Key Insights
The Canada respiratory devices market, valued at approximately $XXX million in 2025, is projected to experience robust growth, exhibiting a compound annual growth rate (CAGR) of 8.73% from 2025 to 2033. This expansion is fueled by several key factors. The rising prevalence of chronic respiratory diseases like asthma, COPD, and cystic fibrosis, coupled with an aging population experiencing increased susceptibility to these conditions, significantly drives demand for respiratory devices. Technological advancements leading to the development of smaller, more portable, and user-friendly devices further contribute to market growth. Increased awareness of respiratory health and improved healthcare infrastructure in Canada also play a pivotal role. The market is segmented into diagnostic and monitoring devices (spirometers, sleep test devices, etc.), therapeutic devices (CPAP, BiPAP, inhalers, etc.), and disposables (masks, breathing circuits, etc.). Each segment contributes uniquely to the overall market value and growth trajectory. Competition among established players like Medtronic, Fisher & Paykel Healthcare, and GE Healthcare, alongside emerging players, fosters innovation and competitive pricing, benefiting consumers. Regional variations within Canada (Eastern, Western, and Central) may exist due to differing healthcare access and prevalence of respiratory diseases across these regions. While precise regional breakdowns are not fully provided, it's reasonable to expect higher market penetration in more densely populated urban areas of Eastern Canada. Further research into specific regional data will reveal a more precise picture of this market segmentation.
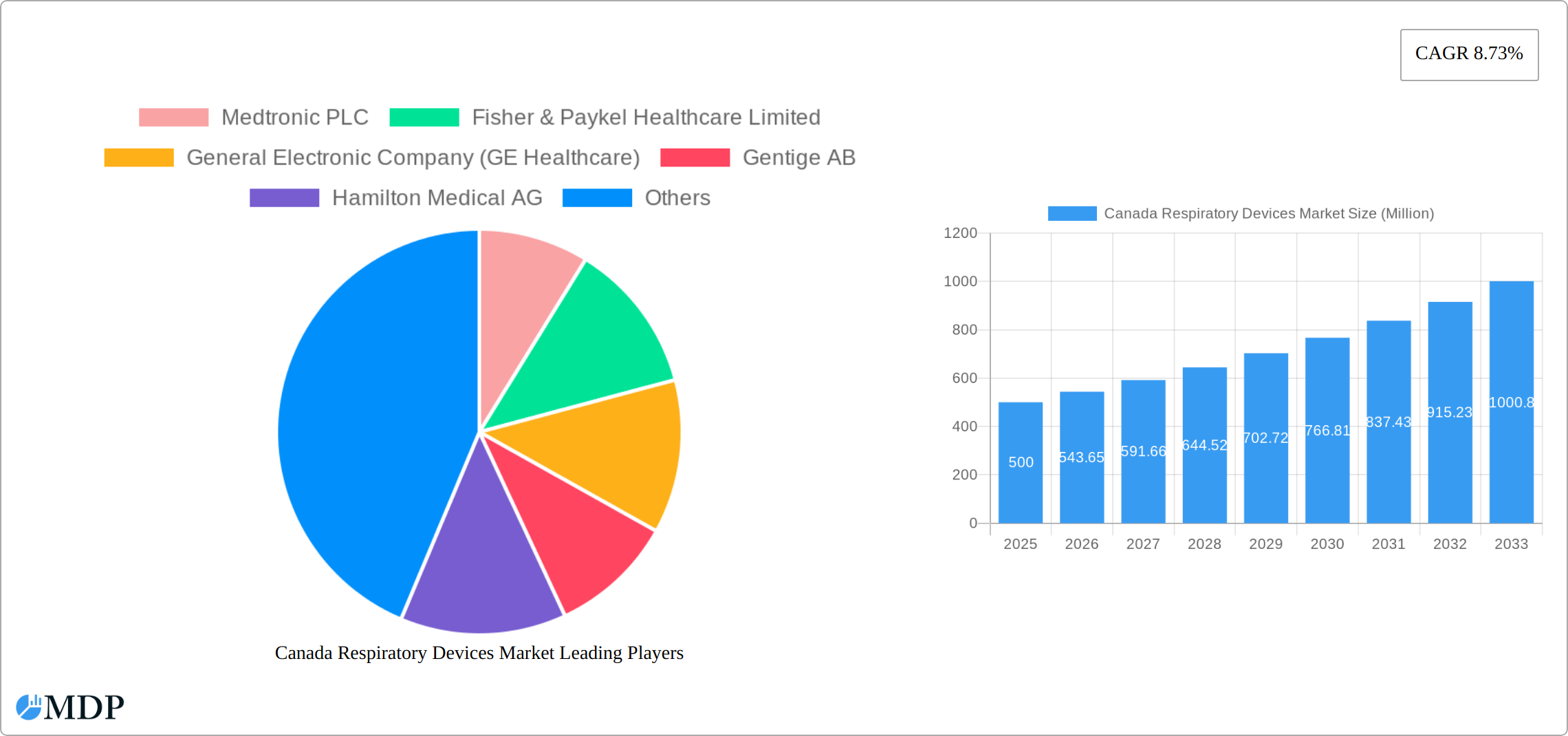
Canada Respiratory Devices Market Market Size (In Million)

