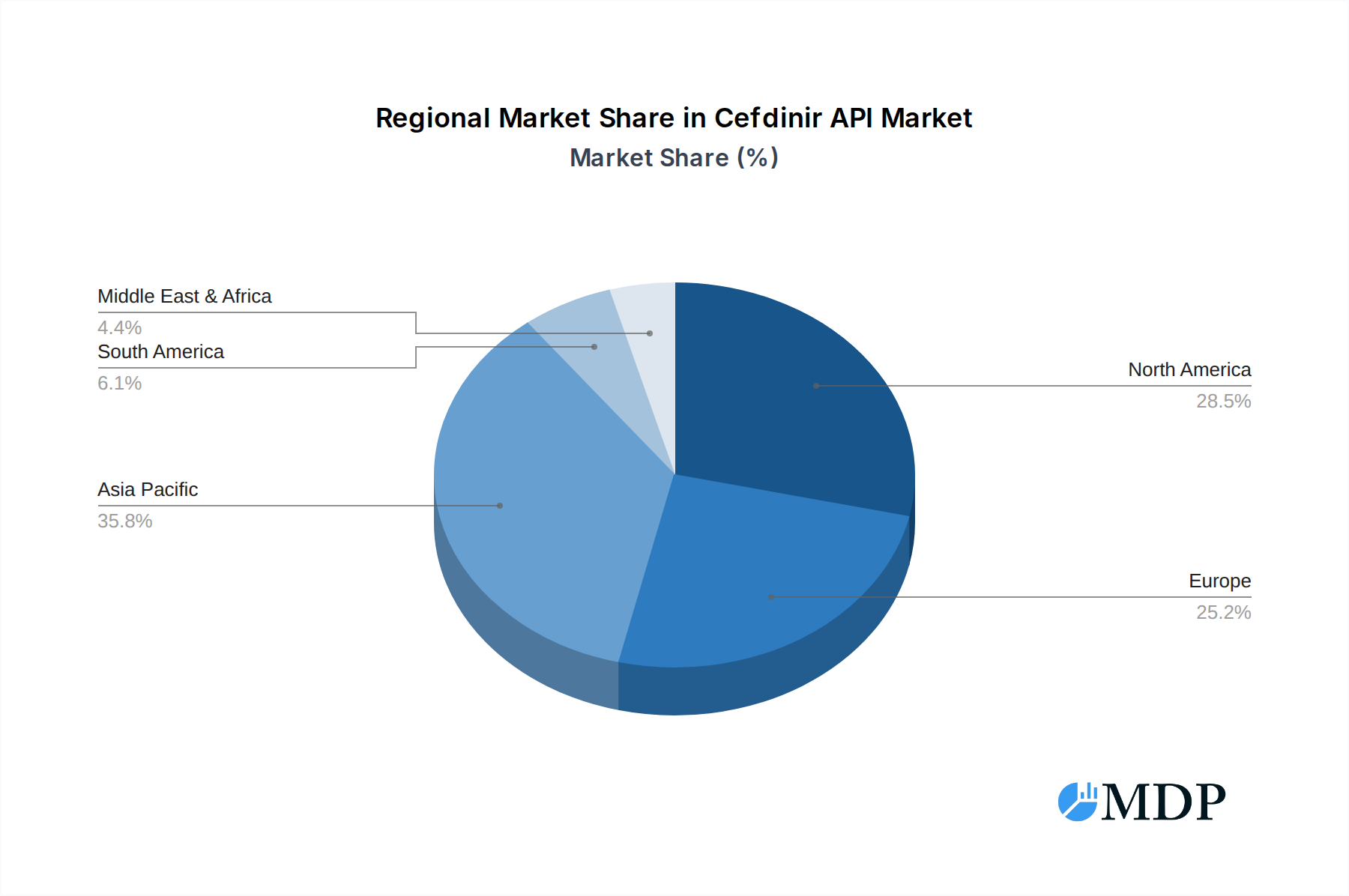
Cefdinir API Regional Market Share

Geographic Coverage of Cefdinir API
Cefdinir API REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.69% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Cefdinir API Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Tablet
- 5.1.2. Capsule
- 5.1.3. Other
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Purity ≥98%
- 5.2.2. Purity ≥99%
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Cefdinir API Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Tablet
- 6.1.2. Capsule
- 6.1.3. Other
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Purity ≥98%
- 6.2.2. Purity ≥99%
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Cefdinir API Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Tablet
- 7.1.2. Capsule
- 7.1.3. Other
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Purity ≥98%
- 7.2.2. Purity ≥99%
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Cefdinir API Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Tablet
- 8.1.2. Capsule
- 8.1.3. Other
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Purity ≥98%
- 8.2.2. Purity ≥99%
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Cefdinir API Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Tablet
- 9.1.2. Capsule
- 9.1.3. Other
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Purity ≥98%
- 9.2.2. Purity ≥99%
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Cefdinir API Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Tablet
- 10.1.2. Capsule
- 10.1.3. Other
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Purity ≥98%
- 10.2.2. Purity ≥99%
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Apeloa Pharmaceutical
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Zhejiang Yongning Pharmaceutical
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Hejia Pharmaceutical
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Fuan Pharmaceutical
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Covalent
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Jeil Pharmaceutical
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 AMICOGEN
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Shiono Chemical
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Hanmi Fine Chemical
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.1 Apeloa Pharmaceutical
List of Figures
- Figure 1: Global Cefdinir API Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Cefdinir API Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Cefdinir API Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Cefdinir API Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Cefdinir API Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Cefdinir API Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Cefdinir API Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Cefdinir API Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Cefdinir API Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Cefdinir API Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Cefdinir API Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Cefdinir API Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Cefdinir API Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Cefdinir API Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Cefdinir API Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Cefdinir API Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Cefdinir API Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Cefdinir API Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Cefdinir API Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Cefdinir API Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Cefdinir API Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Cefdinir API Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Cefdinir API Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Cefdinir API Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Cefdinir API Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Cefdinir API Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Cefdinir API Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Cefdinir API Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Cefdinir API Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Cefdinir API Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Cefdinir API Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Cefdinir API Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Cefdinir API Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Cefdinir API Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Cefdinir API Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Cefdinir API Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Cefdinir API Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Cefdinir API Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Cefdinir API Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Cefdinir API Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Cefdinir API Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Cefdinir API Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Cefdinir API Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Cefdinir API Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Cefdinir API Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Cefdinir API Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Cefdinir API Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Cefdinir API Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Cefdinir API Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Cefdinir API Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Cefdinir API?
The projected CAGR is approximately 6.69%.
2. Which companies are prominent players in the Cefdinir API?
Key companies in the market include Apeloa Pharmaceutical, Zhejiang Yongning Pharmaceutical, Hejia Pharmaceutical, Fuan Pharmaceutical, Covalent, Jeil Pharmaceutical, AMICOGEN, Shiono Chemical, Hanmi Fine Chemical.
3. What are the main segments of the Cefdinir API?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Cefdinir API," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Cefdinir API report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Cefdinir API?
To stay informed about further developments, trends, and reports in the Cefdinir API, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