The market's growth is not without challenges. Healthcare costs and insurance coverage limitations could restrain market expansion, particularly for advanced or specialized respiratory devices. Regulatory hurdles for new device approvals and reimbursement processes also pose a potential obstacle. However, the overall trend points towards a sustained period of growth driven by the increasing burden of respiratory illnesses and the ongoing innovation in respiratory device technology. The Canadian government's initiatives to improve healthcare access and the rising adoption of telehealth services are likely to further support the market's expansion in the forecast period. The market offers significant opportunities for both established manufacturers and emerging players who can innovate to meet the growing and evolving needs of respiratory patients in Canada.
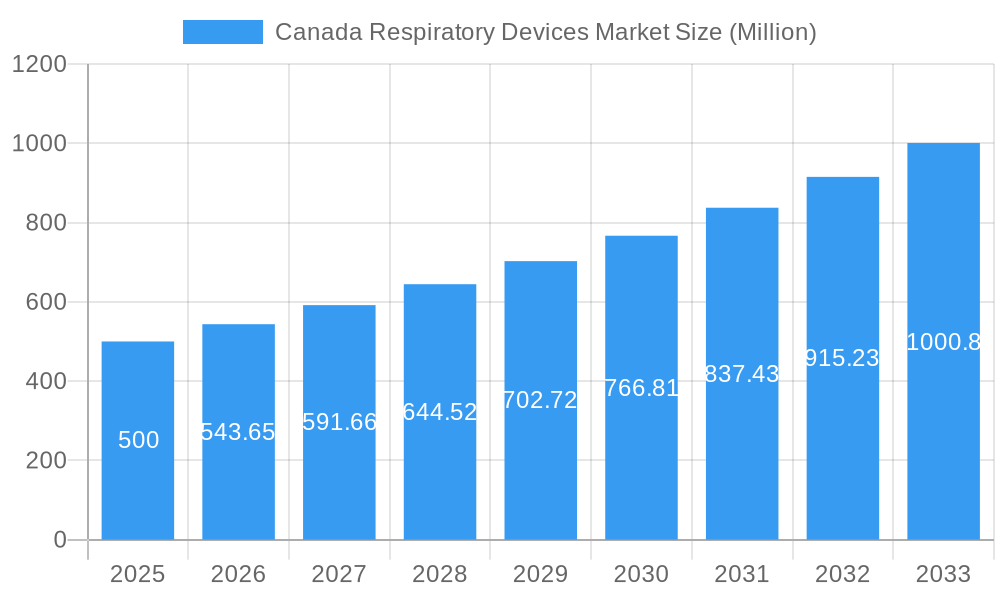
Canada Respiratory Devices Market Company Market Share

Canada Respiratory Devices Market Report: 2019-2033
This comprehensive report provides an in-depth analysis of the Canada Respiratory Devices Market, offering valuable insights for industry stakeholders, investors, and strategic decision-makers. With a focus on market dynamics, trends, and future growth potential, this report covers the period from 2019 to 2033, with a base year of 2025. The market size is projected to reach xx Million by 2033, exhibiting a CAGR of xx% during the forecast period (2025-2033). The report segments the market by device type (Diagnostic and Monitoring, Therapeutic, and Disposables), providing granular analysis of key segments and their growth drivers. Leading players such as Medtronic PLC, Fisher & Paykel Healthcare Limited, and GE Healthcare are profiled, alongside an analysis of their market share and strategic initiatives.
Canada Respiratory Devices Market Dynamics & Concentration
The Canadian respiratory devices market is characterized by a moderate level of concentration, with a few major players holding significant market share. Market share is largely driven by brand recognition, technological innovation, and established distribution networks. The market is experiencing considerable innovation, driven by advancements in sensor technology, artificial intelligence (AI), and telehealth. Stringent regulatory frameworks, including those set by Health Canada, play a crucial role in shaping market dynamics. Substitutes for certain respiratory devices, such as traditional therapies, exist, but the overall market is driven by the need for advanced diagnostic and therapeutic solutions. End-user trends are shifting towards a preference for minimally invasive, user-friendly, and technologically advanced devices, alongside increasing telehealth adoption. M&A activity has been moderate in recent years, with approximately xx deals recorded between 2019 and 2024, reflecting a strategic push towards consolidation and expansion within the market.
- Market Concentration: Moderately concentrated, with top 5 players holding approximately xx% market share in 2024.
- Innovation Drivers: Advancements in sensor technology, AI, and telehealth.
- Regulatory Frameworks: Stringent regulations imposed by Health Canada influence product development and market entry.
- Product Substitutes: Traditional therapies and alternative treatments pose some competition.
- End-User Trends: Growing preference for minimally invasive, user-friendly devices and telehealth solutions.
- M&A Activity: Approximately xx M&A deals between 2019 and 2024.
Canada Respiratory Devices Market Industry Trends & Analysis
The Canadian respiratory devices market is poised for robust growth, fueled by a confluence of factors. The escalating prevalence of chronic respiratory illnesses like asthma, COPD, and cystic fibrosis is a primary driver. This is further amplified by Canada's aging population and a heightened public awareness of respiratory health, leading to increased demand for both diagnostic and therapeutic devices. The market is experiencing a significant technological disruption, with advancements in remote patient monitoring (RPM), AI-powered diagnostics, and miniaturized devices transforming the landscape. These innovations deliver improved patient care, enhanced treatment adherence, and cost-effective solutions. Furthermore, consumer preferences are shifting towards convenient, portable, and user-friendly devices. The competitive landscape remains dynamic, marked by continuous innovation, strategic collaborations, mergers and acquisitions, and the introduction of cutting-edge products. While the market exhibited a CAGR of xx% during the historical period (2019-2024), projections indicate a sustained healthy CAGR of xx% during the forecast period (2025-2033). Market penetration of advanced technologies remains high in urban centers, presenting substantial growth opportunities in underserved rural and remote communities.
Leading Markets & Segments in Canada Respiratory Devices Market
The Canadian respiratory devices market shows significant growth across all provinces and territories, with urban centers experiencing higher adoption rates due to factors such as higher healthcare infrastructure and specialized medical facilities. However, there is substantial growth potential in rural and remote areas. Within the device segments, therapeutic devices, particularly ventilators, CPAP devices, and oxygen concentrators, dominate the market due to the increasing prevalence of chronic respiratory illnesses. This segment is expected to maintain its leading position throughout the forecast period. The demand for disposables, especially masks and breathing circuits, is also substantial, driven by ongoing healthcare needs.
- Key Drivers (Therapeutic Devices): Rising prevalence of chronic respiratory diseases, aging population, improved healthcare infrastructure in urban areas.
- Dominance Analysis: The therapeutic devices segment is the dominant market force, driven by the high prevalence of chronic conditions. This segment holds approximately xx% of the total market share in 2024.
Canada Respiratory Devices Market Product Developments
Recent product innovations underscore a clear trend towards enhancing portability, ease of use, and therapeutic efficacy. Smart inhalers equipped with integrated sensors, connected ventilators facilitating remote monitoring, and miniaturized diagnostic tools exemplify these advancements. These innovations directly contribute to improved patient compliance, better treatment outcomes, and cost-effectiveness within the healthcare system. The market is witnessing a rapid integration of advanced technologies, including AI and machine learning, to optimize device performance and personalize respiratory care. This trend is projected to continue, leading to the creation of even more sophisticated and user-friendly respiratory devices tailored to specific patient needs and preferences.
Key Drivers of Canada Respiratory Devices Market Growth
Several factors are driving growth in the Canadian respiratory devices market. The increasing prevalence of chronic respiratory diseases such as asthma and COPD fuels demand for both diagnostic and therapeutic devices. Technological advancements, including the development of connected devices and AI-powered diagnostic tools, are improving treatment outcomes and patient experience. Government initiatives and supportive healthcare policies aimed at improving respiratory health are encouraging market expansion. The increasing focus on preventative care and early diagnosis is also contributing to market growth.
Challenges in the Canada Respiratory Devices Market Market
The Canadian respiratory devices market faces several challenges. High costs associated with advanced devices can limit accessibility for some patients. Stringent regulatory approvals and compliance requirements can slow down product development and market entry. Supply chain disruptions and fluctuations in raw material prices can impact production and pricing. Intense competition among established players and the entry of new competitors can create pricing pressures. These factors combined could hinder market growth in the coming years.
Emerging Opportunities in Canada Respiratory Devices Market
The Canadian respiratory devices market presents a wealth of compelling opportunities. Ongoing technological advancements, including miniaturization, smart sensors, and seamless AI integration, will pave the way for the development of increasingly sophisticated and effective respiratory devices. The expanding adoption of telehealth and remote patient monitoring (RPM) solutions offers significant potential to enhance access to quality respiratory care, particularly in underserved rural and remote areas. Strategic partnerships between device manufacturers, healthcare providers, and technology companies are crucial for driving innovation and expanding market reach. The growing emphasis on personalized medicine further fuels opportunities for developing tailored respiratory therapies that address individual patient characteristics and needs. This personalized approach promises to optimize treatment efficacy and improve overall patient outcomes.
Leading Players in the Canada Respiratory Devices Market Sector
- Medtronic PLC
- Fisher & Paykel Healthcare Limited
- General Electric Company (GE Healthcare)
- Getinge AB
- Hamilton Medical AG
- Becton Dickinson and Company
- Invacare Corporation
- Drägerwerk AG & Co KGaA
- Nihon Kohden Corporation
- Koninklijke Philips N V
Key Milestones in Canada Respiratory Devices Market Industry
- May 2022: Honeywell launched two new NIOSH-certified respiratory offerings, expanding its portfolio of protective equipment for healthcare professionals. This strengthened Honeywell's position in the market and increased the availability of high-quality respiratory protection.
- February 2022: ZEPHYRx LLC. and Medical International Research (MIR) partnered to market the ZEPHYRx Remote Respiratory Monitoring (RRM) Solution. This partnership marked a significant step towards expanding remote patient monitoring capabilities in North America and improved access to respiratory care.
Strategic Outlook for Canada Respiratory Devices Market Market
The future of the Canadian respiratory devices market looks promising, driven by ongoing technological innovation, rising prevalence of respiratory diseases, and increasing government support for healthcare improvements. Strategic partnerships and collaborations between key players will play a pivotal role in shaping market dynamics and driving future growth. Focus on developing affordable, portable, and user-friendly devices will be crucial for expanding market access and improving patient outcomes. The market is poised for sustained growth driven by the rising adoption of advanced technologies, expanding telehealth, and increased awareness of respiratory health.
Canada Respiratory Devices Market Segmentation
-
1. Diagnostic and Monitoring Devices
- 1.1. Spirometers
- 1.2. Sleep Test Devices
- 1.3. Peak Flow Meters
- 1.4. Pulse Oximeters
- 1.5. Capnographs
- 1.6. Other Diagnostic and Monitoring Devices
-
2. Therapeutic Device
- 2.1. CPAP Devices
- 2.2. BiPAP Devices
- 2.3. Humidifiers
- 2.4. Nebulizers
- 2.5. Oxygen Concentrators
- 2.6. Ventilators
- 2.7. Inhalers
- 2.8. Other Therapeutic Devices
-
3. Disposables
- 3.1. Masks
- 3.2. Breathing Circuit
- 3.3. Other Disposables
Canada Respiratory Devices Market Segmentation By Geography
- 1. Canada
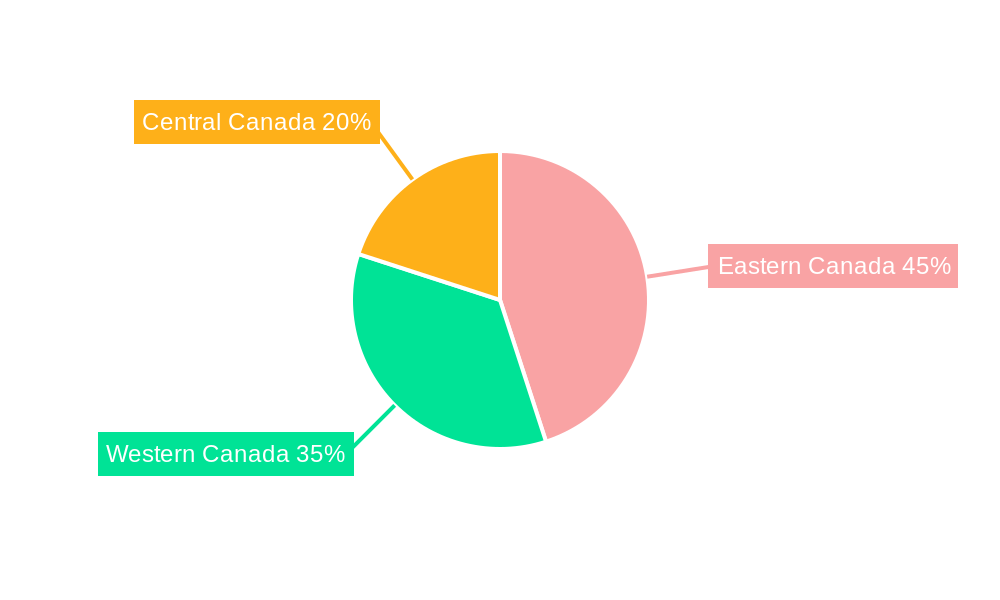
Canada Respiratory Devices Market Regional Market Share

Geographic Coverage of Canada Respiratory Devices Market
Canada Respiratory Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 11% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.2.1. Increasing Prevalence of Respiratory Disorders such as COPD and Asthma; Technological Advancements and Increasing Applications in the Homecare Setting
- 3.3. Market Restrains
- 3.3.1. High Cost of Devices
- 3.4. Market Trends
- 3.4.1. Sleep Test Devices Segment is Expected to Witness a Significant Growth Over the Forecast Period.
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Canada Respiratory Devices Market Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Diagnostic and Monitoring Devices
- 5.1.1. Spirometers
- 5.1.2. Sleep Test Devices
- 5.1.3. Peak Flow Meters
- 5.1.4. Pulse Oximeters
- 5.1.5. Capnographs
- 5.1.6. Other Diagnostic and Monitoring Devices
- 5.2. Market Analysis, Insights and Forecast - by Therapeutic Device
- 5.2.1. CPAP Devices
- 5.2.2. BiPAP Devices
- 5.2.3. Humidifiers
- 5.2.4. Nebulizers
- 5.2.5. Oxygen Concentrators
- 5.2.6. Ventilators
- 5.2.7. Inhalers
- 5.2.8. Other Therapeutic Devices
- 5.3. Market Analysis, Insights and Forecast - by Disposables
- 5.3.1. Masks
- 5.3.2. Breathing Circuit
- 5.3.3. Other Disposables
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. Canada
- 5.1. Market Analysis, Insights and Forecast - by Diagnostic and Monitoring Devices
- 6. Competitive Analysis
- 6.1. Market Share Analysis 2025
- 6.2. Company Profiles
- 6.2.1 Medtronic PLC
- 6.2.1.1. Overview
- 6.2.1.2. Products
- 6.2.1.3. SWOT Analysis
- 6.2.1.4. Recent Developments
- 6.2.1.5. Financials (Based on Availability)
- 6.2.2 Fisher & Paykel Healthcare Limited
- 6.2.2.1. Overview
- 6.2.2.2. Products
- 6.2.2.3. SWOT Analysis
- 6.2.2.4. Recent Developments
- 6.2.2.5. Financials (Based on Availability)
- 6.2.3 General Electronic Company (GE Healthcare)
- 6.2.3.1. Overview
- 6.2.3.2. Products
- 6.2.3.3. SWOT Analysis
- 6.2.3.4. Recent Developments
- 6.2.3.5. Financials (Based on Availability)
- 6.2.4 Gentige AB
- 6.2.4.1. Overview
- 6.2.4.2. Products
- 6.2.4.3. SWOT Analysis
- 6.2.4.4. Recent Developments
- 6.2.4.5. Financials (Based on Availability)
- 6.2.5 Hamilton Medical AG
- 6.2.5.1. Overview
- 6.2.5.2. Products
- 6.2.5.3. SWOT Analysis
- 6.2.5.4. Recent Developments
- 6.2.5.5. Financials (Based on Availability)
- 6.2.6 Becton Dickinson and Company
- 6.2.6.1. Overview
- 6.2.6.2. Products
- 6.2.6.3. SWOT Analysis
- 6.2.6.4. Recent Developments
- 6.2.6.5. Financials (Based on Availability)
- 6.2.7 Invacare Corporation
- 6.2.7.1. Overview
- 6.2.7.2. Products
- 6.2.7.3. SWOT Analysis
- 6.2.7.4. Recent Developments
- 6.2.7.5. Financials (Based on Availability)
- 6.2.8 Dragerwerk AG & Co KGaA
- 6.2.8.1. Overview
- 6.2.8.2. Products
- 6.2.8.3. SWOT Analysis
- 6.2.8.4. Recent Developments
- 6.2.8.5. Financials (Based on Availability)
- 6.2.9 Nihon Kohden Corporation
- 6.2.9.1. Overview
- 6.2.9.2. Products
- 6.2.9.3. SWOT Analysis
- 6.2.9.4. Recent Developments
- 6.2.9.5. Financials (Based on Availability)
- 6.2.10 Koninklijke Philips N V
- 6.2.10.1. Overview
- 6.2.10.2. Products
- 6.2.10.3. SWOT Analysis
- 6.2.10.4. Recent Developments
- 6.2.10.5. Financials (Based on Availability)
- 6.2.1 Medtronic PLC
List of Figures
- Figure 1: Canada Respiratory Devices Market Revenue Breakdown (undefined, %) by Product 2025 & 2033
- Figure 2: Canada Respiratory Devices Market Share (%) by Company 2025
List of Tables
- Table 1: Canada Respiratory Devices Market Revenue undefined Forecast, by Diagnostic and Monitoring Devices 2020 & 2033
- Table 2: Canada Respiratory Devices Market Volume K Units Forecast, by Diagnostic and Monitoring Devices 2020 & 2033
- Table 3: Canada Respiratory Devices Market Revenue undefined Forecast, by Therapeutic Device 2020 & 2033
- Table 4: Canada Respiratory Devices Market Volume K Units Forecast, by Therapeutic Device 2020 & 2033
- Table 5: Canada Respiratory Devices Market Revenue undefined Forecast, by Disposables 2020 & 2033
- Table 6: Canada Respiratory Devices Market Volume K Units Forecast, by Disposables 2020 & 2033
- Table 7: Canada Respiratory Devices Market Revenue undefined Forecast, by Region 2020 & 2033
- Table 8: Canada Respiratory Devices Market Volume K Units Forecast, by Region 2020 & 2033
- Table 9: Canada Respiratory Devices Market Revenue undefined Forecast, by Diagnostic and Monitoring Devices 2020 & 2033
- Table 10: Canada Respiratory Devices Market Volume K Units Forecast, by Diagnostic and Monitoring Devices 2020 & 2033
- Table 11: Canada Respiratory Devices Market Revenue undefined Forecast, by Therapeutic Device 2020 & 2033
- Table 12: Canada Respiratory Devices Market Volume K Units Forecast, by Therapeutic Device 2020 & 2033
- Table 13: Canada Respiratory Devices Market Revenue undefined Forecast, by Disposables 2020 & 2033
- Table 14: Canada Respiratory Devices Market Volume K Units Forecast, by Disposables 2020 & 2033
- Table 15: Canada Respiratory Devices Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 16: Canada Respiratory Devices Market Volume K Units Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Canada Respiratory Devices Market?
The projected CAGR is approximately 11%.
2. Which companies are prominent players in the Canada Respiratory Devices Market?
Key companies in the market include Medtronic PLC, Fisher & Paykel Healthcare Limited, General Electronic Company (GE Healthcare), Gentige AB, Hamilton Medical AG, Becton Dickinson and Company, Invacare Corporation, Dragerwerk AG & Co KGaA, Nihon Kohden Corporation, Koninklijke Philips N V.
3. What are the main segments of the Canada Respiratory Devices Market?
The market segments include Diagnostic and Monitoring Devices, Therapeutic Device, Disposables.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
Increasing Prevalence of Respiratory Disorders such as COPD and Asthma; Technological Advancements and Increasing Applications in the Homecare Setting.
6. What are the notable trends driving market growth?
Sleep Test Devices Segment is Expected to Witness a Significant Growth Over the Forecast Period..
7. Are there any restraints impacting market growth?
High Cost of Devices.
8. Can you provide examples of recent developments in the market?
May 2022: Honeywell launched two new NIOSH-certified respiratory offerings to help meet the needs of healthcare workers. The new products expand Honeywell's protective equipment portfolio for healthcare professionals, incorporating the company's decades of expertise in producing respiratory protection solutions.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K Units.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Canada Respiratory Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Canada Respiratory Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Canada Respiratory Devices Market?
To stay informed about further developments, trends, and reports in the Canada Respiratory Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


